458:, and cell-to-cell contact. The gap junctions in these tissues supported by endocrine signaling arbitrate intracellular signals between cells and larger organ systems by connecting adjacent cells to each other in a tight fit. The Tight fit of the gap junction is such that cells in the tissue can communicate more efficiently and maintain homeostasis. Thus the purpose of the gap junction is to regulate the passage of ions, nutrients, metabolites, second messengers, and small biological molecules. In diabetes the subsequent loss or degradation of Cx36 substantially inhibits insulin production in the pancreas and glucose in the liver which is vital for the production of energy for the entire body. A deficiency of Cx36 adversely affects the ability of the gap junction to operate within these tissues leading a reduction in function and possible disease. Similar symptoms associated with the loss or degradation of the gap junction have been observed in type II diabetes, however, the function of Cx36 in Type 1 and type II diabetes in humans is still unknown. Additionally, the Cx36 connexin is coded for by GJD2 gene, which has a predisposition on the gene locus for type II diabetes, and diabetic syndrome.
205:(ex: Connexin 43 is Cx 43 due to its molecular weight of 43 kDa). Connexons will form the gap junction by docking a hemi-channel to another hemi-channel in an adjacent cell membrane. During this phase, the formation of intercellular channels spanning both of the plasma membranes occurs. Subsequently, this process leads to a better understanding of how electric synapses are facilitated between neurons. Early research identified connexons through their presence in gap junctions. Since then, connexons have been increasingly detected forming channels in single membranes considerably broadening their functionality in cells and tissues.
413:, which is the inability of the body to produce insulin for glucose uptake by cells and degradation in the smaller units of connexons, called connexins, possibly leading to the onset of heart disease. Cardiovascular disease and diabetes, type I and II, affects similar locations within cells of the heart and pancreas. This location is the gap junction, where connexons facilitate rapid cell-to-cell interactions via electrical transmissions. Gap junctions are often present at nerve endings such as in cardiac muscle and are important in maintaining
29:
380:. The molecular mechanism as to how connexons play a role in the conditions listed above has yet to be fully understood and is under further research. Along with their key role in the CNS, connexons are crucial in the functioning of cardiac tissues. The direct connection allows for quick and synchronized firing of neurons in the heart which explains the ability for the heart to beat quickly and change its rate in response to certain stimuli. Connexons also play an essential role in cell development. Specifically, their role in
393:. Connexons cause changes in extracellular glucose concentrations affecting feeding/satiety behavior, sleep-wake cycles, and energy use. Further studies indicate that there is an increase in glucose uptake mediated by connexons (whose mechanism is still not fully understood) and under times of high stress and inflammation. Recent research also indicates that connexons may affect
425:
muscle cells of intercalated discs facilitating synchronized beating of the heart. In the occurrence of cardiovascular disease the Cx43 subunit begins to show signs of oxidative stress, the ability of the heart to counteract the buildup of harmful toxins due to age or diet leading to reduced vascular
384:
dealing with brain development as well as brain repair during certain diseases/pathologies and also assisting in both cell division as well as cell proliferation. The mechanism by which connexons aid in these processes is still being researched however, it is currently understood that this mechanism
323:
regulates the communication between channels in multiple ways by controlling: connexin trafficking from the Golgi
Apparatus, accumulation of connexons to certain areas, and degradation of unnecessary channels. The process of these actions is very complex but involvement of protein phosphorylation is
185:
Connexons contribute to the formation of gap junctions, and are an essential component of the electric synapses in neural pathways. In a single gap junction, connexons will assemble around an aqueous porous membrane, forming a hemi-channel that is composed of connexins. Connexins are the smaller
417:
in the liver and proper function of the kidneys. The gap junction itself is a structure that is a specialized transmembrane protein formed by a connexon hemichannel. Cardiovascular disease and possibly type I and II diabetes, are each associated with a major protein connexin that makes up the gap
449:
excretion and glucose-induced insulin release from gap junctions of the liver and pancreas. Homeostasis in the liver and pancreatic organs are supported by an intricate system of cellular interactions called endocrine signaling. The secretion of hormones into the blood stream to target distant
159:
The assembly of connexins destined for gap junction plaques begins with synthesis of connexins within the cell and ends with the formation of gap junction channel plaques on the cell membrane. The connexin subunit proteins that make up connexons are synthesized on the membranes of the cell's
430:, are present in structural heart disease. However, the mechanisms of Cx43 in the heart are still poorly understood. Overall, these changes in Cx43 expression and oxidant stress can lead to abnormalities in the coordinated beating of the heart, predisposing it to cardiac
363:
Connexons play an imperative role in behavior and neurophysiology. Many of the details surrounding their pathological functions remain unknown as research has only begun recently. In the central nervous system (CNS), connexons play a major role in conditions such as
122:
supplied by a cell on one side of the junction; two connexons from opposing cells normally come together to form the complete intercellular gap junction channel. In some cells, the hemichannel itself is active as a conduit between the cytoplasm and the
172:. The connexons are then delivered to their proper location on the plasma membrane. Connexons then dock with compatible connexons from the neighboring cell to form gap junction channel plaques. A large part of this process is mediated by
213:
Connexon structure is degraded by its removal from the plasma membrane. Connexons will be internalized by the cell itself as a double membrane channel structure (due to the docking of hemi-channels). This is called internalization or
176:
of different enzymes and proteins, allowing and preventing interaction between certain proteins. The connexons forming channels to the cell exterior or in mitochondria will require a somewhat altered path of assembly.
301:– one common type of chemical modulation is through the interaction of Ca and certain domains of connexins. It is not completely understood, however, it is suggested that this interaction causes Ca to block the
263:
and selectivity of the channels is determined by its width as well as the molecular selectivity of connexins such as charge selectivity. Research shows connexons are particularly permeable to
389:(form of extracellular signaling mediated by purine nucleotides and nucleosides such as adenosine and ATP) and permeability to ATP. Other important roles of connexons are glucose sensing and
127:, allowing the transference of ions and small molecules lower than 1-2 KDa. Little is known about this function of connexons besides the new evidence suggesting their key role in
282:
between the interiors of the two cells. Gates can also show voltage sensitivity depending on the difference in voltage from the interior and exterior of the cell (i.e.
450:
organs. However, endocrine signaling in the pancreas and liver operates along short distances in the cellular membrane by way of signaling pathways, ion channels,
278:, ions and glucose. Channels are also voltage sensitive. The connexon channels have voltage-dependent gates that open or close depending on the difference in
218:. Research suggests that gap junctions in general may be internalized using more than one method, but the best known and most studied would be that of
426:
functions. Additionally, reduced Cx43 expression in vascular tissue, which plays a part in ventricular remolding and healing of wounds after a
1010:
186:
protein molecules that make up connexons and play a crucial part to the formation of gap junctions. Structurally, connexins are made up of 4
354:
down neurons. These types of gap-junctions with this type of modulation are often found in neurons in cardiac tissue and vertebrate retina.
421:
In cardiovascular disease, Cx43 (connexin 43), a subunit of a connexon, is a general protein of the gap junction stimulating cardio
979:
766:
Lauf, Undine; Giepmans, Ben N. G.; Lopez, Patricia; Braconnot, Sébastien; Chen, Shu-Chih; Falk, Matthias M. (6 August 2002).
305:
of the channel. Another form of chemical modulation is through the response of the channel to acidification (decrease of
131:. In still other cells connexons have been shown to occur in mitochondrial membranes and appear to play a role in heart
917:"Reduced expression of Cx43 attenuates ventricular remodeling after myocardial infarction via impaired TGF-β signaling"
1003:
768:"Dynamic trafficking and delivery of connexons to the plasma membrane and accretion to gap junctions in living cells"
259:
The properties of individual connexin proteins determine the overall properties of the whole connexon channel. The
231:
294:
Communication between gap-junctions can be modulated/regulated in many ways. The main types of modulation are:
219:
601:"Mitochondrial connexin43 as a new player in the pathophysiology of myocardial ischaemia-reperfusion injury"
996:
599:
Ruiz-Meana, M.; Rodríguez-Sinovas, A.; Cabestrero, A.; Boengler, K.; Heusch, G.; Garcia-Dorado, D. (2008).
556:
Herve, Jean-Claude; Derangeon, Mickael (2012-09-01). "Gap-junction-mediated cell-to-cell communication".
451:
238:
pathways. Lysosomes are able to break down the proteins of the connexon because they contain specific
66:
1237:
455:
61:
118:. This channel allows for bidirectional flow of ions and signaling molecules. The connexon is the
128:
1037:
406:
73:
201:
reside intracellularly. Connexin types can be further differentiated by using their predicted
427:
330:– humoral modulation of gap junction communication is done through many biomolecules such as
260:
190:
161:
719:"Proteins and Mechanisms Regulating Gap-Junction Assembly, Internalization, and Degradation"
386:
8:
915:
Zhang, Yan; Wang, Hongtao; Kovacs, Attila; Kanter, Evelyn; Yamada, Kathryn (2010-02-01).
394:
390:
339:
124:
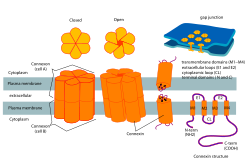
1232:
941:
916:
889:
864:
743:
718:
677:
650:
581:
512:
485:
283:
794:
767:
1041:
975:
946:
894:
840:
799:
748:
682:
622:
573:
517:
377:
351:
331:
310:
267:
1118:
936:
928:
884:
876:
830:
789:
779:
738:
730:
672:
662:
612:
565:
507:
497:
442:
306:
302:
226:
binding to a receptor signaling for a certain part of the membrane to be coated in
585:
28:
835:
818:
438:
320:
173:
169:
932:
1172:
1122:
734:
347:
335:
243:
202:
194:
115:
569:
1226:
1163:
1150:
1029:
969:
598:
502:
309:). It has been found that intracellular acidification causes a change in the
187:
1158:
1089:
1071:
950:
898:
844:
803:
784:
752:
686:
626:
577:
521:
381:
373:
107:
667:
193:
connected by two extracellular loops and one cytoplasmic loop, while both
1206:
1202:
1103:
1098:
1067:
988:
617:
600:
431:
414:
343:
275:
271:
215:
143:
119:
1197:
1167:
1108:
1058:
1020:
198:
79:
1177:
1093:
1053:
880:
235:
139:
111:
1137:
1132:
1080:
1046:
410:
369:
365:
227:
165:
132:
103:
234:. Now present in the cell membrane, connexons will be degraded by
16:
Protein hexamer that forms the pore of gap junctions between cells
1127:
446:
422:
279:
264:
239:
91:
967:
921:
American
Journal of Physiology. Heart and Circulatory Physiology
242:
that are made specifically for this process. It is thought that
1193:
649:
Wright, Josephine; Richards, Toby; Becker, David (2012-03-01).
223:
484:
Cheung, Giselle; Chever, Oana; Rouach, Nathalie (2014-11-04).
230:. This part of the membrane then buds into the cell forming a
168:, or combined with other smaller parts, into connexons in the
49:
138:
Connexons made of the same type of connexins are considered
1210:
142:, while connexons made of differing types of connexins are
765:
865:"Oxidant stress derails the cardiac connexon connection"
486:"Connexons and Pannexons: Newcomers in Neurophysiology"
350:
work in neuronal gap-junctions causing propagation of
914:
819:"The communicating junctions, roles and dysfunctions"
397:, learning, memory, vision, and sensorimotor gating.
313:
of connexins which then reduces the channel activity.
648:
405:Some of the diseases associated with connexons are
823:Biochimica et Biophysica Acta (BBA) - Biomembranes
483:
1224:
772:Proceedings of the National Academy of Sciences
555:
445:diabetes. Cx36 (connexin 36) subunit mediates
1004:
222:. In simple terms this process consists of a
858:
856:
854:
1018:
1011:
997:
712:
710:
708:
706:
704:
702:
700:
698:
696:
27:
968:Andrew L Harris and Darren Locke (2009).
940:
888:
862:
834:
793:
783:
742:
676:
666:
616:
511:
501:
851:
716:
644:
642:
640:
638:
636:
551:
479:
477:
475:
473:
471:
437:Connexons are also associated with both
102:, is an assembly of six proteins called
910:
908:
693:
549:
547:
545:
543:
541:
539:
537:
535:
533:
531:
1225:
992:
816:
633:
468:
249:
246:signals degradation within the cell.
905:
717:Thevenin, Anastasia F (2013-03-07).
528:
358:
974:. New York: Springer. p. 574.
863:Tomaselli, Gordon F. (2010-12-04).
400:
13:
961:
490:Frontiers in Cellular Neuroscience
14:
1249:
869:Journal of Clinical Investigation
655:Cardiology Research and Practice
33:Connexon and connexin structure
810:
759:
592:
208:
1:
461:
289:
254:
220:clathrin-mediated endocytosis
836:10.1016/j.bbamem.2012.10.012
342:. Neurotransmitters such as
149:
7:
933:10.1152/ajpheart.00806.2009
817:Hervé, Jean-Claude (2013).
452:G-protein coupled receptors
154:
10:
1254:
735:10.1152/physiol.00038.2012
180:
164:. These subunits are then
1186:
1148:
1027:
570:10.1007/s00441-012-1485-6
456:tyrosine-kinase receptors
106:that form the pore for a
72:
60:
48:
43:
38:
26:
21:
651:"Connexins And Diabetes"
558:Cell and Tissue Research
503:10.3389/fncel.2014.00348
1038:Cell adhesion molecules
605:Cardiovascular Research
317:Protein Phosphorylation
129:intracellular signaling
785:10.1073/pnas.162055899
407:cardiovascular disease
74:Anatomical terminology
428:myocardial infarction
191:transmembrane domains
162:endoplasmic reticulum
387:purinergic signaling
100:connexin hemichannel
778:(16): 10446–10451.
668:10.1155/2012/496904
395:synaptic plasticity
391:signal transduction
340:bioactive compounds
125:extracellular space
971:Connexins, A Guide
618:10.1093/cvr/cvm062
284:membrane potential
250:Cellular functions
98:, also known as a
67:H1.00.01.1.02025
1220:
1219:
1119:membrane proteins
1042:Adherens junction
981:978-1-934115-46-6
378:neurodegeneration
359:Overall functions
352:action potentials
332:neurotransmitters
311:C-terminal domain
268:second messengers
88:
87:
83:
1245:
1238:Membrane biology
1013:
1006:
999:
990:
989:
985:
955:
954:
944:
912:
903:
902:
892:
881:10.1172/jci41780
860:
849:
848:
838:
814:
808:
807:
797:
787:
763:
757:
756:
746:
714:
691:
690:
680:
670:
646:
631:
630:
620:
596:
590:
589:
553:
526:
525:
515:
505:
481:
401:Related diseases
307:intracellular pH
203:molecular weight
114:of two adjacent
80:edit on Wikidata
77:
31:
19:
18:
1253:
1252:
1248:
1247:
1246:
1244:
1243:
1242:
1223:
1222:
1221:
1216:
1182:
1144:
1023:
1017:
982:
964:
962:Further reading
959:
958:
913:
906:
861:
852:
815:
811:
764:
760:
715:
694:
647:
634:
597:
593:
554:
529:
482:
469:
464:
403:
361:
321:phosphorylation
292:
257:
252:
211:
183:
174:phosphorylation
170:golgi apparatus
157:
152:
84:
34:
17:
12:
11:
5:
1251:
1241:
1240:
1235:
1218:
1217:
1215:
1214:
1200:
1190:
1188:
1184:
1183:
1181:
1180:
1175:
1173:Focal adhesion
1170:
1161:
1155:
1153:
1146:
1145:
1143:
1142:
1141:
1140:
1135:
1130:
1123:Tight junction
1114:
1113:
1112:
1111:
1106:
1101:
1086:
1085:
1084:
1083:
1064:
1063:
1062:
1061:
1051:
1050:
1049:
1034:
1032:
1025:
1024:
1016:
1015:
1008:
1001:
993:
987:
986:
980:
963:
960:
957:
956:
927:(2): H477-87.
904:
850:
809:
758:
692:
632:
611:(2): 325–333.
591:
527:
466:
465:
463:
460:
402:
399:
360:
357:
356:
355:
348:norepinephrine
338:, and various
336:growth factors
325:
314:
291:
288:
256:
253:
251:
248:
244:ubiquitination
210:
207:
182:
179:
156:
153:
151:
148:
86:
85:
76:
70:
69:
64:
58:
57:
52:
46:
45:
41:
40:
36:
35:
32:
24:
23:
15:
9:
6:
4:
3:
2:
1250:
1239:
1236:
1234:
1231:
1230:
1228:
1212:
1208:
1204:
1201:
1199:
1195:
1192:
1191:
1189:
1185:
1179:
1176:
1174:
1171:
1169:
1165:
1164:Hemidesmosome
1162:
1160:
1157:
1156:
1154:
1152:
1147:
1139:
1136:
1134:
1131:
1129:
1126:
1125:
1124:
1120:
1116:
1115:
1110:
1107:
1105:
1102:
1100:
1097:
1096:
1095:
1091:
1088:
1087:
1082:
1079:
1078:
1077:
1073:
1069:
1066:
1065:
1060:
1057:
1056:
1055:
1052:
1048:
1045:
1044:
1043:
1039:
1036:
1035:
1033:
1031:
1026:
1022:
1014:
1009:
1007:
1002:
1000:
995:
994:
991:
983:
977:
973:
972:
966:
965:
952:
948:
943:
938:
934:
930:
926:
922:
918:
911:
909:
900:
896:
891:
886:
882:
878:
874:
870:
866:
859:
857:
855:
846:
842:
837:
832:
828:
824:
820:
813:
805:
801:
796:
791:
786:
781:
777:
773:
769:
762:
754:
750:
745:
740:
736:
732:
729:(2): 93–116.
728:
724:
720:
713:
711:
709:
707:
705:
703:
701:
699:
697:
688:
684:
679:
674:
669:
664:
660:
656:
652:
645:
643:
641:
639:
637:
628:
624:
619:
614:
610:
606:
602:
595:
587:
583:
579:
575:
571:
567:
563:
559:
552:
550:
548:
546:
544:
542:
540:
538:
536:
534:
532:
523:
519:
514:
509:
504:
499:
495:
491:
487:
480:
478:
476:
474:
472:
467:
459:
457:
453:
448:
444:
440:
435:
433:
429:
424:
419:
416:
412:
408:
398:
396:
392:
388:
383:
379:
375:
371:
367:
353:
349:
345:
341:
337:
333:
329:
326:
322:
318:
315:
312:
308:
304:
300:
297:
296:
295:
287:
285:
281:
277:
273:
269:
266:
262:
247:
245:
241:
237:
233:
229:
225:
221:
217:
206:
204:
200:
196:
192:
189:
188:alpha helical
178:
175:
171:
167:
163:
147:
145:
141:
136:
134:
130:
126:
121:
117:
113:
109:
105:
101:
97:
93:
81:
75:
71:
68:
65:
63:
59:
56:
53:
51:
47:
42:
37:
30:
25:
20:
1159:Basal lamina
1090:Cytoskeleton
1075:
1072:Gap junction
1068:Ion channels
1019:Proteins of
970:
924:
920:
875:(1): 87–89.
872:
868:
826:
822:
812:
775:
771:
761:
726:
722:
658:
654:
608:
604:
594:
564:(1): 21–31.
561:
557:
493:
489:
436:
420:
404:
382:neurogenesis
374:inflammation
362:
327:
316:
298:
293:
261:permeability
258:
212:
184:
166:oligomerized
158:
137:
110:between the
108:gap junction
99:
95:
89:
54:
1207:Stereocilia
1151:cell–matrix
1104:Plakoglobin
1099:Desmoplakin
432:arrhythmias
415:homeostasis
344:epinephrine
276:nucleotides
272:amino acids
216:endocytosis
209:Degradation
199:C terminals
144:heteromeric
120:hemichannel
44:Identifiers
1227:Categories
1203:Microvilli
1198:Kinocilium
1168:Tonofibril
1109:Tonofibril
1059:Desmoglein
1021:epithelium
829:(1): 1–3.
723:Physiology
661:: 496904.
462:References
418:junction.
319:– protein
290:Modulation
255:Properties
1233:Cytoplasm
1178:Costamere
1094:Desmosome
1054:Desmosome
1030:cell–cell
385:involves
236:lysosomal
150:Structure
140:homomeric
133:ischaemia
112:cytoplasm
104:connexins
55:connexona
1138:MARVELD2
1133:Occludin
1081:Connexin
1076:Connexon
1047:Cadherin
1028:Lateral/
951:19966054
899:20038808
845:23088917
804:12149451
753:23455769
687:22536530
627:18006437
578:22940728
522:25408635
411:diabetes
370:ischemia
366:epilepsy
299:Chemical
228:clathrin
155:Assembly
96:connexon
22:Connexon
1128:Claudin
942:2822575
890:2798705
744:3768091
678:3303578
513:4219455
496:: 348.
447:insulin
443:Type II
423:myocyte
328:Humoral
280:voltage
265:soluble
240:enzymes
232:vesicle
181:General
92:biology
39:Details
1187:Apical
1149:Basal/
1117:other
978:
949:
939:
897:
887:
843:
802:
795:124935
792:
751:
741:
685:
675:
625:
586:176666
584:
576:
520:
510:
439:Type I
376:, and
324:known.
224:ligand
1194:Cilia
582:S2CID
116:cells
78:[
50:Latin
1211:STRC
976:ISBN
947:PMID
895:PMID
841:PMID
827:1828
800:PMID
749:PMID
683:PMID
659:2012
623:PMID
574:PMID
518:PMID
441:and
409:and
346:and
303:pore
197:and
94:, a
937:PMC
929:doi
925:298
885:PMC
877:doi
873:120
831:doi
790:PMC
780:doi
739:PMC
731:doi
673:PMC
663:doi
613:doi
566:doi
562:352
508:PMC
498:doi
286:).
90:In
1229::
1121::
1092::
1070::
1040::
945:.
935:.
923:.
919:.
907:^
893:.
883:.
871:.
867:.
853:^
839:.
825:.
821:.
798:.
788:.
776:99
774:.
770:.
747:.
737:.
727:28
725:.
721:.
695:^
681:.
671:.
657:.
653:.
635:^
621:.
609:77
607:.
603:.
580:.
572:.
560:.
530:^
516:.
506:.
492:.
488:.
470:^
454:,
434:.
372:,
368:,
334:,
274:,
270:,
146:.
135:.
62:TH
1213:)
1209:(
1205:/
1196:/
1166:/
1074:/
1012:e
1005:t
998:v
984:.
953:.
931::
901:.
879::
847:.
833::
806:.
782::
755:.
733::
689:.
665::
629:.
615::
588:.
568::
524:.
500::
494:8
195:N
82:]
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.