38:
1610:
1619:
1628:
1281:
60:
51:
1646:
2479:
1092:
1637:
1655:
588:, which is close to (14 − pH), so the pOH of pure water is also close to 7. Addition of a base to water will reduce the hydrogen cation concentration and therefore increase the hydroxide ion concentration (decrease pH, increase pOH) even if the base does not itself contain hydroxide. For example,
1350:
In mildly acidic solutions, the hydroxo/hydroxido complexes formed by aluminium are somewhat different from those of boron, reflecting the greater size of Al(III) vs. B(III). The concentration of the species is very dependent on the total aluminium concentration. Various other hydroxo complexes are
1588:
Hydrolysis of Pb in aqueous solution is accompanied by the formation of various hydroxo-containing complexes, some of which are insoluble. The basic hydroxo complex is a cluster of six lead centres with metal–metal bonds surrounding a central oxide ion. The six hydroxide groups lie on the faces of
2229:
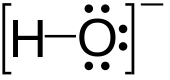
are built up of double layers of hydroxide ions – the aluminium atoms on two-thirds of the octahedral holes between the two layers – and differ only in the stacking sequence of the layers. The structures are similar to the brucite structure. However, whereas the brucite structure can be
1584:
is formed. When solutions containing this ion are acidified, the ion is formed together with some basic hydroxo complexes. The structure of has a triangle of tin atoms connected by bridging hydroxide groups. Tin(IV) hydroxide is unknown but can be regarded as the hypothetical acid from which
1888:
usually have the metal in the +2 (M = Mn, Fe, Co, Ni, Cu, Zn) or +3 (M = Fe, Ru, Rh, Ir) oxidation state. None are soluble in water, and many are poorly defined. One complicating feature of the hydroxides is their tendency to undergo further condensation to the oxides, a process called
1228:
1100:
616:
2242:). The O–H–O distance is very short, at 265 pm; the hydrogen is not equidistant between the oxygen atoms and the short OH bond makes an angle of 12° with the O–O line. A similar type of hydrogen bond has been proposed for other amphoteric hydroxides, including Be(OH)
2230:
described as a close-packed structure in gibbsite the OH groups on the underside of one layer rest on the groups of the layer below. This arrangement led to the suggestion that there are directional bonds between OH groups in adjacent layers. This is an unusual form of
1347:, pure aluminium hydroxide is made to precipitate by reducing the temperature and adding water to the extract, which, by diluting the alkali, lowers the pH of the solution. Basic aluminium hydroxide AlO(OH), which may be present in bauxite, is also amphoteric.
834:, an M−OH bending mode can be observed. For example, in it occurs at 1065 cm. The bending mode for a bridging hydroxide tends to be at a lower frequency as in (955 cm). M−OH stretching vibrations occur below about 600 cm. For example, the
2166:
structure, which gradually freezes in a monoclinically distorted sodium chloride structure at temperatures below about 300 °C. The OH groups still rotate even at room temperature around their symmetry axes and, therefore, cannot be detected by
821:
is a consequence of the small mass of the hydrogen atom as compared to the mass of the oxygen atom, and this makes detection of hydroxyl groups by infrared spectroscopy relatively easy. A band due to an OH group tends to be sharp. However, the
961:
Although the base strength of sodium carbonate solutions is lower than a concentrated sodium hydroxide solution, it has the advantage of being a solid. It is also manufactured on a vast scale (42 million tonnes in 2005) by the
826:
increases when the OH group is involved in hydrogen bonding. A water molecule has an HOH bending mode at about 1600 cm, so the absence of this band can be used to distinguish an OH group from a water molecule.
771:, donating one pair of electrons to each of the atoms being bridged. As illustrated by , metal hydroxides are often written in a simplified format. It can even act as a 3-electron-pair donor, as in the tetramer
1940:
Numerous mixed ligand complexes of these metals with the hydroxide ion exist. In fact, these are in general better defined than the simpler derivatives. Many can be made by deprotonation of the corresponding
1243:, is an acid. Unlike the hydroxides of the alkali and alkaline earth hydroxides, it does not dissociate in aqueous solution. Instead, it reacts with water molecules acting as a Lewis acid, releasing protons.
3667:
Jacobs, H.; Kockelkorn, J.; Tacke, Th. (1985). "Hydroxide des
Natriums, Kaliums und Rubidiums: Einkristallzüchtung und röntgenographische Strukturbestimmung an der bei Raumtemperatur stabilen Modifikation".
684:(114 pm). In aqueous solution the hydroxide ion forms strong hydrogen bonds with water molecules. A consequence of this is that concentrated solutions of sodium hydroxide have high
2019:
A. Some metals, e.g. V, Cr, Nb, Ta, Mo, W, tend to exist in high oxidation states. Rather than forming hydroxides in aqueous solution, they convert to oxo clusters by the process of
2051:
cation in which there is a square of Zr ions with two hydroxide groups bridging between Zr atoms on each side of the square and with four water molecules attached to each Zr atom.
1777:-periodic acid can lose all its protons, eventually forming the periodate ion . It can also be protonated in strongly acidic conditions to give the octahedral ion , completing the
2158:
The hydroxide ion appears to rotate freely in crystals of the heavier alkali metal hydroxides at higher temperatures so as to present itself as a spherical ion, with an effective
1343:, but other hydroxides usually present in the mineral, such as iron hydroxides, do not dissolve because they are not amphoteric. After removal of the insolubles, the so-called
1331:
minerals this equilibrium is manipulated by careful control of temperature and alkali concentration. In the first phase, aluminium dissolves in hot alkaline solution as
5085:
5094:
4378:
4350:
5062:
4388:
4369:
4341:
3265:
Alderighi, L; Dominguez, S.; Gans, P.; Midollini, S.; Sabatini, A.; Vacca, A. (2009). "Beryllium binding to adenosine 5'-phosphates in aqueous solution at 25°C".
5053:
2370:. The base abstracts a proton from a weak acid to give an intermediate that goes on to react with another reagent. Common substrates for proton abstraction are
608:, which decreases the hydrogen cation concentration, which increases the hydroxide ion concentration. pOH can be kept at a nearly constant value with various
1549:) have been characterized. These acids also have hydroxide groups attached to the silicon; the formulas suggest that these acids are protonated forms of poly
3559:
Fortman, George C.; Slawin, Alexandra M. Z.; Nolan, Steven P. (2010). "A Versatile
Cuprous Synthon: (IPr = 1,3 bis(diisopropylphenyl)imidazol-2-ylidene)".
3477:
M. N. Sokolov; E. V. Chubarova; K. A. Kovalenko; I. V. Mironov; A. V. Virovets; E. Peresypkina; V. P. Fedin (2005). "Stabilization of tautomeric forms P(OH)
2937:
Marx, D.; Chandra, A; Tuckerman, M.E. (2010). "Aqueous Basic
Solutions: Hydroxide Solvation, Structural Diffusion, and Comparison to the Hydrated Proton".
2798:
2035:
In some cases, the products of partial hydrolysis of metal ion, described above, can be found in crystalline compounds. A striking example is found with
1351:
found in crystalline compounds. Perhaps the most important is the basic hydroxide AlO(OH), a polymeric material known by the names of the mineral forms
2039:(IV). Because of the high oxidation state, salts of Zr are extensively hydrolyzed in water even at low pH. The compound originally formulated as ZrOCl
4636:
3055:
2218:
1140:
products, including the trimeric ion , which has OH groups bridging between pairs of beryllium ions making a 6-membered ring. At very low pH the
2186:
The hydroxide ion displays cylindrical symmetry in hydroxides of divalent metals Ca, Cd, Mn, Fe, and Co. For example, magnesium hydroxide Mg(OH)
4209:
3967:
2466:
for self-dissociation of ethanol is about 16, so the alkoxide ion is a strong enough base. The addition of an alcohol to an aldehyde to form a
1167:
is a strong base (up to the limit of its solubility, which is very low in pure water), as are the hydroxides of the heavier alkaline earths:
584:
concentration; the pH of pure water is close to 7 at ambient temperatures. The concentration of hydroxide ions can be expressed in terms of
2183:
units. This is consistent with the weakly basic character of LiOH in solution, indicating that the Li–OH bond has much covalent character.
264:
1901:
O). Copper(I) and gold(I) hydroxides are also unstable, although stable adducts of CuOH and AuOH are known. The polymeric compounds M(OH)
1746:
632:
3363:
730:
for this reaction can be specified either as a reaction with dissolved carbon dioxide or as a reaction with carbon dioxide gas (see
639:
by donating a pair of electrons to a Lewis acid. In aqueous solution both hydrogen and hydroxide ions are strongly solvated, with
2470:
is an example of a reaction that can be catalyzed by the presence of hydroxide. Hydroxide can also act as a Lewis-base catalyst.
2802:
2576:
the hydroxide anion can be shuttled into an organic solvent as well, for example in the generation of the reactive intermediate
1009:
Aside from NaOH and KOH, which enjoy very large scale applications, the hydroxides of the other alkali metals also are useful.
2234:
since the two hydroxide ion involved would be expected to point away from each other. The hydrogen atoms have been located by
1800:
As is common outside of the alkali metals, hydroxides of the elements in lower oxidation states are complicated. For example,
4104:
4085:
4066:
4040:
4021:
3886:
3836:
3159:
3124:
3012:
1909:
are in general prepared by increasing the pH of an aqueous solutions of the corresponding metal cations until the hydroxide
4135:
2986:
664:
has been characterized in the solid state. This compound is centrosymmetric and has a very short hydrogen bond (114.5
767:. It is also often found in mixed-ligand complexes of the type , where L is a ligand. The hydroxide ion often serves as a
3988:
2561:
3189:
1980:
1785:= −2, −1, 0, +1. Other acids of iodine(VII) that contain hydroxide groups are known, in particular in salts such as the
3072:
2565:
2440:
The hydroxide ion by itself is not a strong enough base, but it can be converted in one by adding sodium hydroxide to
3856:
3755:
3654:
3594:
3411:
3337:
3252:
2745:
2710:
242:
2226:
3350:
Böhm, Stanislav; Antipova, Diana; Kuthan, Josef (1997). "A study of methanetetraol dehydration to carbonic acid".
842:-active, polarized) and 420 cm (infrared). The same ion has a (HO)–Zn–(OH) bending vibration at 300 cm.
390:
5103:
1141:
3214:
88:
17:
3088:
1717:
there are oxoacids in which the central atom is attached to oxide ions and hydroxide ions. Examples include
219:
2437:
value not less than about 4 log units smaller, or the equilibrium will lie almost completely to the left.
635:
sense as it can accept a proton from a Brønsted–Lowry acid to form a water molecule. It can also act as a
4896:
3485:
and their derivatives by coordination to palladium and nickel atoms in heterometallic clusters with the
572:
of hydroxide ions in pure water is close to 10 mol∙dm, to satisfy the equal charge constraint. The
3190:"The Behavior and Capabilities of Lithium Hydroxide Carbon Dioxide Scrubbers in a Deep Sea Environment"
2460:
2387:
1014:
944:
4975:
4955:
4916:
2282:
3964:
5187:
5120:
5015:
4906:
4566:
4128:
2163:
1893:. Hydroxides of metals in the +1 oxidation state are also poorly defined or unstable. For example,
1742:
779:
528:
457:
433:
3851:
Ouellette, R.J. and Rawn, J.D. "Organic
Chemistry" 1st Ed. Prentice-Hall, Inc., 1996: New Jersey.
2993:
A comprehensive database of published data on equilibrium constants of metal complexes and ligands
2832:
5030:
4945:
4926:
4739:
4684:
4550:
4490:
4407:
4397:
4331:
2573:
1368:
3151:
3145:
2651:
In aqueous solution the ligands L are water molecules, but they may be replaced by other ligands
2102:. In this case the composition is nearer to that of the hydroxide than that of the chloride CuCl
5005:
4985:
4965:
4936:
4579:
4480:
4359:
3984:
3116:
3110:
2827:
2146:
because of its opaque quality, though its use is now restricted because it can be a source for
2071:
3878:
3646:
3329:
3244:
3141:
734:
for values and details). At neutral or acid pH, the reaction is slow, but is catalyzed by the
5040:
4995:
4877:
4735:
4725:
4607:
4058:
3747:
3208:
3106:
1885:
1384:
1372:
752:
in glass are acting as acids. Basic hydroxides, whether solids or in solution, are stored in
78:
3870:
3586:
143:
4886:
4460:
4446:
4436:
3947:
3781:
3721:
2819:
2610:
2569:
831:
727:
551:
469:
228:
119:
1872:. When these acids are neutralized the hydrogen atom is removed from the hydroxide group.
1585:
stannates, with a formula , are derived by reaction with the (Lewis) basic hydroxide ion.
8:
5177:
4867:
4715:
4528:
4456:
4426:
4314:
4264:
4254:
4179:
4158:
4121:
2557:
2371:
2343:
2235:
1290:
1172:
1160:
1109:
1072:
890:
818:
465:
323:
183:
109:
3951:
3785:
3725:
3476:
2823:
1155:
The solubility in water of the other hydroxides in this group increases with increasing
163:
5182:
4789:
4758:
4749:
4523:
4417:
4294:
4218:
4199:
3523:
3282:
2853:
2597:
2172:
1942:
1749:
acid. Many oxoacids of sulfur are known and all feature OH groups that can dissociate.
1609:
1392:
1256:
1125:
839:
823:
738:
581:
778:
When bound to a strongly electron-withdrawing metal centre, hydroxide ligands tend to
4768:
4666:
4626:
4617:
4597:
4540:
4319:
4304:
4274:
4239:
4174:
4100:
4081:
4062:
4036:
4017:
3919:
3882:
3871:
3852:
3832:
3751:
3695:
Enoki, Toshiaki; Tsujikawa, Ikuji (1975). "Magnetic
Behaviours of a Random Magnet, Ni
3650:
3590:
3407:
3367:
3333:
3248:
3155:
3120:
3068:
3008:
2954:
2911:
2861:
2845:
2810:
2621:
pOH signifies the minus the logarithm to base 10 of , alternatively the logarithm of
2355:
2168:
2087:
1869:
1561:
1360:
1168:
1010:
806:
689:
493:
3527:
3286:
1231:
Aluminium hydrolysis as a function of pH. Water molecules attached to Al are omitted
5130:
5071:
4794:
4671:
4592:
4513:
4284:
4249:
4050:
3955:
3911:
3789:
3729:
3677:
3568:
3515:
3359:
3274:
3060:
2946:
2919:
2857:
2837:
2725:
2675:
2347:
2075:
1894:
1881:
1861:
1801:
1664:
1364:
1176:
1068:
1067:
The hydroxide of lithium is preferred to that of sodium because of its lower mass.
901:
850:
810:
628:
624:
481:
473:
461:
453:
437:
410:
360:
284:
3064:
4656:
3971:
3902:
Denmark, S.E.; Beutne, G.L. (2008). "Lewis Base
Catalysis in Organic Synthesis".
2990:
2887:
2771:
2669:
2577:
2495:
2024:
1718:
1669:
768:
609:
429:
2983:
2888:
Kamal Abu-Dari; Kenneth N. Raymond; Derek P. Freyberg (1979). "The bihydroxide (
2796:
2175:
structure. LiOH, however, has a layered structure, made up of tetrahedral Li(OH)
2162:
of about 153 pm. Thus, the high-temperature forms of KOH and NaOH have the
1618:
208:
4470:
3939:
2538:
2400:
2198:
layer structure, with a kind of close-packing of magnesium and hydroxide ions.
2195:
2147:
1914:
1444:
1040:
963:
954:
870:
696:
695:
In solution, exposed to air, the hydroxide ion reacts rapidly with atmospheric
508:
406:
384:
330:
318:
3558:
3519:
3278:
1913:
out of solution. On the converse, the hydroxides dissolve in acidic solution.
1627:
782:
into oxide ligands. For example, the bichromate ion dissociates according to
5171:
4646:
4500:
3681:
3371:
2487:
2367:
2231:
1778:
1752:
1730:
1688:
1679:
1674:
1569:
1479:
has been identified in very dilute aqueous solution. It is a weak acid with p
1448:
1447:
is also known as carbonic anhydride, meaning that it forms by dehydration of
1324:
1277:
of boron are known, which, in the protonated form, contain hydroxide groups.
1156:
1024:
989:
882:
814:
731:
640:
569:
512:
485:
477:
425:
352:
3868:
2841:
2775:
2767:
2763:
1280:
3923:
3915:
3794:
3769:
3264:
2958:
2849:
2542:
2411:
2159:
1996:
1969:
1466:
1076:
971:
967:
996:, are among the principal ores used for the manufacture of metallic iron.
37:
2483:
2383:
2202:
2119:
2111:
1910:
1645:
1080:
835:
700:
577:
489:
445:
3733:
3317:
2923:
984:, is the principal ore from which the metal is manufactured. Similarly,
4229:
4189:
3364:
10.1002/(SICI)1097-461X(1997)62:3<315::AID-QUA10>3.0.CO;2-8
2665:
2467:
2123:
2048:
1693:
1240:
1137:
1117:
1036:
1028:
905:
669:
636:
305:
174:
3572:
2950:
2642:
In this context proton is the term used for a solvated hydrogen cation
970:(another name for sodium carbonate) acts on insoluble esters, such as
896:
Solutions containing the hydroxide ion are generated when a salt of a
3959:
2522:
hydrolysis under alkaline conditions (also known as base hydrolysis)
2115:
2091:
2055:
2036:
2016:
1714:
1706:
1702:
1557:
1306:
1208:
1184:
1180:
1149:
1121:
1032:
981:
897:
878:
858:
764:
685:
665:
535:
500:
59:
688:
due to the formation of an extended network of hydrogen bonds as in
383:
Except where otherwise noted, data are given for materials in their
2572:. The reaction medium for KOH and NaOH is usually water but with a
2499:
2478:
2456:
2239:
2222:
2214:
2210:
1992:
1865:
1824:
1594:
1550:
1469:
is the name given to a variety of compounds with a generic formula
1356:
1352:
1274:
1091:
985:
974:, commonly known as fats, to hydrolyze them and make them soluble.
753:
749:
449:
418:
364:
50:
3005:
Infrared and Raman spectra of
Inorganic and Coordination compounds
2664:
derived from "period", but from "iodine": per-iodic acid (compare
2552:
Other cases where hydroxide can act as a nucleophilic reagent are
2396:
value for dissociation of a C–H bond is extremely high, but the pK
817:
in the region centered around 3500 cm. The high frequency of
142:
3869:
Pine, S.H.; Hendrickson, J.B.; Cram, D.J.; Hammond, G.S. (1980).
2503:
2441:
2415:
2351:
2327: = 2, and M = Ca, Mg, Mn, Fe, Co, Ni, Cu, or Zn; hence
2191:
2143:
2020:
1922:
1890:
1828:
1710:
1344:
1328:
1211:, which is a mixture of the strong bases NaOH and KOH with Ca(OH)
977:
966:. An example of the use of sodium carbonate as an alkali is when
885:. Worldwide production in 2004 was approximately 60 million
866:
589:
195:
2090:
is made up of copper, carbonate and hydroxide ions. The mineral
1636:
763:, forming such complexes as tetrahydroxoaluminate/tetrahydroxido
759:
The hydroxide ion can function as a typical electron-pair donor
2609:
Strictly speaking pH is the cologarithm of the hydrogen cation
2375:
1864:: +1, +3, +5, or +7, and A = Cl, Br, or I. The only oxoacid of
1832:
1227:
1099:
760:
741:, which effectively creates hydroxide ions at the active site.
735:
643:
between oxygen and hydrogen atoms. Indeed, the bihydroxide ion
441:
414:
2734:
4113:
2936:
2553:
2519:
2418:. Dissociation can occur in the presence of a suitable base.
2403:
of a carbonyl compound are about 3 log units lower. Typical p
2379:
1654:
886:
862:
745:
615:
524:
371:
335:
154:
132:
2684:
2534:
is an example of a hydroxide ion serving as a nucleophile.
1179:. A solution or suspension of calcium hydroxide is known as
3985:"Illustrated Glossary of Organic Chemistry: Saponification"
3937:
Mullins, J. J. (2008). "Six
Pillars of Organic Chemistry".
2687:
2257:
A number of mixed hydroxides are known with stoichiometry A
2127:
1493: = 13.2 at 25 °C. It is usually written as H
993:
874:
585:
511:
of the hydroxide ion, but covalent compounds which contain
421:
2693:
2058:
is a typical example of a basic carbonate. The formula, Cu
3196:. USNA-TSPR-157. Archived from the original on 2009-08-24
2737:
2699:
2546:
2094:
is an example of a basic chloride. It has the formula, Cu
1745:
with the liberation of hydrogen cations as in a standard
1148:
gives the soluble tetrahydroxoberyllate or tetrahydroxido
854:
592:
solutions have a pH greater than 7 due to the reaction NH
460:
in aqueous solution, liberating solvated hydroxide ions.
2277:. As the formula suggests these substances contain M(OH)
1875:
3585:
Juan J. Borrás-Almenar, Eugenio
Coronado, Achim Müller
573:
3147:
Nature's
Building Blocks: An A–Z Guide to the Elements
3112:
Nature's Building Blocks: An A–Z Guide to the Elements
3052:
1741:. In these compounds one or more hydroxide groups can
3666:
3647:
Diffuse neutron scattering from crystalline materials
2746:
2728:
2711:
2702:
2678:
2078:. Indeed, in the past the formula was written as CuCO
1572:
is treated with alkali the pyramidal hydroxo complex
4011:
2731:
2696:
2681:
1207:+ OH illustrates the basicity of calcium hydroxide.
904:
is used as an alkali, for example, by virtue of the
3901:
3770:"The Function of Hydrogen in Intermolecular Forces"
2801:; Chandler, D.; Hutter, J.; Parrinello, M. (2001).
2762:Crystal structures are illustrated at Web mineral:
2690:
2030:
1593:tetrahedra. In strongly alkaline solutions soluble
1505:is generally accepted. Other silicic acids such as
1395:or methanetetrol) is unstable in aqueous solution:
3670:Zeitschrift für Anorganische und Allgemeine Chemie
3349:
1297:is amphoteric and dissolves in alkaline solution.
4080:(3rd ed.). Oxford: Oxford University Press.
4049:
3767:
2909:) anion. A very short, symmetric hydrogen bond".
1095:Trimeric hydrolysis product of beryllium dication
857:and caustic soda, are used in the manufacture of
5169:
4030:
3989:Department of Chemistry & Biochemistry, UCLA
3826:
3053:Cetin Kurt, Jürgen Bittner. "Sodium Hydroxide".
1897:Ag(OH) decomposes spontaneously to the oxide (Ag
1767:O but is better described structurally as Te(OH)
1327:for the production of pure aluminium oxide from
207:
4012:Holleman, A.F.; Wiberg, E.; Wiberg, N. (2001).
3827:Hattori, H.; Misono, M.; Ono, Y., eds. (1994).
3150:. Oxford, UK: Oxford University Press. p.
3115:. Oxford, UK: Oxford University Press. p.
2482:Nucleophilic acyl substitution with an anionic
830:When the OH group is bound to a metal ion in a
619:Schematic representation of the bihydroxide ion
488:. Both the hydroxide ion and hydroxy group are
118:
3694:
3056:Ullmann's Encyclopedia of Industrial Chemistry
744:Solutions containing the hydroxide ion attack
568:has a value close to 10 at 25 °C, so the
4129:
4075:
1816:, in equilibrium with a small amount of P(OH)
889:. The principal method of manufacture is the
699:, acting as an acid, to form, initially, the
523:The hydroxide ion is naturally produced from
3746:Athanasios K. Karamalidis, David A. Dzombak
3245:A manual for the chemical analysis of metals
2473:
2171:. The room-temperature form of NaOH has the
1600:
3632:
3630:
3628:
3626:
3607:
3605:
3603:
4136:
4122:
4099:(3rd. ed.). Oxford: Clarendon Press.
3352:International Journal of Quantum Chemistry
1144:is formed. Addition of hydroxide to Be(OH)
1105:Water molecules attached to Be are omitted
182:
4031:Housecroft, C. E.; Sharpe, A. G. (2008).
3982:
3976:
3793:
2831:
2047:O was found to be the chloride salt of a
1921:is amphoteric, forming the tetrahydroxido
1812:predominantly has the structure OP(H)(OH)
1136:of −11.7. Addition of acid gives soluble
227:
3714:Journal of the Physical Society of Japan
3623:
3600:
3002:
2996:
2600:and the concentration of hydroxide ions
2477:
1378:
1279:
1226:
1103:Beryllium hydrolysis as a function of pH
1098:
1090:
1086:
614:
3936:
3904:Angewandte Chemie International Edition
3748:Surface Complexation Modeling: Gibbsite
3239:
3237:
3235:
3233:
3187:
3081:
3046:
2672:), and it is thus pronounced per-iodic
2338:
2153:
2007:, has similarities with sulfuric acid H
1383:Carbon forms no simple hydroxides. The
1222:
1023:of −0.36. Lithium hydroxide is used in
999:
668:) that is similar to the length in the
14:
5170:
4164:
3139:
3104:
2979:
2977:
1701:In the higher oxidation states of the
1568:was prepared in anhydrous media. When
800:
576:of a solution is equal to the decimal
4856:
4117:
4094:
3176:Acids and Bases (Essential Chemistry)
3036:
2883:
2881:
2494:The hydroxide ion is intermediate in
1876:Transition and post-transition metals
1235:The simplest hydroxide of boron B(OH)
162:
3230:
3039:Metal–Ligand and Related Vibrations
2974:
2562:nucleophilic aliphatic substitution
1755:is often written with the formula H
1187:carbon dioxide. The reaction Ca(OH)
198:
24:
4076:Shriver, D.F; Atkins, P.W (1999).
3768:Bernal, J.D.; Megaw, H.D. (1935).
3178:. Infobase Publishing (2009). p43.
2878:
2566:nucleophilic aromatic substitution
2285:may be represented by the formula
2209:has four major crystalline forms:
1991:though it has a much more complex
1653:
1644:
1635:
1626:
1617:
1608:
1359:, depending on crystal structure.
1075:, and the hydroxides of the other
58:
49:
25:
5199:
3645:Victoria M. Nield, David A. Keen
3587:Polyoxometalate Molecular Science
3194:US Naval Academy Technical Report
2545:from animal fat (the ester) with
2526:RC(O)OR + OH ⇌ RCO(O)H + OR ⇌ RCO
2361:
2142:, which has been used as a white
2110:. Copper forms hydroxyphosphate (
2070:shows that it is halfway between
1965:M(OH) + BH (L = ligand, B = base)
992:(γ-FeO(OH)), basic hydroxides of
464:is a multi-million-ton per annum
432:. It is an important but usually
3649:, Oxford University Press, 2001
3095:United States Geological Survey.
3007:. Part A (5th ed.). Wiley.
2803:"Autoionization in liquid water"
2724:
2674:
2031:Basic salts containing hydroxide
1183:and can be used to test for the
1004:
518:
36:
4035:(3rd ed.). Prentice Hall.
4005:
3930:
3895:
3862:
3845:
3820:
3811:
3802:
3761:
3740:
3688:
3660:
3639:
3614:
3579:
3552:
3543:
3534:
3506:core (M = Ni, Pd; Q = S, Se)".
3470:
3461:
3452:
3443:
3434:
3425:
3416:
3396:
3387:
3378:
3343:
3322:
3311:
3302:
3293:
3258:
3221:
3181:
3168:
3133:
3098:
3030:
3021:
2756:
2654:
2645:
2366:The hydroxide ion may act as a
1937:in strongly alkaline solution.
845:
838:ion has bands at 470 cm (
809:of compounds containing the OH
503:substances which bear the word
387:(at 25 °C , 100 kPa).
4143:
4097:Structural Inorganic Chemistry
2965:
2930:
2790:
2636:
2615:
2603:
2590:
1789:periodate ion that occurs in K
941:
554:for this reaction, defined as
13:
1:
3983:Hardinger, Steven A. (2017).
3065:10.1002/14356007.a24_345.pub2
2784:
2596:denotes the concentration of
2281:octahedral structural units.
1781:series, , E = Sn, Sb, Te, I;
1597:ions are formed, including .
3750:, John Wiley and Sons, 2010
3247:, ASTM International, 1996,
3027:Nakamoto, Part B, p. 57
492:and can act as catalysts in
7:
3467:Greenwood, pp. 873–874
3431:Greenwood, pp. 383–384
45:
10:
5204:
4858:
3540:Holleman, pp. 711–718
2238:experiments on α-AlO(OH) (
2122:), and nitrate compounds.
1120:. The hydroxide itself is
953:= 10.33 at 25 °C and zero
484: –OH of atoms is the
452:. The hydroxide ion forms
434:minor constituent of water
424:held together by a single
4166:
4151:
4055:Chemistry of the Elements
4053:; Earnshaw, Alan (1997).
3520:10.1007/s11172-005-0296-1
3508:Russian Chemical Bulletin
3279:10.1080/00958970802474862
3213:: CS1 maint: unfit URL (
2474:As a nucleophilic reagent
2283:Layered double hydroxides
2015:; for example, both form
1601:Other main-group elements
1560:have been characterized.
1556:Few hydroxo complexes of
1305:(solid) + OH (aq) ⇌
1027:purification systems for
1013:is a strong base, with a
853:solutions, also known as
428:, and carries a negative
381:
341:
277:
260:
238:
102:
87:
77:
72:
44:
35:
3682:10.1002/zaac.19855311217
3406:, Wiley, New York, 1979
3332:, Academic Press, 2008,
3308:Housectroft, p. 263
3087:Kostick, Dennis (2006).
3041:. London: Edward Arnold.
2583:
2430:The base should have a p
2194:) crystallizes with the
1501:, but the formula Si(OH)
472:compound HO is the
3877:. McGraw–Hill. p.
3404:The Chemistry of Silica
3318:Bayer process chemistry
3299:Housecroft, p. 241
3093:2005 Minerals Yearbook,
3059:. Weinheim: Wiley-VCH.
2971:Greenwood, p. 1168
2842:10.1126/science.1056991
2574:phase-transfer catalyst
2426:R' + B ⇌ RC(O)CHR' + BH
1369:thallium(III) hydroxide
980:, a basic hydroxide of
900:is dissolved in water.
627:the hydroxide ion is a
507:in their names are not
3916:10.1002/anie.200604943
3829:Acid–Base catalysis II
3795:10.1098/rspa.1935.0157
3549:Greenwood, p. 853
3458:Greenwood, p. 781
3449:Greenwood, p. 705
3440:Greenwood, p. 395
3422:Greenwood, p. 384
3393:Greenwood, p. 346
3384:Greenwood, p. 310
3227:Holleman, p. 1108
2491:
1983:with phosphoric acid H
1886:post-transition metals
1880:The hydroxides of the
1658:
1649:
1640:
1631:
1622:
1613:
1287:
1232:
1106:
1096:
620:
413:OH. It consists of an
63:
54:
4059:Butterworth-Heinemann
4051:Greenwood, Norman N.
3140:Emsley, John (2001).
3105:Emsley, John (2001).
3003:Nakamoto, K. (1997).
2570:elimination reactions
2481:
1657:
1648:
1639:
1630:
1621:
1612:
1385:hypothetical compound
1379:Carbon group elements
1373:Thallium(I) hydroxide
1371:are also amphoteric.
1283:
1230:
1102:
1094:
1087:Alkaline earth metals
618:
251:InChI=1S/H2O/h1H2/p-1
89:Systematic IUPAC name
62:
53:
3188:Jaunsen, JR (1989).
3037:Adams, D.M. (1967).
2410:values are 16.7 for
2339:In organic reactions
2154:Structural chemistry
1995:oxoanion chemistry.
1486: = 9.84, p
1223:Boron group elements
1000:Inorganic hydroxides
832:coordination complex
756:plastic containers.
748:. In this case, the
728:equilibrium constant
552:equilibrium constant
476:. The corresponding
470:electrically neutral
468:. The corresponding
436:. It functions as a
4095:Wells, A.F (1962).
4078:Inorganic Chemistry
4033:Inorganic Chemistry
4014:Inorganic Chemistry
3952:2008JChEd..85...83M
3786:1935RSPSA.151..384B
3734:10.1143/JPSJ.39.317
3726:1975JPSJ...39..317E
3330:Inorganic chemistry
2924:10.1021/ja00507a059
2824:2001Sci...291.2121G
2818:(5511): 2121–2124.
2558:Cannizzaro reaction
2447:OH + EtOH ⇌ EtO + H
2350:are two well-known
2344:Potassium hydroxide
2236:neutron diffraction
1589:the two external Pb
1291:Aluminium hydroxide
1173:strontium hydroxide
1161:Magnesium hydroxide
1110:Beryllium hydroxide
1073:potassium hydroxide
891:chloralkali process
819:molecular vibration
801:Vibrational spectra
625:an aqueous solution
313: g·mol
32:
4016:. Academic press.
3970:2011-07-07 at the
3817:Wells, p. 555
3808:Wells, p. 557
3636:Wells, p. 548
3620:Wells, p. 393
3611:Wells, p. 561
3589:, Springer, 2003,
2989:2017-06-19 at the
2539:manufacturing soap
2537:Early methods for
2492:
1981:shows similarities
1943:metal aquo complex
1835:have the formula O
1659:
1650:
1641:
1632:
1623:
1614:
1393:orthocarbonic acid
1375:is a strong base.
1288:
1286:aluminate(III) ion
1233:
1126:solubility product
1107:
1097:
1043:from exhaled gas.
739:carbonic anhydrase
621:
466:commodity chemical
391:Infobox references
342:Related compounds
64:
55:
30:
5165:
5164:
5159:
5158:
4106:978-0-19-855125-6
4087:978-0-19-850330-9
4068:978-0-08-037941-8
4042:978-0-13-175553-6
4023:978-0-12-352651-9
3888:978-0-07-050115-7
3873:Organic chemistry
3838:978-0-444-98655-9
3573:10.1021/om100733n
3567:(17): 3966–3972.
3243:Thomas R. Dulski
3161:978-0-19-850340-8
3126:978-0-19-850340-8
3014:978-0-471-16394-7
2984:IUPAC SC-Database
2951:10.1021/cr900233f
2918:(13): 3688–3689.
2912:J. Am. Chem. Soc.
2797:Geissler, P. L.;
2356:organic chemistry
2323:. Most commonly,
2169:X-ray diffraction
2088:crystal structure
1882:transition metals
1870:hypofluorous acid
1699:
1698:
1562:Tin(II) hydroxide
1477:Orthosilicic acid
1361:Gallium hydroxide
1215:, is used as a CO
1169:calcium hydroxide
1124:in water, with a
1011:Lithium hydroxide
690:hydrogen fluoride
494:organic chemistry
399:Chemical compound
397:
396:
348:Related compounds
144:Interactive image
95:(not recommended)
68:
67:
27:Chemical compound
16:(Redirected from
5195:
5136:
5126:
5113:
5100:
5091:
5081:
5068:
5059:
5046:
5036:
5021:
5011:
5001:
4991:
4981:
4971:
4961:
4951:
4942:
4932:
4922:
4912:
4902:
4892:
4883:
4873:
4800:
4774:
4764:
4755:
4745:
4731:
4721:
4690:
4677:
4652:
4642:
4632:
4623:
4613:
4603:
4585:
4572:
4556:
4546:
4534:
4506:
4496:
4486:
4476:
4466:
4452:
4442:
4432:
4423:
4413:
4403:
4394:
4384:
4375:
4365:
4356:
4347:
4337:
4325:
4300:
4290:
4280:
4270:
4260:
4235:
4225:
4215:
4205:
4195:
4185:
4155:
4154:
4138:
4131:
4124:
4115:
4114:
4110:
4091:
4072:
4057:(2nd ed.).
4046:
4027:
4000:
3999:
3997:
3995:
3980:
3974:
3963:
3960:10.1021/ed085p83
3934:
3928:
3927:
3910:(9): 1560–1638.
3899:
3893:
3892:
3876:
3866:
3860:
3849:
3843:
3842:
3824:
3818:
3815:
3809:
3806:
3800:
3799:
3797:
3780:(873): 384–420.
3765:
3759:
3744:
3738:
3737:
3692:
3686:
3685:
3664:
3658:
3643:
3637:
3634:
3621:
3618:
3612:
3609:
3598:
3583:
3577:
3576:
3556:
3550:
3547:
3541:
3538:
3532:
3531:
3505:
3504:
3503:
3495:
3494:
3474:
3468:
3465:
3459:
3456:
3450:
3447:
3441:
3438:
3432:
3429:
3423:
3420:
3414:
3400:
3394:
3391:
3385:
3382:
3376:
3375:
3347:
3341:
3326:
3320:
3315:
3309:
3306:
3300:
3297:
3291:
3290:
3262:
3256:
3241:
3228:
3225:
3219:
3218:
3212:
3204:
3202:
3201:
3185:
3179:
3172:
3166:
3165:
3137:
3131:
3130:
3102:
3096:
3085:
3079:
3078:
3050:
3044:
3042:
3034:
3028:
3025:
3019:
3018:
3000:
2994:
2981:
2972:
2969:
2963:
2962:
2945:(4): 2174–2216.
2934:
2928:
2927:
2908:
2907:
2906:
2898:
2897:
2885:
2876:
2875:
2873:
2872:
2866:
2860:. Archived from
2835:
2807:
2794:
2778:
2760:
2754:
2749:
2744:
2743:
2740:
2739:
2736:
2733:
2730:
2718:
2714:
2709:
2708:
2705:
2704:
2701:
2698:
2695:
2692:
2689:
2686:
2683:
2680:
2658:
2652:
2649:
2643:
2640:
2634:
2633:
2631:
2630:
2627:
2619:
2613:
2607:
2601:
2598:hydrogen cations
2594:
2556:hydrolysis, the
2517:
2516:
2515:
2348:sodium hydroxide
2322:
2320:
2319:
2307:
2306:
2304:
2303:
2297:
2232:hydrogen bonding
2205:hydroxide Al(OH)
2130:carbonate, (PbCO
2076:copper hydroxide
2072:copper carbonate
2025:polyoxometalates
1936:
1935:
1934:
1895:silver hydroxide
1862:oxidation number
1854:
1852:
1851:
1848:
1845:
1823:The oxoacids of
1802:phosphorous acid
1665:Phosphorous acid
1605:
1604:
1583:
1582:
1581:
1535:pyrosilicic acid
1507:metasilicic acid
1432:
1431:
1430:
1414:
1413:
1412:
1365:indium hydroxide
1342:
1341:
1340:
1317:
1316:
1315:
1267:
1266:
1265:
1206:
1205:
1204:
1177:barium hydroxide
1069:Sodium hydroxide
988:(α-FeO(OH)) and
942:
938:
937:
936:
922:
921:
920:
902:Sodium carbonate
851:Sodium hydroxide
815:absorption bands
811:functional group
807:infrared spectra
722:
721:
720:
683:
682:
681:
663:
662:
661:
653:
652:
610:buffer solutions
607:
606:
605:
474:hydroxyl radical
462:Sodium hydroxide
456:, some of which
411:chemical formula
312:
299:
298:
297:
285:Chemical formula
231:
211:
200:
186:
166:
146:
122:
46:
40:
33:
29:
21:
5203:
5202:
5198:
5197:
5196:
5194:
5193:
5192:
5188:Water chemistry
5168:
5167:
5166:
5161:
5160:
5135:
5131:
5125:
5121:
5112:
5108:
5104:
5102:
5099:
5095:
5093:
5090:
5086:
5080:
5076:
5072:
5070:
5067:
5063:
5061:
5058:
5054:
5045:
5041:
5035:
5031:
5020:
5016:
5010:
5006:
5000:
4996:
4990:
4986:
4980:
4976:
4970:
4966:
4960:
4956:
4950:
4946:
4944:
4941:
4937:
4931:
4927:
4921:
4917:
4911:
4907:
4901:
4897:
4891:
4887:
4885:
4882:
4878:
4872:
4868:
4799:
4795:
4773:
4769:
4763:
4759:
4757:
4754:
4750:
4744:
4740:
4738:
4730:
4726:
4720:
4716:
4689:
4685:
4676:
4672:
4651:
4647:
4641:
4637:
4631:
4627:
4625:
4622:
4618:
4612:
4608:
4602:
4598:
4584:
4580:
4571:
4567:
4555:
4551:
4545:
4541:
4533:
4529:
4505:
4501:
4495:
4491:
4485:
4481:
4475:
4471:
4465:
4461:
4459:
4451:
4447:
4441:
4437:
4431:
4427:
4425:
4422:
4418:
4412:
4408:
4402:
4398:
4396:
4393:
4389:
4383:
4379:
4377:
4374:
4370:
4364:
4360:
4358:
4355:
4351:
4349:
4346:
4342:
4336:
4332:
4324:
4320:
4299:
4295:
4289:
4285:
4279:
4275:
4269:
4265:
4259:
4255:
4234:
4230:
4223:
4219:
4217:
4214:
4210:
4204:
4200:
4194:
4190:
4184:
4180:
4147:
4142:
4107:
4088:
4069:
4043:
4024:
4008:
4003:
3993:
3991:
3981:
3977:
3972:Wayback Machine
3935:
3931:
3900:
3896:
3889:
3867:
3863:
3850:
3846:
3839:
3825:
3821:
3816:
3812:
3807:
3803:
3774:Proc. R. Soc. A
3766:
3762:
3745:
3741:
3711:
3707:
3700:
3693:
3689:
3665:
3661:
3644:
3640:
3635:
3624:
3619:
3615:
3610:
3601:
3584:
3580:
3561:Organometallics
3557:
3553:
3548:
3544:
3539:
3535:
3502:
3499:
3498:
3497:
3493:
3490:
3489:
3488:
3486:
3484:
3480:
3475:
3471:
3466:
3462:
3457:
3453:
3448:
3444:
3439:
3435:
3430:
3426:
3421:
3417:
3401:
3397:
3392:
3388:
3383:
3379:
3348:
3344:
3328:James E. House
3327:
3323:
3316:
3312:
3307:
3303:
3298:
3294:
3263:
3259:
3242:
3231:
3226:
3222:
3206:
3205:
3199:
3197:
3186:
3182:
3173:
3169:
3162:
3138:
3134:
3127:
3103:
3099:
3086:
3082:
3075:
3051:
3047:
3035:
3031:
3026:
3022:
3015:
3001:
2997:
2991:Wayback Machine
2982:
2975:
2970:
2966:
2935:
2931:
2905:
2902:
2901:
2900:
2896:
2893:
2892:
2891:
2889:
2886:
2879:
2870:
2868:
2864:
2805:
2795:
2791:
2787:
2782:
2781:
2761:
2757:
2747:
2727:
2723:
2716:
2712:
2677:
2673:
2670:perchloric acid
2659:
2655:
2650:
2646:
2641:
2637:
2628:
2625:
2624:
2622:
2620:
2616:
2608:
2604:
2595:
2591:
2586:
2578:dichlorocarbene
2529:
2514:
2511:
2510:
2509:
2507:
2502:ion F, and the
2496:nucleophilicity
2476:
2464:
2455:to produce the
2450:
2436:
2425:
2409:
2401:alpha hydrogens
2399:
2394:
2364:
2341:
2318:
2315:
2314:
2313:
2305:
2299:
2293:
2292:
2290:
2289:
2288:
2286:
2280:
2276:
2272:
2268:
2264:
2260:
2253:
2249:
2245:
2213:(most stable),
2208:
2189:
2182:
2178:
2173:thallium iodide
2164:sodium chloride
2156:
2141:
2137:
2133:
2109:
2105:
2101:
2097:
2085:
2081:
2069:
2065:
2061:
2046:
2042:
2033:
2014:
2010:
2006:
2002:
1990:
1986:
1979:
1975:
1964:
1958:
1954:
1933:
1930:
1929:
1928:
1926:
1920:
1908:
1904:
1900:
1878:
1855:
1849:
1846:
1840:
1839:
1837:
1819:
1815:
1811:
1807:
1796:
1792:
1770:
1766:
1762:
1758:
1740:
1736:
1728:
1724:
1719:phosphoric acid
1670:Phosphoric acid
1603:
1592:
1580:
1577:
1576:
1575:
1573:
1567:
1548:
1544:
1540:
1532:
1528:
1524:
1516:
1512:
1504:
1500:
1496:
1492:
1485:
1474:
1462:
1458:
1454:
1440:
1436:
1429:
1426:
1425:
1424:
1422:
1418:
1411:
1408:
1407:
1406:
1404:
1402:
1390:
1381:
1339:
1336:
1335:
1334:
1332:
1314:
1311:
1310:
1309:
1307:
1304:
1296:
1285:
1264:
1261:
1260:
1259:
1257:
1254:
1250:
1238:
1225:
1218:
1214:
1203:
1200:
1199:
1198:
1196:
1194:
1190:
1166:
1147:
1135:
1115:
1104:
1089:
1062:
1058:
1054:
1050:
1021:
1007:
1002:
951:
940:
935:
932:
931:
930:
928:
926:
919:
916:
915:
914:
912:
848:
803:
796:
774:
769:bridging ligand
719:
716:
715:
714:
712:
710:
680:
677:
676:
675:
673:
660:
657:
656:
655:
651:
648:
647:
646:
644:
604:
601:
600:
599:
597:
595:
582:hydrogen cation
563:
545:
539:
529:self-ionization
521:
509:ionic compounds
430:electric charge
400:
393:
388:
375:
370:
368:
363:
359:
356:
349:
310:
296:
294:
293:
292:
290:
287:
273:
268:
267:
256:
253:
252:
246:
245:
234:
214:
201:
189:
169:
149:
136:
125:
112:
98:
97:
83:
28:
23:
22:
15:
12:
11:
5:
5201:
5191:
5190:
5185:
5180:
5163:
5162:
5157:
5156:
5153:
5150:
5147:
5144:
5141:
5138:
5133:
5128:
5123:
5118:
5115:
5110:
5106:
5097:
5088:
5083:
5078:
5074:
5065:
5056:
5051:
5048:
5043:
5038:
5033:
5028:
5024:
5023:
5018:
5013:
5008:
5003:
4998:
4993:
4988:
4983:
4978:
4973:
4968:
4963:
4958:
4953:
4948:
4939:
4934:
4929:
4924:
4919:
4914:
4909:
4904:
4899:
4894:
4889:
4880:
4875:
4870:
4865:
4861:
4860:
4857:
4854:
4853:
4850:
4847:
4844:
4841:
4838:
4835:
4832:
4829:
4826:
4823:
4820:
4817:
4814:
4811:
4808:
4805:
4802:
4797:
4792:
4786:
4785:
4782:
4779:
4776:
4771:
4766:
4761:
4752:
4747:
4742:
4733:
4728:
4723:
4718:
4713:
4710:
4707:
4704:
4701:
4698:
4695:
4692:
4687:
4682:
4679:
4674:
4669:
4663:
4662:
4659:
4654:
4649:
4644:
4639:
4634:
4629:
4620:
4615:
4610:
4605:
4600:
4595:
4590:
4587:
4582:
4577:
4574:
4569:
4564:
4561:
4558:
4553:
4548:
4543:
4538:
4536:
4531:
4526:
4520:
4519:
4516:
4511:
4508:
4503:
4498:
4493:
4488:
4483:
4478:
4473:
4468:
4463:
4454:
4449:
4444:
4439:
4434:
4429:
4420:
4415:
4410:
4405:
4400:
4391:
4386:
4381:
4372:
4367:
4362:
4353:
4344:
4339:
4334:
4329:
4327:
4322:
4317:
4311:
4310:
4307:
4302:
4297:
4292:
4287:
4282:
4277:
4272:
4267:
4262:
4257:
4252:
4246:
4245:
4242:
4237:
4232:
4227:
4221:
4212:
4207:
4202:
4197:
4192:
4187:
4182:
4177:
4171:
4170:
4167:
4165:
4163:
4161:
4153:
4152:
4149:
4148:
4141:
4140:
4133:
4126:
4118:
4112:
4111:
4105:
4092:
4086:
4073:
4067:
4047:
4041:
4028:
4022:
4007:
4004:
4002:
4001:
3975:
3940:J. Chem. Educ.
3929:
3894:
3887:
3861:
3844:
3837:
3819:
3810:
3801:
3760:
3739:
3709:
3702:
3696:
3687:
3659:
3638:
3622:
3613:
3599:
3578:
3551:
3542:
3533:
3500:
3491:
3482:
3478:
3469:
3460:
3451:
3442:
3433:
3424:
3415:
3395:
3386:
3377:
3358:(3): 315–322.
3342:
3321:
3310:
3301:
3292:
3267:J. Coord. Chem
3257:
3229:
3220:
3180:
3174:Lew. Kristi.,
3167:
3160:
3132:
3125:
3097:
3080:
3074:978-3527306732
3073:
3045:
3029:
3020:
3013:
2995:
2973:
2964:
2929:
2903:
2894:
2877:
2788:
2786:
2783:
2780:
2779:
2755:
2653:
2644:
2635:
2614:
2602:
2588:
2587:
2585:
2582:
2532:
2531:
2527:
2512:
2475:
2472:
2462:
2453:
2452:
2448:
2434:
2428:
2427:
2423:
2407:
2397:
2392:
2363:
2362:Base catalysis
2360:
2340:
2337:
2316:
2291:
2278:
2274:
2270:
2266:
2262:
2258:
2251:
2247:
2243:
2206:
2196:cadmium iodide
2187:
2180:
2176:
2155:
2152:
2148:lead poisoning
2139:
2135:
2131:
2107:
2103:
2099:
2095:
2083:
2079:
2067:
2063:
2059:
2044:
2040:
2032:
2029:
2012:
2008:
2004:
2000:
1988:
1984:
1977:
1973:
1967:
1966:
1960:
1956:
1950:
1931:
1918:
1915:Zinc hydroxide
1906:
1902:
1898:
1877:
1874:
1836:
1817:
1813:
1809:
1805:
1794:
1790:
1768:
1764:
1760:
1756:
1747:Brønsted–Lowry
1738:
1734:
1726:
1722:
1697:
1696:
1691:
1682:
1677:
1672:
1667:
1661:
1660:
1651:
1642:
1633:
1624:
1615:
1602:
1599:
1590:
1578:
1565:
1546:
1542:
1538:
1530:
1526:
1522:
1519:disilicic acid
1514:
1510:
1502:
1498:
1494:
1490:
1483:
1470:
1460:
1456:
1452:
1445:Carbon dioxide
1442:
1441:
1438:
1434:
1427:
1420:
1416:
1409:
1400:
1388:
1380:
1377:
1337:
1321:
1320:
1312:
1302:
1294:
1271:
1270:
1262:
1252:
1248:
1236:
1224:
1221:
1216:
1212:
1201:
1192:
1188:
1164:
1145:
1133:
1113:
1088:
1085:
1065:
1064:
1060:
1056:
1052:
1048:
1041:carbon dioxide
1019:
1006:
1003:
1001:
998:
964:Solvay process
959:
958:
955:ionic strength
949:
933:
924:
917:
871:drinking water
847:
844:
802:
799:
797:of about 5.9.
794:
788:
787:
772:
724:
723:
717:
708:
697:carbon dioxide
678:
658:
649:
641:hydrogen bonds
633:Brønsted–Lowry
602:
593:
566:
565:
561:
548:
547:
543:
537:
520:
517:
513:hydroxy groups
407:diatomic anion
398:
395:
394:
389:
385:standard state
382:
379:
378:
373:
366:
354:
350:
347:
344:
343:
339:
338:
333:
331:Conjugate base
327:
326:
321:
319:Conjugate acid
315:
314:
308:
302:
301:
295:
288:
283:
280:
279:
275:
274:
272:
271:
263:
262:
261:
258:
257:
255:
254:
250:
249:
241:
240:
239:
236:
235:
233:
232:
224:
222:
216:
215:
213:
212:
204:
202:
194:
191:
190:
188:
187:
179:
177:
171:
170:
168:
167:
159:
157:
151:
150:
148:
147:
139:
137:
130:
127:
126:
124:
123:
115:
113:
108:
105:
104:
100:
99:
92:
91:
85:
84:
81:
75:
74:
70:
69:
66:
65:
56:
42:
41:
26:
9:
6:
4:
3:
2:
5200:
5189:
5186:
5184:
5181:
5179:
5176:
5175:
5173:
5154:
5151:
5148:
5145:
5142:
5139:
5137:
5129:
5127:
5119:
5116:
5114:
5101:
5092:
5084:
5082:
5069:
5060:
5052:
5049:
5047:
5039:
5037:
5029:
5026:
5025:
5022:
5014:
5012:
5004:
5002:
4994:
4992:
4984:
4982:
4974:
4972:
4964:
4962:
4954:
4952:
4943:
4935:
4933:
4925:
4923:
4915:
4913:
4905:
4903:
4895:
4893:
4884:
4876:
4874:
4866:
4863:
4862:
4855:
4851:
4848:
4845:
4842:
4839:
4836:
4833:
4830:
4827:
4824:
4821:
4818:
4815:
4812:
4809:
4806:
4803:
4801:
4793:
4791:
4788:
4787:
4783:
4780:
4777:
4775:
4767:
4765:
4756:
4748:
4746:
4737:
4734:
4732:
4724:
4722:
4714:
4711:
4708:
4705:
4702:
4699:
4696:
4693:
4691:
4683:
4680:
4678:
4670:
4668:
4665:
4664:
4660:
4658:
4655:
4653:
4645:
4643:
4635:
4633:
4624:
4616:
4614:
4606:
4604:
4596:
4594:
4591:
4588:
4586:
4578:
4575:
4573:
4565:
4562:
4559:
4557:
4549:
4547:
4539:
4537:
4535:
4527:
4525:
4522:
4521:
4517:
4515:
4512:
4509:
4507:
4499:
4497:
4489:
4487:
4479:
4477:
4469:
4467:
4458:
4455:
4453:
4445:
4443:
4435:
4433:
4424:
4416:
4414:
4406:
4404:
4395:
4387:
4385:
4376:
4368:
4366:
4357:
4348:
4340:
4338:
4330:
4328:
4326:
4318:
4316:
4313:
4312:
4308:
4306:
4303:
4301:
4293:
4291:
4283:
4281:
4273:
4271:
4263:
4261:
4253:
4251:
4248:
4247:
4243:
4241:
4238:
4236:
4228:
4226:
4216:
4208:
4206:
4198:
4196:
4188:
4186:
4178:
4176:
4173:
4172:
4168:
4162:
4160:
4157:
4156:
4150:
4146:
4139:
4134:
4132:
4127:
4125:
4120:
4119:
4116:
4108:
4102:
4098:
4093:
4089:
4083:
4079:
4074:
4070:
4064:
4060:
4056:
4052:
4048:
4044:
4038:
4034:
4029:
4025:
4019:
4015:
4010:
4009:
3990:
3986:
3979:
3973:
3969:
3966:
3961:
3957:
3953:
3949:
3945:
3942:
3941:
3933:
3925:
3921:
3917:
3913:
3909:
3905:
3898:
3890:
3884:
3880:
3875:
3874:
3865:
3858:
3857:0-02-390171-3
3854:
3848:
3840:
3834:
3830:
3823:
3814:
3805:
3796:
3791:
3787:
3783:
3779:
3775:
3771:
3764:
3757:
3756:0-470-58768-7
3753:
3749:
3743:
3735:
3731:
3727:
3723:
3719:
3715:
3706:
3699:
3691:
3683:
3679:
3675:
3671:
3663:
3657:, p. 276
3656:
3655:0-19-851790-4
3652:
3648:
3642:
3633:
3631:
3629:
3627:
3617:
3608:
3606:
3604:
3596:
3595:1-4020-1242-X
3592:
3588:
3582:
3574:
3570:
3566:
3562:
3555:
3546:
3537:
3529:
3525:
3521:
3517:
3513:
3509:
3473:
3464:
3455:
3446:
3437:
3428:
3419:
3413:
3412:0-471-02404-X
3409:
3405:
3399:
3390:
3381:
3373:
3369:
3365:
3361:
3357:
3353:
3346:
3340:, p. 764
3339:
3338:0-12-356786-6
3335:
3331:
3325:
3319:
3314:
3305:
3296:
3288:
3284:
3280:
3276:
3272:
3268:
3261:
3254:
3253:0-8031-2066-4
3250:
3246:
3240:
3238:
3236:
3234:
3224:
3216:
3210:
3195:
3191:
3184:
3177:
3171:
3163:
3157:
3153:
3149:
3148:
3143:
3136:
3128:
3122:
3118:
3114:
3113:
3108:
3101:
3094:
3091:, chapter in
3090:
3084:
3076:
3070:
3066:
3062:
3058:
3057:
3049:
3040:
3033:
3024:
3016:
3010:
3006:
2999:
2992:
2988:
2985:
2980:
2978:
2968:
2960:
2956:
2952:
2948:
2944:
2940:
2933:
2925:
2921:
2917:
2914:
2913:
2884:
2882:
2867:on 2007-06-25
2863:
2859:
2855:
2851:
2847:
2843:
2839:
2834:
2833:10.1.1.6.4964
2829:
2825:
2821:
2817:
2813:
2812:
2804:
2800:
2793:
2789:
2777:
2773:
2769:
2765:
2759:
2752:
2751:
2742:
2722:, and not as
2721:
2720:
2707:
2671:
2667:
2663:
2657:
2648:
2639:
2618:
2612:
2606:
2599:
2593:
2589:
2581:
2579:
2575:
2571:
2567:
2563:
2559:
2555:
2550:
2548:
2544:
2543:triglycerides
2540:
2535:
2525:
2524:
2523:
2521:
2505:
2501:
2497:
2489:
2488:leaving group
2485:
2480:
2471:
2469:
2465:
2458:
2446:
2445:
2444:
2443:
2438:
2433:
2421:
2420:
2419:
2417:
2413:
2406:
2402:
2395:
2391:
2385:
2381:
2377:
2373:
2369:
2368:base catalyst
2359:
2357:
2353:
2349:
2345:
2336:
2334:
2331: =
2330:
2326:
2311:
2302:
2296:
2284:
2255:
2241:
2237:
2233:
2228:
2224:
2220:
2219:nordstrandite
2216:
2212:
2204:
2199:
2197:
2193:
2184:
2174:
2170:
2165:
2161:
2151:
2149:
2145:
2129:
2125:
2121:
2117:
2114:), arsenate (
2113:
2093:
2089:
2077:
2073:
2057:
2052:
2050:
2038:
2028:
2026:
2022:
2018:
1998:
1994:
1982:
1971:
1963:
1953:
1948:
1947:
1946:
1944:
1938:
1924:
1916:
1912:
1896:
1892:
1887:
1883:
1873:
1871:
1867:
1863:
1859:
1856:A(OH), where
1843:
1834:
1830:
1826:
1821:
1803:
1798:
1788:
1784:
1780:
1779:isoelectronic
1776:
1772:
1754:
1753:Telluric acid
1750:
1748:
1744:
1732:
1731:sulfuric acid
1720:
1716:
1712:
1708:
1704:
1695:
1692:
1690:
1689:periodic acid
1686:
1683:
1681:
1680:Telluric acid
1678:
1676:
1675:Sulfuric acid
1673:
1671:
1668:
1666:
1663:
1662:
1656:
1652:
1647:
1643:
1638:
1634:
1629:
1625:
1620:
1616:
1611:
1607:
1606:
1598:
1596:
1586:
1571:
1570:tin(II) oxide
1563:
1559:
1554:
1552:
1536:
1520:
1508:
1489:
1482:
1478:
1473:
1468:
1464:
1450:
1449:carbonic acid
1446:
1421:
1398:
1397:
1396:
1394:
1386:
1376:
1374:
1370:
1366:
1362:
1358:
1354:
1348:
1346:
1330:
1326:
1325:Bayer process
1318:
1300:
1299:
1298:
1292:
1284:Tetrahydroxo-
1282:
1278:
1276:
1273:A variety of
1268:
1246:
1245:
1244:
1242:
1229:
1220:
1210:
1186:
1182:
1178:
1174:
1170:
1162:
1158:
1157:atomic number
1153:
1151:
1143:
1139:
1131:
1127:
1123:
1119:
1111:
1101:
1093:
1084:
1082:
1078:
1077:alkali metals
1074:
1070:
1046:
1045:
1044:
1042:
1038:
1034:
1030:
1026:
1025:breathing gas
1022:
1018:
1012:
1005:Alkali metals
997:
995:
991:
990:lepidocrocite
987:
983:
979:
975:
973:
972:triglycerides
969:
965:
956:
952:
948:
911:
910:
909:
907:
903:
899:
894:
892:
888:
884:
883:drain cleaner
880:
876:
872:
868:
864:
860:
856:
852:
843:
841:
837:
833:
828:
825:
820:
816:
812:
808:
798:
793:
785:
784:
783:
781:
776:
770:
766:
762:
757:
755:
751:
747:
742:
740:
737:
733:
732:Carbonic acid
729:
706:
705:
704:
702:
698:
693:
691:
687:
671:
667:
642:
638:
634:
630:
626:
617:
613:
611:
591:
587:
583:
579:
575:
571:
570:concentration
560:
557:
556:
555:
553:
541:
534:
533:
532:
530:
526:
519:Hydroxide ion
516:
514:
510:
506:
502:
497:
495:
491:
487:
486:hydroxy group
483:
479:
475:
471:
467:
463:
459:
455:
451:
447:
443:
439:
435:
431:
427:
426:covalent bond
423:
420:
416:
412:
408:
404:
392:
386:
380:
377:
369:
362:
358:
351:
346:
345:
340:
337:
334:
332:
329:
328:
325:
322:
320:
317:
316:
309:
307:
304:
303:
289:
286:
282:
281:
276:
270:
269:
266:
259:
248:
247:
244:
237:
230:
226:
225:
223:
221:
218:
217:
210:
206:
205:
203:
197:
193:
192:
185:
181:
180:
178:
176:
173:
172:
165:
161:
160:
158:
156:
153:
152:
145:
141:
140:
138:
134:
129:
128:
121:
117:
116:
114:
111:
107:
106:
101:
96:
90:
86:
80:
76:
71:
61:
57:
52:
48:
47:
43:
39:
34:
19:
18:Hydroxide ion
4144:
4096:
4077:
4054:
4032:
4013:
4006:Bibliography
3992:. Retrieved
3978:
3943:
3938:
3932:
3907:
3903:
3897:
3872:
3864:
3847:
3831:. Elsevier.
3828:
3822:
3813:
3804:
3777:
3773:
3763:
3742:
3717:
3713:
3704:
3697:
3690:
3673:
3669:
3662:
3641:
3616:
3581:
3564:
3560:
3554:
3545:
3536:
3511:
3507:
3472:
3463:
3454:
3445:
3436:
3427:
3418:
3403:
3402:R. K. Iler,
3398:
3389:
3380:
3355:
3351:
3345:
3324:
3313:
3304:
3295:
3273:(1): 14–22.
3270:
3266:
3260:
3223:
3209:cite journal
3198:. Retrieved
3193:
3183:
3175:
3170:
3146:
3135:
3111:
3100:
3092:
3083:
3054:
3048:
3038:
3032:
3023:
3004:
2998:
2967:
2942:
2938:
2932:
2915:
2910:
2869:. Retrieved
2862:the original
2815:
2809:
2792:
2772:Norstrandite
2758:
2661:
2660:The name is
2656:
2647:
2638:
2617:
2605:
2592:
2551:
2536:
2533:
2498:between the
2493:
2454:
2439:
2431:
2429:
2412:acetaldehyde
2404:
2389:
2384:carbon acids
2365:
2342:
2332:
2328:
2324:
2309:
2300:
2294:
2273:, and AM(OH)
2256:
2250:, and Fe(OH)
2225:. All these
2200:
2185:
2160:ionic radius
2157:
2118:), sulfate (
2054:The mineral
2053:
2034:
1997:Chromic acid
1970:Vanadic acid
1968:
1961:
1951:
1939:
1911:precipitates
1879:
1857:
1841:
1822:
1799:
1786:
1782:
1774:
1773:
1751:
1700:
1684:
1587:
1555:
1534:
1518:
1506:
1487:
1480:
1476:
1471:
1467:Silicic acid
1465:
1443:
1382:
1349:
1322:
1289:
1272:
1234:
1154:
1129:
1108:
1081:strong bases
1066:
1016:
1008:
976:
968:washing soda
960:
946:
895:
849:
846:Applications
829:
813:have strong
804:
791:
789:
777:
758:
743:
725:
694:
622:
567:
558:
549:
522:
504:
498:
490:nucleophiles
402:
401:
103:Identifiers
94:
3676:(12): 119.
3597:, p. 4
3255:p. 100
3142:"Aluminium"
3107:"Aluminium"
2799:Dellago, C.
2484:nucleophile
2414:and 19 for
2126:is a basic
2120:brochantite
2112:libethenite
1715:noble gases
1239:, known as
1219:absorbent.
1047:2 LiOH + CO
1037:rebreathers
881:, and as a
836:tetrahedral
701:bicarbonate
692:solutions.
578:cologarithm
446:nucleophile
336:Oxide anion
278:Properties
164:CHEBI:16234
5178:Hydroxides
5172:Categories
4145:Hydroxides
3720:(2): 317.
3514:(3): 615.
3481:and HP(OH)
3200:2008-06-17
3089:"Soda Ash"
3043:Chapter 5.
2871:2017-10-25
2785:References
2666:iodic acid
2468:hemiacetal
2227:polymorphs
2203:amphoteric
2179:and (OH)Li
2124:White lead
2049:tetrameric
2023:, forming
2017:acid salts
1868:is F(OH),
1743:dissociate
1707:chalcogens
1703:pnictogens
1694:Xenic acid
1319: (aq)
1241:boric acid
1138:hydrolysis
1118:amphoteric
1039:to remove
1033:submarines
1029:spacecraft
906:hydrolysis
879:detergents
824:band width
670:bifluoride
637:Lewis base
531:reaction:
478:covalently
458:dissociate
306:Molar mass
229:9159UV381P
175:ChemSpider
131:3D model (
120:14280-30-9
110:CAS Number
93:Oxidanide
79:IUPAC name
31:Hydroxide
5183:Oxyanions
3994:April 10,
3946:(1): 83.
3758:pp. 15 ff
3372:1097-461X
2939:Chem. Rev
2828:CiteSeerX
2568:, and in
2486:(Nu) and
2459:ion. The
2116:olivenite
2092:atacamite
2056:malachite
2037:zirconium
1959:) + B ⇌ L
1905:and M(OH)
1558:germanium
1551:oxyanions
1275:oxyanions
1209:Soda lime
1185:weak acid
1181:limewater
1152:anion, .
1150:beryllate
1128:log
1122:insoluble
1079:are also
982:aluminium
908:reaction
898:weak acid
765:aluminate
750:silicates
686:viscosity
542:+ OH ⇌ 2H
505:hydroxide
501:inorganic
403:Hydroxide
82:Hydroxide
3968:Archived
3924:18236505
3528:93718865
3287:93623985
2987:Archived
2959:20170203
2850:11251111
2776:Doyleite
2768:Bayerite
2764:Gibbsite
2611:activity
2541:treated
2500:fluoride
2457:ethoxide
2372:alcohols
2352:reagents
2246:, Zn(OH)
2240:diaspore
2223:doyleite
2215:bayerite
2211:gibbsite
2106:·3Cu(OH)
1993:vanadate
1866:fluorine
1825:chlorine
1711:halogens
1595:plumbate
1357:diaspore
1353:boehmite
1142:aqua ion
986:goethite
867:textiles
790:with a p
754:airtight
450:catalyst
448:, and a
419:hydrogen
4859:
4220:[NH
3948:Bibcode
3782:Bibcode
3722:Bibcode
2858:1081091
2820:Bibcode
2811:Science
2632:
2623:
2442:ethanol
2422:RC(O)CH
2416:acetone
2376:phenols
2298:⁄
2192:brucite
2144:pigment
2138:·Pb(OH)
2082:·Cu(OH)
2021:olation
1923:zincate
1891:olation
1860:is the
1853:
1838:
1829:bromine
1533:), and
1459:(OC(OH)
1433:+ H ⇌ H
1345:red mud
1329:bauxite
1323:In the
1195:⇌ Ca +
978:Bauxite
707:OH + CO
631:in the
590:ammonia
580:of the
527:by the
300:
196:PubChem
5132:Cm(OH)
5122:Am(OH)
5096:Np(OH)
5087:Np(OH)
5042:Th(OH)
5032:Ac(OH)
5017:Yb(OH)
5007:Tm(OH)
4997:Er(OH)
4987:Ho(OH)
4977:Dy(OH)
4967:Tb(OH)
4957:Gd(OH)
4947:Eu(OH)
4938:Eu(OH)
4928:Sm(OH)
4918:Pm(OH)
4908:Nd(OH)
4898:Pr(OH)
4888:Ce(OH)
4879:Ce(OH)
4869:La(OH)
4796:Ra(OH)
4770:Bi(OH)
4760:Pb(OH)
4751:Pb(OH)
4741:Tl(OH)
4727:Hg(OH)
4717:Au(OH)
4686:Lu(OH)
4673:Ba(OH)
4648:Te(OH)
4638:Sb(OH)
4628:Sn(OH)
4619:Sn(OH)
4609:In(OH)
4599:Cd(OH)
4581:Rh(OH)
4568:Tc(OH)
4552:Zr(OH)
4530:Sr(OH)
4502:As(OH)
4492:Ge(OH)
4482:Ga(OH)
4472:Zn(OH)
4462:Cu(OH)
4448:Ni(OH)
4438:Co(OH)
4428:Fe(OH)
4419:Fe(OH)
4409:Mn(OH)
4399:Cr(OH)
4390:Cr(OH)
4361:Ti(OH)
4352:Ti(OH)
4343:Ti(OH)
4333:Sc(OH)
4321:Ca(OH)
4276:Si(OH)
4266:Al(OH)
4256:Mg(OH)
4181:Be(OH)
4103:
4084:
4065:
4039:
4020:
3922:
3885:
3855:
3835:
3754:
3653:
3593:
3526:
3410:
3370:
3336:
3285:
3251:
3158:
3123:
3071:
3011:
2957:
2856:
2848:
2830:
2386:. The
2382:, and
2380:amines
2221:, and
2098:Cl(OH)
2086:. The
1927:Zn(OH)
1917:Zn(OH)
1833:iodine
1831:, and
1729:, and
1713:, and
1574:Sn(OH)
1564:Sn(OH)
1367:, and
1333:Al(OH)
1308:Al(OH)
1301:Al(OH)
1293:Al(OH)
1175:, and
1163:Mg(OH)
1112:Be(OH)
1035:, and
887:tonnes
786:⇌ + H
780:ionise
761:ligand
736:enzyme
596:+ H ⇌
480:bound
442:ligand
415:oxygen
311:17.007
265:SMILES
73:Names
5064:U(OH)
5055:U(OH)
4542:Y(OH)
4380:V(OH)
4371:V(OH)
4296:S(OH)
4286:P(OH)
4231:O(OH)
4211:N(OH)
4201:C(OH)
4191:B(OH)
3524:S2CID
3283:S2CID
2865:(PDF)
2854:S2CID
2806:(PDF)
2715:-eye-
2584:Notes
2554:amide
2530:+ HOR
2520:Ester
2504:amide
2269:M(OH)
2261:M(OH)
1775:Ortho
1685:Ortho
1399:C(OH)
1387:C(OH)
1258:B(OH)
1247:B(OH)
939:+ OH
875:soaps
863:paper
840:Raman
746:glass
703:ion.
525:water
499:Many
482:group
454:salts
409:with
405:is a
324:Water
243:InChI
155:ChEBI
133:JSmol
5109:(OH)
5077:(OH)
4790:FrOH
4736:TlOH
4667:CsOH
4593:AgOH
4524:RbOH
4514:BrOH
4457:CuOH
4305:ClOH
4250:NaOH
4175:LiOH
4101:ISBN
4082:ISBN
4063:ISBN
4037:ISBN
4018:ISBN
3996:2023
3920:PMID
3883:ISBN
3853:ISBN
3833:ISBN
3752:ISBN
3708:(OH)
3651:ISBN
3591:ISBN
3408:ISBN
3368:ISSN
3334:ISBN
3249:ISBN
3215:link
3156:ISBN
3121:ISBN
3069:ISBN
3009:ISBN
2955:PMID
2846:PMID
2774:and
2750:-ee-
2748:PEER
2549:.
2506:ion
2346:and
2201:The
2128:lead
2074:and
2066:(OH)
1955:M(OH
1925:ion
1884:and
1787:meso
1255:O ⇌
1191:+ CO
1051:→ Li
994:iron
927:O ⇌
877:and
861:and
859:pulp
805:The
726:The
672:ion
629:base
550:The
444:, a
440:, a
438:base
422:atom
417:and
220:UNII
5155:No
5152:Md
5149:Fm
5146:Es
5143:Cf
5140:Bk
5117:Pu
5105:NpO
5050:Pa
5027:**
4852:Og
4849:Ts
4846:Lv
4843:Mc
4840:Fl
4837:Nh
4834:Cn
4831:Rg
4828:Ds
4825:Mt
4822:Hs
4819:Bh
4816:Sg
4813:Db
4810:Rf
4807:Lr
4804:**
4784:Rn
4781:At
4778:Po
4712:Pt
4709:Ir
4706:Os
4703:Re
4697:Ta
4694:Hf
4661:Xe
4657:IOH
4589:Pd
4576:Ru
4563:Mo
4560:Nb
4518:Kr
4510:Se
4315:KOH
4309:Ar
4244:Ne
4240:FOH
4224:]OH
4169:He
4159:HOH
3965:pdf
3956:doi
3912:doi
3879:206
3790:doi
3778:151
3730:doi
3712:".
3678:doi
3674:531
3569:doi
3516:doi
3360:doi
3275:doi
3152:209
3061:doi
2947:doi
2943:110
2920:doi
2916:101
2838:doi
2816:291
2735:ɪər
2719:-ik
2713:PUR
2685:ɜːr
2662:not
2547:lye
2490:(L)
2354:in
2287:(X)
2265:, A
2043:·8H
2003:CrO
1797:O.
1793:·8H
1763:·2H
1759:TeO
1517:),
1513:SiO
1497:SiO
1463:).
1423:HCO
1415:+ H
1405:HCO
1355:or
1269:+ H
1251:+ H
1197:HCO
1116:is
1059:+ H
929:HCO
923:+ H
855:lye
713:HCO
623:In
586:pOH
209:961
199:CID
184:936
5174::
5073:UO
4864:*
4700:W
4681:*
4061:.
3987:.
3954:.
3944:85
3918:.
3908:47
3906:.
3881:.
3788:.
3776:.
3772:.
3728:.
3718:39
3716:.
3703:1−
3701:Mg
3672:.
3625:^
3602:^
3565:29
3563:.
3522:.
3512:54
3510:.
3496:MQ
3487:Mo
3366:.
3356:62
3354:.
3281:.
3271:62
3269:.
3232:^
3211:}}
3207:{{
3192:.
3154:.
3144:.
3119:.
3117:24
3109:.
3067:.
2976:^
2953:.
2941:.
2880:^
2852:.
2844:.
2836:.
2826:.
2814:.
2808:.
2770:,
2766:,
2741:-/
2717:OD
2688:aɪ
2668:,
2580:.
2564:,
2560:,
2518:.
2508:NH
2461:pK
2378:,
2374:,
2358:.
2335:.
2254:.
2217:,
2150:.
2062:CO
2027:.
2011:SO
1987:PO
1976:VO
1945:.
1844:−1
1827:,
1820:.
1808:PO
1771:.
1737:SO
1725:PO
1709:,
1705:,
1553:.
1541:Si
1537:(H
1525:Si
1521:(H
1509:(H
1491:a2
1484:a1
1475:.
1455:CO
1437:CO
1403:→
1363:,
1171:,
1159:.
1134:sp
1083:.
1071:,
1055:CO
1031:,
950:a2
913:CO
893:.
873:,
869:,
865:,
775:.
711:⇌
674:HF
666:pm
612:.
598:NH
574:pH
564:=
515:.
496:.
361:OH
291:OH
5134:3
5124:3
5111:3
5107:2
5098:4
5089:3
5079:2
5075:2
5066:3
5057:2
5044:4
5034:3
5019:3
5009:3
4999:3
4989:3
4979:3
4969:3
4959:3
4949:3
4940:2
4930:3
4920:3
4910:3
4900:3
4890:4
4881:3
4871:3
4798:2
4772:3
4762:4
4753:2
4743:3
4729:2
4719:3
4688:3
4675:2
4650:6
4640:3
4630:4
4621:2
4611:3
4601:2
4583:3
4570:4
4554:4
4544:3
4532:2
4504:3
4494:2
4484:3
4474:2
4464:2
4450:2
4440:2
4430:3
4421:2
4411:2
4401:3
4392:2
4382:3
4373:2
4363:4
4354:3
4345:2
4335:3
4323:2
4298:2
4288:3
4278:4
4268:3
4258:2
4233:2
4222:4
4213:3
4203:4
4193:3
4183:2
4137:e
4130:t
4123:v
4109:.
4090:.
4071:.
4045:.
4026:.
3998:.
3962:.
3958::
3950::
3926:.
3914::
3891:.
3859:.
3841:.
3798:.
3792::
3784::
3736:.
3732::
3724::
3710:2
3705:p
3698:p
3684:.
3680::
3575:.
3571::
3530:.
3518::
3501:4
3492:3
3483:2
3479:3
3374:.
3362::
3289:.
3277::
3217:)
3203:.
3164:.
3129:.
3077:.
3063::
3017:.
2961:.
2949::
2926:.
2922::
2904:2
2899:O
2895:3
2890:H
2874:.
2840::
2822::
2753:.
2738:ɪ
2732:p
2729:ˌ
2726:/
2706:/
2703:k
2700:ɪ
2697:d
2694:ɒ
2691:ˈ
2682:p
2679:ˌ
2676:/
2629:/
2626:1
2528:2
2513:2
2463:a
2451:O
2449:2
2435:a
2432:K
2424:2
2408:a
2405:K
2398:a
2393:a
2390:K
2388:p
2333:x
2329:q
2325:z
2321:O
2317:2
2312:H
2310:y
2308:·
2301:n
2295:q
2279:6
2275:6
2271:6
2267:2
2263:6
2259:3
2252:3
2248:2
2244:2
2207:3
2190:(
2188:2
2181:4
2177:4
2140:2
2136:2
2134:)
2132:3
2108:2
2104:2
2100:3
2096:2
2084:2
2080:3
2068:2
2064:3
2060:2
2045:2
2041:2
2013:4
2009:2
2005:4
2001:2
1999:H
1989:4
1985:3
1978:4
1974:3
1972:H
1962:n
1957:2
1952:n
1949:L
1932:4
1919:2
1907:3
1903:2
1899:2
1858:n
1850:2
1847:/
1842:n
1818:3
1814:2
1810:3
1806:3
1804:H
1795:2
1791:4
1783:z
1769:6
1765:2
1761:4
1757:2
1739:4
1735:2
1733:H
1727:4
1723:3
1721:H
1687:-
1591:4
1579:3
1566:2
1547:7
1545:O
1543:2
1539:6
1531:5
1529:O
1527:2
1523:2
1515:3
1511:2
1503:4
1499:4
1495:4
1488:K
1481:K
1472:n
1461:2
1457:3
1453:2
1451:H
1439:3
1435:2
1428:3
1419:O
1417:3
1410:3
1401:4
1391:(
1389:4
1338:4
1313:4
1303:3
1295:3
1263:4
1253:2
1249:3
1237:3
1217:2
1213:2
1202:3
1193:2
1189:2
1165:2
1146:2
1132:*
1130:K
1114:2
1063:O
1061:2
1057:3
1053:2
1049:2
1020:b
1017:K
1015:p
957:)
947:K
945:p
943:(
934:3
925:2
918:3
795:a
792:K
773:4
718:3
709:2
679:2
659:2
654:O
650:3
645:H
603:4
594:3
562:w
559:K
546:O
544:2
540:O
538:3
536:H
376:O
374:2
372:H
367:2
365:O
357:H
355:2
353:O
135:)
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.