1733:
46:
1498:
37:
290:
197:
1255:
825:
1866:
997:
1272:
Cyclophosphazenes such as hexachlorophosphazene are distinguished by notable stability and equal P–N bond lengths which, in many such cyclic molecules, would imply delocalization or even aromaticity. To account for these features, early bonding models starting from the mid-1950s invoked a delocalised
1956:
Among these, the best structurally characterised are the 1:1 adducts with aluminium trichloride or with gallium trichloride; they are found with the Al/Ga atom bound to a N and assume a more prominently distorted chair conformation compared to the free hexachlorophosphazene. The adducts also exhibit
1517:
at approximately 60 °C affords the pure trimer free of the tetramer. Reaction conditions such as temperature may also be tuned to maximise the yield of the trimer at the expense of the other possible products; nonetheless, commercial samples of hexachlorophosphazene usually contain appreciable
1258:
Depictions of P–N bonding in a general cyclotriphosphazene: left, a representation of alternating single and double P–N bonds (does not account for equal bond lengths), used as a matter of convention; middle, the earlier proposed delocalised ring system (discredited due to infeasibility of P
1889:
The nitrogen centres of hexachlorophosphazene are weakly basic, and this Lewis base behaviour has been suggested to play a role in the polymerisation mechanism. Specifically, hexachlorophosphazene has been reported to form adducts of various stoichiometries with Lewis acids
1100:
Occasionally, commercial or suggested practical applications have been reported, too, utilising hexachlorophosphazene as a precursor chemical. Derivatives of noted interest include the hexalkoxyphosphazene lubricants obtained from
2495:"Structural investigations of phosphorus–nitrogen compounds. 7. Relationships between physical properties, electron densities, reaction mechanisms and hydrogen-bonding motifs of N3P3Cl(6 − n)(NHBu t ) n derivatives"
1732:
1497:
833:
1224:
spectroscopy is the usual method for assaying hexachlorophosphazene and its reactions. Hexachlorophosphazene exhibits a single resonance at 20.6 ppm as all P environments are chemically equivalent.
805:
1865:
2085:
species), resulting from the nucleophilic hexasubstitution of the hexachlorophosphazene P atoms, have attracted interest for their high thermal and chemical stability as well as their low
2108:
properties have been investigated. Some of them appear promising for future applications as fibre- or membrane-forming high performance materials, since they combine transparency,
891:
1487:
1439:(HCl) side product. Since ammonium chloride is insoluble in chlorinated solvents, workup is facilitated. For the reaction under such conditions, the following
883:
2089:. Certain hexalkoxyphosphazenes (such as the hexa-phenoxy derivative) have been put to commercial use as fireproof materials and high temperature lubricants.
36:
1962:
1010:
2493:
Bartlett, Stewart W.; Coles, Simon J.; Davies, David B.; Hursthouse, Michael B.; i̇Bişogˇlu, Hanife; Kiliç, Adem; Shaw, Robert A.; Ün, İlker (2006).
1157:
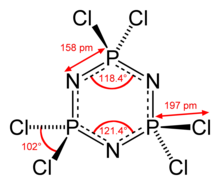
atoms bonded to each phosphorus atom. Hexachlorophosphazene molecule contains six equivalent P–N bonds, for which the adjacent P–N distances are 157
45:
134:
17:
339:
1810:. 250 °C induces polymerisation. The tetramer also polymerises in this manner, although more slowly. The conversion is a type of
1966:
1221:
887:
1005:
2564:
2368:
1873:
This polydichlorophosphazene product is the starting material for a wide class of polymeric compounds, collectively known as
704:
2605:
Heston, Amy J.; Panzner, Matthew J.; Youngs, Wiley J.; Tessier, Claire A. (2005). "Lewis Acid
Adducts of [PCl2N]3".
2139:, where the polyphosphazenes confer fire resistance, imperviousness to oils, and flexibility even at very low temperatures.
1798:
is due to the combined steric effects and oxygen lone pair π-backdonation (which deactivates already substituted P atoms).
2740:
Nielsen, Morris L.; Cranford, Garland (2007) . "Trimeric
Phosphonitrile Chloride and Tetrameric Phosphonitrile Chloride".
1296:
contribution is negligible, invalidating the earlier hypothesis. Instead, a charge separated model is generally accepted.
1097:. There is large academic interest in the compound relating to the phosphorus-nitrogen bonding and phosphorus reactivity.
1064:
1501:
The three major cyclophosphazene products resulting from the reaction of phosphorus pentachloride and ammonium chloride
1263:
participation); right, the most accurate description to current knowledge, where the majority of the bonding is ionic
2649:"A nitrogen and phosphorus enriched pyridine bridged inorganic–organic hybrid material for supercapacitor application"
2073:
rings. Solid films of the trimer and tetramer will not undergo any chemical change under such irradiation conditions.
1395:
Modern syntheses are based on the developments by Schenk and Römer who used ammonium chloride in place of ammonia and
2777:
2757:
2475:
304:
2816:
2012:
in chloroform, though for this application the tetramer octachlorotetraphosphazene usually proves more effective.
1017:
2148:
Discovery of cyclophosphazenes: Liebig-Wöhler, Briefwechsel vol. 1, 63; Ann. Chem. (Liebig), vol. 11 (1834), 146.
1292:
Starting from the late 1980s, more modern calculations and the lack of spectroscopic evidence reveal that the P 3
2801:
1177:
786:
435:
2170:"Syntheses and structures of cyclic and short-chain linear phosphazenes bearing 4-phenylphenoxy side groups"
2806:
2086:
1869:
Hexachlorotriphosphazene ROP and subsequent nucleophilic substitution for desired polyphosphazene synthesis
861:
247:
192:
2168:
Allcock, Harry R.; Ngo, Dennis C.; Parvez, Masood; Whittle, Robert R.; Birdsall, William J. (1991-03-01).
2158:
2152:
154:
2821:
2796:
1811:
268:
875:
1491:
1109:, or chemically resistant inorganic polymers with desirable thermal and mechanical properties known as
2121:
2093:
1834:
1741:
1428:
1311:
1102:
946:
204:
2648:
1818:, but is overall not very well understood. Prolonged heating of the polymer at higher temperatures (
1736:
A SN2 substitution at hexachlorotriphosphazene. A trigonal bipyramidal transition state is proposed.
1581:
1527:
1335:
1195:
961:
731:
675:
285:
1936:
1695:
756:
539:
2166:
Example of hexalkoxyphosphazene synthesis from hexachlorophosphazene and structure description:
1299:
According to this description, the P–N bond is viewed as a very polarised one (between notional
1235:
stretches. Other bands are found at 860 and 500–600 cm, respectively assigned to ring and ν
2470:
Mark, J. E.; Allcock, H. R.; West, R. “Inorganic
Polymers” Prentice Hall, Englewood, NJ: 1992.
956:
1405:. By replacing ammonia with ammonium chloride allows the reaction to proceed without a strong
824:
2826:
2360:
1815:
1745:
1600:
1243:
1228:
1210:
1080:
941:
847:
817:
58:
1242:
Hexachlorophosphazene and many of its derivatives have been characterized by single crystal
174:
1927:
1585:
464:
256:
1526:
The mechanism of the above reaction has not been resolved, but it has been suggested that
879:
8:
1909:
1636:
1551:
1406:
1357:
289:
196:
110:
100:
1961:
behaviour in solution for temperatures down to −60 °C, which can be monitored with
1331:
2676:
2231:
2055:
2001:
derivatives, postulated to go through a cyclophosphazene pyridinium salt intermediate.
1958:
1891:
1620:
1514:
1506:
1385:
1076:
1033:
669:
612:
2811:
2773:
2753:
2680:
2668:
2622:
2570:
2560:
2516:
2471:
2430:
2422:
2364:
2297:
2223:
2189:
2097:
1978:
1945:
1900:
1589:
1572:
1436:
1361:
1327:
577:
2235:
2772:
Holleman, A. F.; Wiberg, E. "Inorganic
Chemistry" Academic Press: San Diego, 2001.
2745:
2707:
2660:
2614:
2506:
2414:
2352:
2289:
2277:
2215:
2181:
2005:
1977:
Hexachlorophosphazene has also found applications in research by enabling aromatic
1823:
1795:
1752:
by alkoxide proceeds via displacement of chloride at separate phosphorus centers:
1396:
1194:
ring in hexachlorophosphazene deviates from planarity and is slightly ruffled (see
1037:
895:
627:
552:
449:
362:
236:
2278:"Regio- and stereochemical control in substitution reactions of cyclophosphazenes"
2051:
1918:
1874:
1854:~ 15000. It was first observed in the late 19th century and its form after chain
1770:
1699:
1131:
1110:
1060:
951:
684:
2749:
2556:
Phosphorus-nitrogen compounds ; cyclic, linear, and high polymeric systems
2136:
2113:
2109:
2021:
1998:
1877:. Substitution of the chloride groups by other nucleophilic groups, especially
1315:
1281:
1166:
1114:
988:
2511:
2494:
2200:
Novel hexalkoxyphosphazene synthesis not starting from hexachlorophosphazene:
1254:
867:
2790:
2711:
2672:
2574:
2426:
2301:
2227:
2193:
2105:
1830:
1440:
1432:
1307:), with sufficient ionic character to account for most of the bond strength.
424:
414:
185:
85:
2,2,4,4,6,6-hexachloro-2,2,4,4,6,6-hexahydro-1,3,5,2,4,6-triazatriphosphorine
1360:
to yield a new substance that could be washed with cold water to remove the
2626:
2520:
2434:
2009:
1726:
1719:
1597:
1593:
1424:
652:
583:
2554:
2219:
1547:(tetrachlorophosphonium hexachlorophosphate(V)) and the reaction proceeds
1399:
1090:
966:
918:
899:
591:
490:
2293:
2203:
2185:
2024:
react forming clear liquids identified as alkyl-substituted derivatives
2664:
2157:. American Chemical Journal, vol. 17, p. 275.H. N. Stokes (1896),
2117:
1855:
1389:
1373:
1150:
1068:
1045:
512:
486:
460:
383:
205:
165:
2618:
2418:
2169:
903:
2132:
2101:
1859:
1158:
853:
2725:
R. Klement (1963). "Phosphonitrilic
Chlorides". In G. Brauer (ed.).
2695:
2402:
2401:
Chaplin, Adrian B.; Harrison, John A.; Dyson, Paul J. (2005-11-01).
987:
Except where otherwise noted, data are given for materials in their
1982:
1878:
1749:
1722:
1702:
1510:
1402:
1381:
1377:
1154:
1146:
1106:
1072:
1048:
1042:
976:
2092:
Polyphosphazenes obtained from polymerised hexachlorophosphazene (
911:
133:
2082:
1563:
1346:
1094:
971:
871:
404:
223:
1490:), 3 (the trimer hexachlorotriphosphazene), and 4 (the tetramer
1330:
in 1834. In that report he describes experiments conducted with
2128:
1994:
1231:, the 1370 and 1218 cm vibrational bands are assigned to ν
516:
145:
1881:
as laid out above, yields numerous characterised derivatives.
2492:
1326:
The synthesis of hexachlorophosphazene was first reported by
691:
123:
2151:
First reports on their polymerisation: H. N. Stokes (1895),
1310:
The rest (~15%) of the bond strength may be attributed to a
273:
2204:"A Novel Synthesis of Hexasubstituted Cyclotriphosphazenes"
419:
112 to 114 °C (234 to 237 °F; 385 to 387 K)
2604:
62:
2,2,4,4,6,6-Hexachloro-1,3,5,2λ,4λ,6λ-triazatriphosphinine
2160:
2163:
American
Chemical Journal, vol. 18 issue 8, p. 629.
2167:
1518:
amounts of octachlorotetraphosphazene, even up to 40%.
2202:
Ye, Chengfeng; Zhang, Zefu; Liu, Weimin (2002-01-01).
2020:
Both the trimer and tetramer in hydrocarbon solutions
2403:"Revisiting the Electronic Structure of Phosphazenes"
1814:(ROP). The ROP mechanism is found to be catalysed by
2744:. Inorganic Syntheses. Vol. 6. pp. 94–97.
2499:
2400:
2729:. Vol. 1. NY, NY: Academic Press. p. 575.
2727:
2127:Polyphosphazene-based components have been used in
1635:and subsequent HCl elimination, creates a growing
2351:
1318:into π-accepting σ* molecular orbitals on the P.
1120:
1089:(phosphazyl dichloride). Its classification as a
2788:
1125:
235:
2739:
2647:Dhiman, Nisha; Mohanty, Paritosh (2019-10-28).
2050:= 3, 4. Such reactions proceed under prolonged
313:InChI=1S/Cl6N3P3/c1-10(2)7-11(3,4)9-12(5,6)8-10
109:
2552:
2096:) have garnered attention within the field of
1858:has been called "inorganic rubber" due to its
1801:
1161:. This is characteristically shorter than the
907:
323:InChI=1/Cl6N3P3/c1-10(2)7-11(3,4)9-12(5,6)8-10
2646:
1505:Purification by sublimation gives mainly the
2015:
1165:. 177 pm P–N bonds in the valence saturated
1216:
2724:
2347:
2345:
2343:
2341:
2339:
2337:
2335:
2333:
2331:
2201:
2081:The hexalkoxyphosphazenes (especially the
1718:Hexachlorophosphazene reacts readily with
288:
195:
173:
2693:
2510:
2329:
2327:
2325:
2323:
2321:
2319:
2317:
2315:
2313:
2311:
255:
2174:Journal of the American Chemical Society
1486:can usually take values of 2 (the dimer
1372:) coproduct. The new compound contained
1287:
1273:π system arising from the overlap of N 2
1253:
1176:symmetry, and each phosphorus center is
284:
14:
2789:
2308:
1521:
186:
2466:
2464:
2275:
2058:) illumination without affecting the
1698:leads to the formation of one of the
316:Key: UBIJTWDKTYCPMQ-UHFFFAOYSA-N
153:
2642:
2640:
2638:
2636:
2600:
2598:
2596:
2594:
2592:
2590:
2588:
2586:
2584:
2548:
2546:
2544:
2542:
2540:
2538:
2536:
2534:
2532:
2530:
2488:
2486:
2484:
2462:
2460:
2458:
2456:
2454:
2452:
2450:
2448:
2446:
2444:
2396:
2394:
2392:
2390:
2388:
2386:
2384:
2382:
2380:
2276:Allen, Christopher W. (1991-03-01).
2271:
2269:
2267:
2265:
2263:
2261:
2259:
2257:
2255:
2253:
1713:
2154:On the chloronitrides of phosphorus
2004:The compound may also be used as a
1972:
1067:backbone consisting of alternating
326:Key: UBIJTWDKTYCPMQ-UHFFFAOYAJ
226:
24:
2142:
1864:
1731:
1496:
1314:interaction: the N lone pairs can
25:
2838:
2633:
2581:
2527:
2481:
2441:
2377:
2250:
1884:
1806:Heating hexachlorophosphazene to
1267:
347:N1=P(N=P(N=P1(Cl)Cl)(Cl)Cl)(Cl)Cl
1997:, resulting in 4,4'-substituted
1627:which through further attack of
995:
823:
44:
35:
2766:
2733:
2718:
2076:
1944:, but no isolable product with
1829:The structure of the inorganic
1093:highlights its relationship to
991:(at 25 °C , 100 kPa).
73:Phosphonitrilic chloride trimer
2687:
1213:species is completely planar.
1180:with a Cl–P–Cl angle of 101°.
1121:Structure and characterisation
1105:of hexachlorophosphazene with
1075:atoms, and can be viewed as a
787:Occupational safety and health
13:
1:
2243:
2008:reagent for the synthesis of
1126:Bond lengths and conformation
79:Hexachlorocyclotriphosphazene
18:Hexachlorocyclotriphosphazene
2559:. New York: Academic Press.
2087:glass transition temperature
1708:
1562:(tetrachlorophosphonium) by
1321:
1316:donate some electron density
7:
1812:ring-opening polymerisation
1802:Ring-opening polymerisation
1535:is found in its ionic form
1130:Hexachlorophosphazene is a
82:Triphosphonitrilic chloride
10:
2843:
2750:10.1002/9780470132371.ch28
1822:. 350 °C) will cause
1492:octachlorotetraphosphazene
1388:. It was sensitive toward
1249:
1153:atoms, and two additional
1117:of hexachlorophosphazene.
429:decomposes (above 167 °C)
2512:10.1107/S0108768106000851
2357:Chemistry of the Elements
2355:; Earnshaw, Alan (1997).
2094:poly(dichlorophosphazene)
2016:Photochemical degradation
1835:Poly(dichlorophosphazene)
1429:1,1,2,2-tetrachloroethane
1312:negative hyperconjugation
1103:nucleophilic substitution
985:
947:Poly(dichlorophosphazene)
927:
804:
784:
779:
697:
570:
355:
335:
300:
93:
67:
57:
52:
43:
34:
2712:10.1002/jlac.18340110202
2696:"Nachtrag der Redaction"
2653:New Journal of Chemistry
2208:Synthetic Communications
1488:tetrachlorodiphosphazene
1336:phosphorus pentachloride
1217:Characterisation methods
1172:The molecule possesses D
962:Tetrasulfur tetranitride
862:Precautionary statements
732:Enthalpy of vaporization
76:Hexachlorotriphosphazene
2817:Phosphorus heterocycles
2553:Allcock, H. R. (1972).
757:Enthalpy of sublimation
540:Magnetic susceptibility
1870:
1837:) comprises a linear –
1737:
1502:
1264:
1145:core with alternating
957:Trithiazyl trichloride
641: = 12.83 Å,
637: = 13.87 Å,
530:50.7 wt % (60 °C)
527:38.9 wt % (40 °C)
524:27.7 wt % (20 °C)
504:53.7 wt % (60 °C)
501:36.8 wt % (40 °C)
498:22.3 wt % (20 °C)
478:39.2 wt % (60 °C)
475:35.6 wt % (40 °C)
472:24.5 wt % (20 °C)
30:Hexachlorophosphazene
2802:Nitrogen heterocycles
2361:Butterworth-Heinemann
2353:Greenwood, Norman N.
2220:10.1081/SCC-120002003
2112:flexibility, tunable
1868:
1765:+ 3 NaOR → (NPCl(OR))
1735:
1696:intramolecular attack
1596:) creates a reactive
1500:
1435:, which tolerate the
1288:Modern bonding models
1257:
1244:X-ray crystallography
1211:hexafluorophosphazene
1081:hypothetical compound
1059:. The molecule has a
1030:Hexachlorophosphazene
942:Hexafluorophosphazene
2122:desirable properties
2120:, and various other
1993:-dialkylanilines or
1409:associated with the
1209:ring in the related
1198:). By contrast, the
465:carbon tetrachloride
2807:Inorganic compounds
2742:Inorganic Syntheses
2659:(42): 16670–16675.
2607:Inorganic Chemistry
2407:Inorganic Chemistry
2294:10.1021/cr00002a002
2186:10.1021/ja00007a041
1552:nucleophilic attack
1522:Formation mechanism
1407:exothermic reaction
678:(slightly ruffled)
645: = 6.09 Å
450:Solubility in water
443:60 °C at 0.05 Torr
409:1.98 g/mL at 25 °C
391: g·mol
31:
2822:Six-membered rings
2797:Chlorine compounds
2694:J. Liebig (1834).
2665:10.1039/C9NJ03976G
2098:inorganic polymers
1981:reactions between
1871:
1781:+ 3 NaOR → (NP(OR)
1738:
1694:until an eventual
1515:vacuum sublimation
1503:
1386:elemental analysis
1384:, on the basis of
1334:. They found that
1265:
1196:chair conformation
1113:produced from the
1034:inorganic compound
1018:Infobox references
928:Related compounds
29:
2619:10.1021/ic050974y
2613:(19): 6518–6520.
2566:978-0-323-14751-4
2419:10.1021/ic0511266
2413:(23): 8407–8417.
2370:978-0-08-037941-8
1714:Substitution at P
1437:hydrogen chloride
1362:ammonium chloride
1026:Chemical compound
1024:
1023:
934:Related compounds
848:Hazard statements
578:Crystal structure
399:colourless solid
269:CompTox Dashboard
135:Interactive image
16:(Redirected from
2834:
2781:
2770:
2764:
2763:
2737:
2731:
2730:
2722:
2716:
2715:
2691:
2685:
2684:
2644:
2631:
2630:
2602:
2579:
2578:
2550:
2525:
2524:
2514:
2490:
2479:
2468:
2439:
2438:
2398:
2375:
2374:
2359:(2nd ed.).
2349:
2306:
2305:
2282:Chemical Reviews
2273:
2239:
2197:
2180:(7): 2628–2634.
2072:
2045:
2006:peptide coupling
1973:Coupling reagent
1951:
1942:
1933:
1924:
1915:
1906:
1897:
1875:polyphosphazenes
1849:
1824:depolymerisation
1796:regioselectivity
1790:
1773:
1686:
1661:
1634:
1623:
1579:
1569:
1561:
1546:
1533:
1478:
1422:
1415:
1371:
1355:
1344:
1306:
1302:
1208:
1193:
1144:
1111:polyphosphazenes
1088:
1058:
1038:chemical formula
1008:
1002:
999:
998:
913:
909:
905:
901:
897:
893:
889:
885:
881:
877:
873:
869:
855:
827:
771:
746:
721:
698:Thermochemistry
628:Lattice constant
605:
604:
553:Refractive index
390:
377:
363:Chemical formula
293:
292:
277:
275:
259:
239:
228:
207:
199:
188:
177:
157:
137:
113:
48:
39:
32:
28:
21:
2842:
2841:
2837:
2836:
2835:
2833:
2832:
2831:
2787:
2786:
2785:
2784:
2771:
2767:
2760:
2738:
2734:
2723:
2719:
2692:
2688:
2645:
2634:
2603:
2582:
2567:
2551:
2528:
2491:
2482:
2469:
2442:
2399:
2378:
2371:
2350:
2309:
2274:
2251:
2246:
2145:
2143:Further reading
2137:shock absorbers
2079:
2071:
2065:
2059:
2044:
2038:
2032:
2025:
2022:photochemically
2018:
1975:
1950:
1946:
1941:
1937:
1932:
1928:
1923:
1919:
1914:
1910:
1905:
1901:
1896:
1892:
1887:
1848:
1842:
1838:
1804:
1788:
1784:
1780:
1776:
1768:
1764:
1760:
1756:
1716:
1711:
1684:
1680:
1676:
1672:
1668:
1664:
1659:
1655:
1651:
1647:
1643:
1632:
1628:
1618:
1614:
1610:
1606:
1577:
1573:
1568:
1564:
1559:
1555:
1544:
1540:
1536:
1532:
1528:
1524:
1472:
1466:
1462:
1454:
1447:
1421:
1417:
1414:
1410:
1369:
1365:
1354:
1350:
1343:
1339:
1324:
1304:
1300:
1290:
1270:
1252:
1238:
1234:
1219:
1207:
1203:
1199:
1192:
1188:
1184:
1175:
1143:
1139:
1135:
1134:, containing a
1132:cyclic molecule
1128:
1123:
1087:
1083:
1057:
1053:
1040:
1027:
1020:
1015:
1014:
1013: ?)
1004:
1000:
996:
992:
981:
952:Polyphosphazene
935:
864:
850:
836:
820:
797:
772:
769:
763:
759:
747:
744:
738:
734:
722:
719:
713:
709:
706:
705:Std enthalpy of
687:
672:
670:Molecular shape
660:
646:
630:
621:
615:
603:
600:
599:
598:
594:
580:
563:
561:
546:−149×10 cm/mol
543:
533:
507:
481:
452:
440:
437:
388:
376:
372:
368:
365:
351:
348:
343:
342:
331:
328:
327:
324:
318:
317:
314:
308:
307:
296:
278:
271:
262:
242:
229:
217:
180:
160:
140:
127:
116:
103:
89:
88:
63:
23:
22:
15:
12:
11:
5:
2840:
2830:
2829:
2824:
2819:
2814:
2809:
2804:
2799:
2783:
2782:
2765:
2758:
2732:
2717:
2686:
2632:
2580:
2565:
2526:
2505:(2): 321–329.
2480:
2440:
2376:
2369:
2307:
2288:(2): 119–135.
2248:
2247:
2245:
2242:
2241:
2240:
2214:(2): 203–209.
2198:
2164:
2149:
2144:
2141:
2118:hydrophobicity
2114:hydrophilicity
2078:
2075:
2067:
2061:
2040:
2034:
2027:
2017:
2014:
1999:phenylpyridine
1974:
1971:
1948:
1939:
1930:
1921:
1912:
1903:
1894:
1886:
1885:Lewis basicity
1883:
1844:
1840:
1803:
1800:
1792:
1791:
1786:
1782:
1778:
1774:
1766:
1762:
1758:
1715:
1712:
1710:
1707:
1692:
1691:
1682:
1678:
1674:
1670:
1666:
1662:
1657:
1653:
1649:
1645:
1630:
1625:
1624:
1616:
1612:
1608:
1575:
1566:
1557:
1542:
1538:
1530:
1523:
1520:
1480:
1479:
1468:
1464:
1460:
1452:
1419:
1412:
1392:by hot water.
1367:
1358:exothermically
1352:
1341:
1323:
1320:
1289:
1286:
1269:
1268:Early analyses
1266:
1251:
1248:
1236:
1232:
1218:
1215:
1205:
1201:
1190:
1186:
1173:
1141:
1137:
1127:
1124:
1122:
1119:
1115:polymerisation
1085:
1055:
1051:
1025:
1022:
1021:
1016:
994:
993:
989:standard state
986:
983:
982:
980:
979:
974:
969:
964:
959:
954:
949:
944:
938:
936:
933:
930:
929:
925:
924:
923:Non-flammable
921:
915:
914:
892:P305+P351+P338
884:P303+P361+P353
880:P301+P330+P331
865:
860:
857:
856:
851:
846:
843:
842:
837:
832:
829:
828:
821:
816:
813:
812:
802:
801:
800:mild irritant
798:
795:
792:
791:
782:
781:
777:
776:
773:
767:
761:
755:
752:
751:
748:
742:
736:
730:
727:
726:
725:−812.4 kJ/mol
723:
717:
711:
703:
700:
699:
695:
694:
688:
683:
680:
679:
673:
668:
665:
664:
661:
651:
648:
647:
633:
631:
626:
623:
622:
619:
616:
611:
608:
607:
601:
595:
590:
587:
586:
581:
576:
573:
572:
568:
567:
566:1.62 (589 nm)
564:
559:
551:
548:
547:
544:
538:
535:
534:
532:
531:
528:
525:
521:
519:
509:
508:
506:
505:
502:
499:
495:
493:
483:
482:
480:
479:
476:
473:
469:
467:
457:
456:
453:
448:
445:
444:
441:
434:
431:
430:
427:
421:
420:
417:
411:
410:
407:
401:
400:
397:
393:
392:
386:
380:
379:
374:
370:
366:
361:
358:
357:
353:
352:
350:
349:
346:
338:
337:
336:
333:
332:
330:
329:
325:
322:
321:
319:
315:
312:
311:
303:
302:
301:
298:
297:
295:
294:
281:
279:
267:
264:
263:
261:
260:
252:
250:
244:
243:
241:
240:
232:
230:
222:
219:
218:
216:
215:
211:
209:
201:
200:
190:
182:
181:
179:
178:
170:
168:
162:
161:
159:
158:
150:
148:
142:
141:
139:
138:
130:
128:
121:
118:
117:
115:
114:
106:
104:
99:
96:
95:
91:
90:
87:
86:
83:
80:
77:
74:
70:
69:
65:
64:
61:
55:
54:
50:
49:
41:
40:
9:
6:
4:
3:
2:
2839:
2828:
2825:
2823:
2820:
2818:
2815:
2813:
2810:
2808:
2805:
2803:
2800:
2798:
2795:
2794:
2792:
2779:
2778:0-12-352651-5
2775:
2769:
2761:
2759:9780470132371
2755:
2751:
2747:
2743:
2736:
2728:
2721:
2713:
2709:
2705:
2701:
2697:
2690:
2682:
2678:
2674:
2670:
2666:
2662:
2658:
2654:
2650:
2643:
2641:
2639:
2637:
2628:
2624:
2620:
2616:
2612:
2608:
2601:
2599:
2597:
2595:
2593:
2591:
2589:
2587:
2585:
2576:
2572:
2568:
2562:
2558:
2557:
2549:
2547:
2545:
2543:
2541:
2539:
2537:
2535:
2533:
2531:
2522:
2518:
2513:
2508:
2504:
2500:
2496:
2489:
2487:
2485:
2477:
2476:0-13-465881-7
2473:
2467:
2465:
2463:
2461:
2459:
2457:
2455:
2453:
2451:
2449:
2447:
2445:
2436:
2432:
2428:
2424:
2420:
2416:
2412:
2408:
2404:
2397:
2395:
2393:
2391:
2389:
2387:
2385:
2383:
2381:
2372:
2366:
2362:
2358:
2354:
2348:
2346:
2344:
2342:
2340:
2338:
2336:
2334:
2332:
2330:
2328:
2326:
2324:
2322:
2320:
2318:
2316:
2314:
2312:
2303:
2299:
2295:
2291:
2287:
2283:
2279:
2272:
2270:
2268:
2266:
2264:
2262:
2260:
2258:
2256:
2254:
2249:
2237:
2233:
2229:
2225:
2221:
2217:
2213:
2209:
2205:
2199:
2195:
2191:
2187:
2183:
2179:
2175:
2171:
2165:
2162:
2161:
2156:
2155:
2150:
2147:
2146:
2140:
2138:
2134:
2130:
2125:
2123:
2119:
2115:
2111:
2107:
2106:thermoplastic
2103:
2099:
2095:
2090:
2088:
2084:
2074:
2070:
2064:
2057:
2053:
2049:
2043:
2037:
2031:
2023:
2013:
2011:
2010:oligopeptides
2007:
2002:
2000:
1996:
1992:
1988:
1984:
1980:
1970:
1968:
1964:
1960:
1954:
1952:
1943:
1934:
1925:
1916:
1907:
1898:
1882:
1880:
1876:
1867:
1863:
1861:
1857:
1856:cross-linking
1853:
1850:chain, where
1847:
1836:
1832:
1831:chloropolymer
1827:
1825:
1821:
1817:
1813:
1809:
1799:
1797:
1794:The observed
1775:
1772:
1755:
1754:
1753:
1751:
1747:
1743:
1734:
1730:
1728:
1724:
1721:
1706:
1704:
1701:
1697:
1690:
1663:
1642:
1641:
1640:
1639:intermediate
1638:
1622:
1605:
1604:
1603:
1602:
1599:
1595:
1591:
1587:
1583:
1580:
1570:
1553:
1550:
1534:
1519:
1516:
1512:
1508:
1499:
1495:
1493:
1489:
1485:
1476:
1471:
1458:
1450:
1446:
1445:
1444:
1442:
1441:stoichiometry
1438:
1434:
1433:chlorobenzene
1430:
1427:solvents are
1426:
1408:
1404:
1401:
1398:
1393:
1391:
1387:
1383:
1379:
1375:
1363:
1359:
1348:
1337:
1333:
1329:
1319:
1317:
1313:
1308:
1297:
1295:
1285:
1283:
1280:
1276:
1262:
1256:
1247:
1245:
1240:
1230:
1225:
1223:
1214:
1212:
1197:
1181:
1179:
1170:
1168:
1164:
1160:
1156:
1152:
1148:
1133:
1118:
1116:
1112:
1108:
1104:
1098:
1096:
1092:
1082:
1078:
1074:
1070:
1066:
1062:
1050:
1047:
1044:
1039:
1035:
1031:
1019:
1012:
1007:
990:
984:
978:
975:
973:
970:
968:
965:
963:
960:
958:
955:
953:
950:
948:
945:
943:
940:
939:
937:
932:
931:
926:
922:
920:
917:
916:
866:
863:
859:
858:
852:
849:
845:
844:
841:
838:
835:
831:
830:
826:
822:
819:
815:
814:
810:
808:
803:
799:
794:
793:
789:
788:
783:
778:
774:
766:
758:
754:
753:
749:
741:
733:
729:
728:
724:
716:
708:
702:
701:
696:
693:
689:
686:
685:Dipole moment
682:
681:
677:
674:
671:
667:
666:
662:
658:
654:
653:Formula units
650:
649:
644:
640:
636:
632:
629:
625:
624:
617:
614:
610:
609:
596:
593:
589:
588:
585:
582:
579:
575:
574:
569:
565:
558:
554:
550:
549:
545:
541:
537:
536:
529:
526:
523:
522:
520:
518:
514:
511:
510:
503:
500:
497:
496:
494:
492:
488:
485:
484:
477:
474:
471:
470:
468:
466:
462:
459:
458:
454:
451:
447:
446:
442:
439:
433:
432:
428:
426:
425:Boiling point
423:
422:
418:
416:
415:Melting point
413:
412:
408:
406:
403:
402:
398:
395:
394:
387:
385:
382:
381:
367:
364:
360:
359:
354:
345:
344:
341:
334:
320:
310:
309:
306:
299:
291:
287:
286:DTXSID4061331
283:
282:
280:
270:
266:
265:
258:
254:
253:
251:
249:
246:
245:
238:
234:
233:
231:
225:
221:
220:
213:
212:
210:
208:
203:
202:
198:
194:
191:
189:
187:ECHA InfoCard
184:
183:
176:
172:
171:
169:
167:
164:
163:
156:
155:ChEMBL2022081
152:
151:
149:
147:
144:
143:
136:
132:
131:
129:
125:
120:
119:
112:
108:
107:
105:
102:
98:
97:
92:
84:
81:
78:
75:
72:
71:
66:
60:
56:
51:
47:
42:
38:
33:
27:
19:
2827:Phosphazenes
2768:
2741:
2735:
2726:
2720:
2703:
2699:
2689:
2656:
2652:
2610:
2606:
2555:
2502:
2498:
2410:
2406:
2356:
2285:
2281:
2211:
2207:
2177:
2173:
2159:
2153:
2126:
2091:
2080:
2077:Applications
2068:
2062:
2047:
2041:
2035:
2029:
2019:
2003:
1990:
1986:
1976:
1955:
1888:
1872:
1851:
1845:
1828:
1819:
1807:
1805:
1793:
1746:substitution
1742:nucleophilic
1739:
1720:alkali metal
1717:
1693:
1688:
1626:
1601:intermediate
1598:nucleophilic
1594:side product
1582:dissociation
1548:
1525:
1504:
1483:
1481:
1474:
1469:
1456:
1448:
1425:chlorocarbon
1394:
1325:
1309:
1298:
1293:
1291:
1278:
1274:
1271:
1260:
1241:
1226:
1220:
1182:
1171:
1162:
1129:
1099:
1029:
1028:
839:
806:
796:Main hazards
785:
775:76.2 kJ/mol
764:
750:55.2 kJ/mol
739:
714:
656:
642:
638:
634:
584:orthorhombic
556:
94:Identifiers
68:Other names
26:
2706:: 139–150.
2102:elastomeric
2056:mercury arc
1985:and either
1860:elastomeric
1816:Lewis acids
1652:] → [Cl
1592:(the major
1586:Elimination
1400:chlorinated
1229:IR spectrum
1178:tetrahedral
1169:analogues.
1167:phosphazane
1091:phosphazene
1065:unsaturated
967:Polythiazyl
919:Flash point
834:Signal word
790:(OHS/OSH):
613:Point group
597:62 (Pnma, D
592:Space group
491:cyclohexane
455:decomposes
436:Sublimation
396:Appearance
356:Properties
193:100.012.160
2791:Categories
2700:Ann. Pharm
2244:References
2133:fuel lines
1862:behaviour.
1777:(NPCl(OR))
1677:] → HN=PCl
1648:+ [PCl
1615:] → HN=PCl
1611:+ [PCl
1423:. Typical
1390:hydrolysis
1328:von Liebig
1151:phosphorus
1069:phosphorus
818:Pictograms
571:Structure
513:Solubility
487:Solubility
461:Solubility
438:conditions
384:Molar mass
257:7VR28MTM9D
166:ChemSpider
122:3D model (
101:CAS Number
59:IUPAC name
2681:208761169
2673:1369-9261
2575:838102247
2427:0020-1669
2302:0009-2665
2228:0039-7911
2194:0002-7863
1959:fluxional
1879:alkoxides
1839:(N=P(−Cl)
1833:product (
1723:alkoxides
1709:Reactions
1703:oligomers
1685:+ HCl + H
1669:+ [Cl
1541:][PCl
1443:applies:
1322:Synthesis
1107:alkoxides
1036:with the
888:P304+P340
809:labelling
707:formation
214:213-376-8
206:EC Number
2812:Nitrides
2627:16156607
2521:16552166
2435:16270979
2236:97319633
2110:backbone
2046:, where
1983:pyridine
1979:coupling
1789:+ 3 NaCl
1750:chloride
1629:[PCl
1619:+ HCl +
1556:[PCl
1537:[PCl
1511:tetramer
1403:solvents
1356:) react
1282:orbitals
1155:chlorine
1147:nitrogen
1073:nitrogen
977:Borazine
780:Hazards
542:(χ)
111:940-71-6
2129:O-rings
2083:aryloxy
1673:P−N=PCl
1660:] + HCl
1656:P−N=PCl
1637:acyclic
1574:[NH
1513:. Slow
1463:→ (NPCl
1451:[NH
1366:[NH
1347:ammonia
1277:and P 3
1250:Bonding
1095:benzene
1079:of the
1011:what is
1009: (
972:Benzene
405:Density
378:
224:PubChem
2776:
2756:
2679:
2671:
2625:
2573:
2563:
2519:
2474:
2433:
2425:
2367:
2300:
2234:
2226:
2192:
2100:. The
1995:indole
1727:amides
1700:cyclic
1681:−N=PCl
1644:HN=PCl
1571:(from
1507:trimer
1482:where
1455:]Cl +
1380:, and
1345:) and
1332:Wöhler
1227:In it
1077:trimer
1061:cyclic
1032:is an
1006:verify
1003:
840:Danger
768:sublim
517:xylene
389:347.64
340:SMILES
237:220225
175:190959
146:ChEMBL
53:Names
2677:S2CID
2232:S2CID
2026:(NPCl
1967:P-NMR
1757:(NPCl
1397:inert
1222:P-NMR
1084:N≡PCl
676:chair
369:(NPCl
305:InChI
124:JSmol
2774:ISBN
2754:ISBN
2669:ISSN
2623:PMID
2571:OCLC
2561:ISBN
2517:PMID
2472:ISBN
2431:PMID
2423:ISSN
2365:ISBN
2298:ISSN
2224:ISSN
2190:ISSN
2135:and
2104:and
1965:and
1938:VOCl
1929:TaCl
1911:GaCl
1902:AlBr
1893:AlCl
1771:NaCl
1769:+ 3
1744:poly
1740:The
1725:and
1689:etc.
1509:and
1303:and
1237:P–Cl
1183:The
1149:and
1071:and
912:P501
908:P405
904:P363
900:P321
896:P310
876:P280
872:P264
868:P260
854:H314
248:UNII
2746:doi
2708:doi
2661:doi
2615:doi
2507:doi
2415:doi
2290:doi
2216:doi
2182:doi
2178:113
2116:or
2052:UVC
1947:BCl
1748:of
1590:HCl
1588:of
1584:).
1578:]Cl
1554:of
1549:via
1529:PCl
1494:).
1477:HCl
1459:PCl
1431:or
1418:PCl
1370:]Cl
1340:PCl
1233:P–N
807:GHS
743:vap
718:298
515:in
489:in
463:in
274:EPA
227:CID
2793::
2752:.
2704:11
2702:.
2698:.
2675:.
2667:.
2657:43
2655:.
2651:.
2635:^
2621:.
2611:44
2609:.
2583:^
2569:.
2529:^
2515:.
2503:62
2501:.
2497:.
2483:^
2443:^
2429:.
2421:.
2411:44
2409:.
2405:.
2379:^
2363:.
2310:^
2296:.
2286:91
2284:.
2280:.
2252:^
2230:.
2222:.
2212:32
2210:.
2206:.
2188:.
2176:.
2172:.
2131:,
2124:.
2028:2−
1969:.
1953:.
1935:,
1926:,
1920:SO
1917:,
1908:,
1899:,
1843:−)
1826:.
1820:ca
1808:ca
1729:.
1705:.
1687:,
1665:NH
1607:NH
1565:NH
1473:+
1411:NH
1382:Cl
1376:,
1351:NH
1284:.
1246:.
1239:.
1174:3h
1163:ca
1159:pm
1063:,
1049:Cl
910:,
906:,
902:,
898:,
894:,
890:,
886:,
882:,
878:,
874:,
870:,
811::
760:(Δ
735:(Δ
710:(Δ
690:0
663:4
620:3h
606:)
602:2h
2780:.
2762:.
2748::
2714:.
2710::
2683:.
2663::
2629:.
2617::
2577:.
2523:.
2509::
2478:.
2437:.
2417::
2373:.
2304:.
2292::
2238:.
2218::
2196:.
2184::
2069:n
2066:N
2063:n
2060:P
2054:(
2048:n
2042:n
2039:)
2036:x
2033:R
2030:x
1991:N
1989:,
1987:N
1963:N
1949:3
1940:3
1931:5
1922:3
1913:3
1904:3
1895:3
1852:n
1846:n
1841:2
1787:3
1785:)
1783:2
1779:3
1767:3
1763:3
1761:)
1759:2
1683:3
1679:2
1675:3
1671:3
1667:3
1658:3
1654:3
1650:4
1646:3
1633:]
1631:4
1621:H
1617:3
1613:4
1609:3
1576:4
1567:3
1560:]
1558:4
1545:]
1543:6
1539:4
1531:5
1484:n
1475:n
1470:n
1467:)
1465:2
1461:5
1457:n
1453:4
1449:n
1420:5
1416:/
1413:3
1378:N
1374:P
1368:4
1364:(
1353:3
1349:(
1342:5
1338:(
1305:N
1301:P
1294:d
1279:d
1275:p
1261:d
1259:3
1206:3
1204:N
1202:3
1200:P
1191:3
1189:N
1187:3
1185:P
1142:3
1140:N
1138:3
1136:P
1086:2
1056:3
1054:)
1052:2
1046:P
1043:N
1041:(
1001:N
770:)
765:H
762:f
745:)
740:H
737:f
720:)
715:H
712:f
692:D
659:)
657:Z
655:(
643:c
639:b
635:a
618:D
562:)
560:D
557:n
555:(
375:3
373:)
371:2
276:)
272:(
126:)
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.