186:
731:
33:
1146:
1494:
1456:
1447:
792:
1321:
926:
67:
5201:
3988:
5225:
4012:
5237:
5213:
4000:
1181:
802:
amounts to be effective and require additional synthetic steps to append and remove the auxiliary. However, in some cases the only available stereoselective methodology relies on chiral auxiliaries and these reactions tend to be versatile and very well-studied, allowing the most time-efficient access
1268:
The development of enantioselective synthesis was initially slow, largely due to the limited range of techniques available for their separation and analysis. Diastereomers possess different physical properties, allowing separation by conventional means, however at the time enantiomers could only be
1173:-glucose by plants to be due to the influence of optically active substances within chlorophyll. Fischer also successfully performed what would now be regarded as the first example of enantioselective synthesis, by enantioselectively elongating sugars via a process which would eventually become the
1312:
to separate chiral amino acids. Although
Dalgliesh was not the first to observe such separations, he correctly attributed the separation of enantiomers to differential retention by the chiral cellulose. This was expanded upon in 1960, when Klem and Reed first reported the use of chirally-modified
995:
This can make it very difficult to determine whether a process has produced a single enantiomer (and crucially which enantiomer it is) as well as making it hard to separate enantiomers from a reaction which has not been 100% enantioselective. Fortunately, enantiomers behave differently in the
157:
Put more simply: it is the synthesis of a compound by a method that favors the formation of a specific enantiomer or diastereomer. Enantiomers are stereoisomers that have opposite configurations at every chiral center. Diastereomers are stereoisomers that differ at one or more chiral centers.
1371:
Early research into the teratogenic mechanism, using mice, suggested that one enantiomer of thalidomide was teratogenic while the other possessed all the therapeutic activity. This theory was later shown to be incorrect and has now been superseded by a body of research. However it raised the
1019:
to convert the enantiomers into a diastereomers, in much the same way as chiral auxiliaries. These have different physical properties and hence can be separated and analysed using conventional methods. Special chiral derivitizing agents known as 'chiral resolution agents' are used in the
913:
Chiral pool synthesis is one of the simplest and oldest approaches for enantioselective synthesis. A readily available chiral starting material is manipulated through successive reactions, often using achiral reagents, to obtain the desired target molecule. This can meet the criteria for
783:
A chiral auxiliary is an organic compound which couples to the starting material to form a new compound which can then undergo diastereoselective reactions via intramolecular asymmetric induction. At the end of the reaction the auxiliary is removed, under conditions that will not cause
1137:
geometry of carbon. Structural models prior to this work had been two-dimensional, and van 't Hoff and Le Bel theorized that the arrangement of groups around this tetrahedron could dictate the optical activity of the resulting compound through what became known as the
1255:
argued that natural and artificial compounds were fundamentally different and that chirality was simply a manifestation of the 'vital force' which could only exist in natural compounds. Unlike
Fischer, Marckwald had performed an enantioselective reaction upon an achiral,
718:
ligands. Most enantioselective catalysts are effective at low substrate/catalyst ratios. Given their high efficiencies, they are often suitable for industrial scale synthesis, even with expensive catalysts. A versatile example of enantioselective synthesis is
470:
2569:
Notz, Wolfgang; Tanaka, Fujie; Barbas, Carlos F. (1 August 2004). "Enamine-Based
Organocatalysis with Proline and Diamines: The Development of Direct Catalytic Asymmetric Aldol, Mannich, Michael, and Diels−Alder Reactions".
1486:. In common with Knowles' findings, Noyori's results for the enantiomeric excess for this first-generation ligand were disappointingly low: 6%. However continued research eventually led to the development of the
2752:
Researches on the molecular asymmetry of natural organic products, English translation of French original, published by
Alembic Club Reprints (Vol. 14, pp. 1–46) in 1905, facsimile reproduction by SPIE in a 1990
475:
This temperature dependence means the rate difference, and therefore the enantioselectivity, is greater at lower temperatures. As a result, even small energy-barrier differences can lead to a noticeable effect.
1750:
Lepola U, Wade A, Andersen HF (May 2004). "Do equivalent doses of escitalopram and citalopram have similar efficacy? A pooled analysis of two positive placebo-controlled studies in major depressive disorder".
1722:
Theodore J. Leitereg; Dante G. Guadagni; Jean Harris; Thomas R. Mon; Roy
Teranishi (1971). "Chemical and sensory data supporting the difference between the odors of the enantiomeric carvones".
1015:, or on a large scale to separate chirally impure materials. However this process can require large amount of chiral packing material which can be expensive. A common alternative is to use a
710:. Catalysis is effective for a broader range of transformations than any other method of enantioselective synthesis. The chiral metal catalysts are almost invariably rendered chiral by using
1687:
Gal, Joseph (2012). "The
Discovery of Stereoselectivity at Biological Receptors: Arnaldo Piutti and the Taste of the Asparagine Enantiomers-History and Analysis on the 125th Anniversary".
3637:
Sharpless, K. Barry; Patrick, Donald W.; Truesdale, Larry K.; Biller, Scott A. (1975). "New reaction. Stereospecific vicinal oxyamination of olefins by alkyl imido osmium compounds".
353:
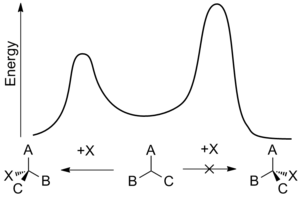
Enantioselectivity is usually determined by the relative rates of an enantiodifferentiating step—the point at which one reactant can become either of two enantiomeric products. The
1360:, eventually causing birth defects in more than 10,000 babies. The disaster prompted many countries to introduce tougher rules for the testing and licensing of drugs, such as the
932:
Chiral pool synthesis is especially attractive for target molecules having similar chirality to a relatively inexpensive naturally occurring building-block such as a sugar or
3554:
H. Nozaki; H. Takaya; S. Moriuti; R. Noyori (1968). "Homogeneous catalysis in the decomposition of diazo compounds by copper chelates: Asymmetric carbenoid reactions".
1559:
has allowed the tailoring of enzymes to specific processes, permitting an increased range of selective transformations. For example, in the asymmetric hydrogenation of
3610:
Jacobsen, Eric N.; Marko, Istvan.; Mungall, William S.; Schroeder, Georg.; Sharpless, K. Barry. (1988). "Asymmetric dihydroxylation via ligand-accelerated catalysis".
3430:
Gil-Av, Emanuel; Feibush, Binyamin; Charles-Sigler, Rosita (1966). "Separation of enantiomers by gas liquid chromatography with an optically active stationary phase".
1548:
1479:
387:
4184:
3731:
Ensley, Harry E.; Parnell, Carol A.; Corey, Elias J. (1978). "Convenient synthesis of a highly efficient and recyclable chiral director for asymmetric induction".
841:. The high reagent specificity can be a problem, however, as it often requires that a wide range of biocatalysts be screened before an effective reagent is found.
326:
mixture. Enantioselective synthesis can be achieved by using a chiral feature that favors the formation of one enantiomer over another through interactions at the
3696:
Hinckley, Conrad C. (1969). "Paramagnetic shifts in solutions of cholesterol and the dipyridine adduct of trisdipivalomethanatoeuropium(III). A shift reagent".
1384:) were first published in 1966; allowing enantiomers to be more easily and accurately described. The same year saw first successful enantiomeric separation by
1587:, the synthesis of chiral products from achiral precursors and without the use of optically active catalysts or auxiliaries. It is relevant to the discussion
4189:
3457:
Vineyard, B. D.; Knowles, W. S.; Sabacky, M. J.; Bachman, G. L.; Weinkauff, D. J. (1977). "Asymmetric hydrogenation. Rhodium chiral bisphosphine catalyst".
3083:
Bijvoet, J. M.; Peerdeman, A. F.; van Bommel, A. J. (1951). "Determination of the
Absolute Configuration of Optically Active Compounds by Means of X-Rays".
1050:. The most frequently employed technique to carry out chiral analysis involves separation science procedures, specifically chiral chromatographic methods.
883:, then enantioselective synthesis can be achieved; for example a number of carbon–carbon bond forming reactions become enantioselective in the presence of
1552:
1551:. Enzyme-catalyzed enantioselective reactions became more and more common during the 1980s, particularly in industry, with their applications including
1223:
reported in 1904. A slight excess of the levorotary form of the product of the reaction, 2-methylbutyric acid, was produced; as this product is also a
3668:(1969). "α-Methoxy-α-trifluoromethylphenylacetic acid, a versatile reagent for the determination of enantiomeric composition of alcohols and amines".
2286:
1930:
1657:
1628:
1066:
2520:
4296:
3054:"CXXVII.Studies in asymmetric synthesis. I. Reduction of menthyl benzoylformate. II. Action of magnesium alkyl haloids on menthyl benzoylformate"
1235:—this result constitutes the first recorded total synthesis with enantioselectivity, as well other firsts (as Koskinen notes, first "example of
4050:
4397:
3830:
4402:
1356:
First synthesized in 1953, thalidomide was widely prescribed for morning sickness from 1957 to 1962, but was soon found to be seriously
1247:"). This observation is also of historical significance, as at the time enantioselective synthesis could only be understood in terms of
1345:
While it was known that the different enantiomers of a drug could have different activities, with significant early work being done by
936:. However, the number of possible reactions the molecule can undergo is restricted and tortuous synthetic routes may be required (e.g.
1584:
3497:
2805:[On the relations which exist between the atomic formulas of organic compounds and the rotatory power of their solutions].
2547:
2341:
Schmid, A.; Dordick, J. S.; Hauer, B.; Kiener, A.; Wubbolts, M.; Witholt, B. (2001). "Industrial biocatalysis today and tomorrow".
4380:
3403:
3371:
2161:
314:
As such enantioselective synthesis is of great importance but it can also be difficult to achieve. Enantiomers possess identical
2803:"Sur les relations qui existent entre les formules atomiques des corps organiques et le pouvoir rotatoire de leurs dissolutions"
4424:
1012:
977:
3024:
2863:
2684:
2325:
2271:
2214:
2143:
1980:
1427:. Knowles was also the first to apply enantioselective metal catalysis to industrial-scale synthesis; while working for the
4436:
4375:
3918:
3014:
1506:
4289:
4004:
1794:
Hyttel, J.; Bøgesø, K. P.; Perregaard, J.; Sánchez, C. (1992). "The pharmacological effect of citalopram resides in the (
213:
and will often react differently with the various enantiomers of a given compound. Examples of this selectivity include:
17:
1042:
The separation and analysis of component enantiomers of a racemic drugs or pharmaceutical substances are referred to as
4043:
2262:
Evans, D. A.; Helmchen, G.; Rüping, M. (2007). "Chiral
Auxiliaries in Asymmetric Synthesis". In Christmann, M. (ed.).
3038:
Much of this early work was published in German, however contemporary
English accounts can be found in the papers of
2649:
2504:
2445:
1021:
1208:
4222:
3823:
1057:
of a substance can also be determined using certain optical methods. The oldest method for doing this is to use a
5241:
3873:
3785:
Wandrey, Christian; Liese, Andreas; Kihumbu, David (2000). "Industrial
Biocatalysis: Past, Present, and Future".
1572:
1487:
1126:
758:
5120:
4282:
3519:
Knowles, W. S. (March 1986). "Application of organometallic catalysis to the commercial production of L-DOPA".
2715:
Gal, Joseph (January 2011). "Louis Pasteur, language, and molecular chirality. I. Background and Dissymmetry".
1139:
837:. Biocatalysts are more commonly used in industry than in academic research; for example in the production of
161:
Enantioselective synthesis is a key process in modern chemistry and is particularly important in the field of
4563:
4317:
4207:
4036:
2824:
1174:
4217:
3039:
2766:
Pedro Cintas (2007). "Tracing the Origins and Evolution of Chirality and Handedness in Chemical Language".
1004:
1997:
Bauer, Eike B. (2012). "Chiral-at-metal complexes and their catalytic applications in organic synthesis".
1547:. Around the same time enantioselective organocatalysis was developed, with pioneering work including the
5263:
4840:
4327:
3992:
3973:
3816:
937:
829:
to living cells, to perform chemical transformations. The advantages of these reagents include very high
761:
with BINAP/Ru requires a β-ketone, although another catalyst, BINAP/diamine-Ru, widens the scope to α,β-
5217:
4766:
4717:
4670:
3583:
Katsuki, Tsutomu; Sharpless, K. Barry (1980). "The first practical method for asymmetric epoxidation".
3126:
Dalgliesh, C. E. (1952). "756. The optical resolution of aromatic amino-acids on paper chromatograms".
1372:
importance of chirality in drug design, leading to increased research into enantioselective synthesis.
190:
4355:
4179:
3908:
3853:
1609:
1361:
1105:
showed that certain chemicals could rotate the plane of a beam of polarised light, a property called
1047:
1016:
996:
presence of other chiral materials and this can be exploited to allow their separation and analysis.
973:
702:
Enantioselective catalysis (known traditionally as "asymmetric catalysis") is performed using chiral
37:
5268:
5110:
5026:
4665:
4105:
3883:
1408:
1404:
720:
803:
to enantiomerically pure products. Additionally, the products of auxiliary-directed reactions are
5048:
4959:
4922:
4806:
4732:
4553:
4536:
4479:
3948:
3362:
1501:
Sharpless complemented these reduction reactions by developing a range of asymmetric oxidations (
1416:
1346:
1305:
968:
etc.) and so behave identically to each other. As a result, they will migrate with an identical R
3149:
Klemm, L.H.; Reed, David (1960). "Optical resolution by molecular complexation chromatography".
1288:
It was not until the 1950s that major progress really began. Driven in part by chemists such as
1252:
185:
4966:
4954:
4845:
4710:
4484:
4350:
3963:
3913:
2034:"Steering Asymmetric Lewis Acid Catalysis Exclusively with Octahedral Metal-Centered Chirality"
1510:
1301:
1270:
1077:
888:
465:{\displaystyle {\frac {k_{1}}{k_{2}}}=10^{\frac {\Delta \Delta G^{*}}{T\times 1.98\times 2.3}}}
3636:
1260:
starting material, albeit with a chiral organocatalyst (as we now understand this chemistry).
377:*, means that the relative rates for opposing stereochemical outcomes at a given temperature,
5115:
5012:
4997:
4927:
4850:
4682:
4632:
4541:
4466:
4365:
4243:
4157:
4074:
3958:
3758:
Sariaslani, F.Sima; Rosazza, John P.N. (1984). "Biocatalysis in natural products chemistry".
3556:
3401:; Günter Helmchen (1982). "Basic Principles of the CIP-System and Proposals for a Revision".
2497:
Asymmetric organocatalysis – from biomimetic concepts to applications in asymmetric synthesis
1721:
1502:
1297:
1277:(where one enantiomer is selectively destroyed). The only tool for analysing enantiomers was
1081:
989:
908:
880:
808:
335:
315:
210:
135:
1520:
During the same period, methods were developed to allow the analysis of chiral compounds by
5105:
5060:
4835:
4655:
4585:
4342:
4322:
4253:
4238:
4089:
3878:
3858:
3528:
3092:
2974:"Biosynthesis of lovastatin and related metabolites formed by fungal iterative PKS enzymes"
2802:
2350:
1724:
1423:. This experimental catalyst was employed in an asymmetric hydrogenation with a modest 15%
1365:
1309:
1236:
1166:
1162:
1076:
One of the most accurate ways of determining the chirality of compound is to determine its
707:
331:
299:
221:
45:
3609:
8:
5128:
5082:
5007:
4980:
4878:
4860:
4813:
4751:
4647:
4627:
4496:
4491:
4392:
4151:
4028:
3893:
2235:
2233:
Glorius, F.; Gnas, Y. (2006). "Chiral Auxiliaries – Principles and Recent Applications".
1556:
1424:
1065:
in the product against a 'standard' of known composition. It is also possible to perform
1054:
830:
754:
730:
174:
151:
3532:
3357:
3327:
3302:
3096:
2354:
2080:
5273:
5205:
5171:
5033:
5002:
4883:
4825:
4523:
4506:
4501:
4456:
4419:
4409:
4370:
4212:
4110:
3839:
3358:
3340:
3251:
3226:
3202:
3177:
3108:
2605:
Bertelsen, Søren; Jørgensen, Karl Anker (2009). "Organocatalysis—after the gold rush".
2534:
Dalko, Peter I.; Moisan, Lionel (15 October 2001). "Enantioselective Organocatalysis".
2514:
2374:
2116:
1823:
1776:
1412:
1400:
1392:
1385:
1274:
1240:
1102:
1008:
981:
123:
3569:
3443:
3287:
3162:
1867:
1842:
1349:, this was not accounted for in early drug design and testing. However, following the
757:. Most catalysts are effective for only one type of asymmetric reaction. For example,
322:
and hence should be produced in equal amounts by an undirected process – leading to a
5224:
5186:
5151:
5134:
5072:
4990:
4985:
4913:
4898:
4868:
4789:
4756:
4727:
4722:
4697:
4687:
4607:
4595:
4474:
4387:
4199:
4011:
3938:
3933:
3928:
3863:
3771:
3713:
3501:
3332:
3256:
3207:
3020:
2995:
2859:
2783:
2732:
2680:
2645:
2622:
2587:
2551:
2500:
2441:
2418:
2366:
2321:
2267:
2210:
2178:
2139:
2108:
2100:
2061:
2053:
2014:
1976:
1913:
1872:
1815:
1768:
1764:
1704:
965:
956:
The two enantiomers of a molecule possess many of the same physical properties (e.g.
860:
766:
742:
370:
362:
347:
295:
131:
3344:
3042:, with continuing analysis and commentary in modern reviews such as Koskinen (2012).
2296:
2120:
1940:
1827:
1780:
1667:
1638:
5229:
5146:
4801:
4660:
4637:
4590:
4531:
4248:
4174:
4084:
4016:
3923:
3898:
3794:
3767:
3740:
3705:
3678:
3646:
3619:
3592:
3565:
3536:
3493:
3466:
3439:
3412:
3380:
3322:
3314:
3283:
3246:
3242:
3238:
3197:
3193:
3189:
3158:
3131:
3112:
3100:
3065:
2985:
2954:
2923:
2892:
2775:
2724:
2672:
2614:
2579:
2543:
2475:
2408:
2378:
2358:
2300:
2291:
2244:
2170:
2092:
2045:
2006:
1960:
1944:
1935:
1903:
1862:
1854:
1807:
1760:
1732:
1696:
1671:
1662:
1642:
1633:
1525:
1514:
1428:
1420:
1278:
1106:
1062:
864:
778:
724:
327:
71:
32:
3456:
1308:
in 1951. Chiral chromatography was introduced a year later by Dalgliesh, who used
1149:
Marckwald's brucine-catalyzed enantioselective decarboxylation of 2-ethyl-2-methyl
5087:
5043:
5038:
4932:
4908:
4742:
4705:
4558:
4548:
4431:
4258:
4146:
4126:
4100:
3968:
3665:
3398:
3366:
3016:
New Beer in an Old Bottle. Eduard Buchner and the Growth of Biochemical Knowledge
2159:
M. Heitbaum; F. Glorius; I. Escher (2006). "Asymmetric Heterogeneous Catalysis".
2096:
2049:
1604:
1293:
1244:
1224:
1216:
1204:
1188:
1125:
a year later. The origin of chirality itself was finally described in 1874, when
1043:
896:
892:
850:
834:
162:
92:
1493:
1396:
999:
Enantiomers do not migrate identically on chiral chromatographic media, such as
714:, but it is possible to generate chiral-at-metal complexes composed entirely of
4971:
4949:
4944:
4939:
4894:
4890:
4873:
4830:
4761:
4622:
4617:
4602:
4414:
4332:
3943:
3888:
2033:
1513:) during the 1970s and 1980s. With the asymmetric oxyamination reaction, using
1289:
1196:
1145:
1085:
941:
914:
enantioselective synthesis when a new chiral species is created, such as in an
799:
267:
3318:
2896:
737:
The design of new catalysts is dominated by the development of new classes of
5257:
5176:
5065:
5021:
4746:
4580:
4575:
4568:
4446:
4141:
3953:
3670:
3429:
2958:
2927:
2665:
Snyder, Lloyd R.; Kirkland, Joseph J.; Glajch, Joseph L. (28 February 1997).
2295:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
2195:
Asymmetric Catalysis on Industrial Scale, (Blaser, Schmidt), Wiley-VCH, 2004.
2104:
2057:
1968:
1964:
1939:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1666:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1637:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
1588:
1544:
1130:
1110:
1070:
961:
957:
804:
711:
354:
288:
83:
2304:
1948:
1908:
1891:
1793:
1675:
1646:
1455:
1446:
1084:. However this is a labour-intensive process which requires that a suitable
948:
starting material, which can be expensive if it is not naturally occurring.
350:
required to form one enantiomer lower than that of the opposing enantiomer.
5053:
4903:
4818:
4794:
4784:
4776:
4677:
4612:
4511:
4360:
4263:
4136:
4079:
3868:
3505:
3416:
3384:
3336:
3260:
3211:
2999:
2787:
2779:
2736:
2666:
2626:
2591:
2555:
2422:
2413:
2396:
2370:
2182:
2174:
2112:
2065:
2018:
1917:
1876:
1772:
1708:
1411:, which they developed independently in 1968. Knowles replaced the achiral
1232:
1220:
1150:
1003:
or standard media that has been chirally modified. This forms the basis of
915:
891:
being a prime example. Organocatalysis often employs natural compounds and
820:
791:
785:
750:
170:
147:
139:
3717:
3498:
10.1002/1521-3773(20020617)41:12<1998::AID-ANIE1998>3.0.CO;2-8
2676:
2548:
10.1002/1521-3773(20011015)40:20<3726::AID-ANIE3726>3.0.CO;2-D
2248:
1819:
1431:
he developed an enantioselective hydrogenation step for the production of
1388:
an important development as the technology was in common use at the time.
1203:
The first enantioselective chemical synthesis is most often attributed to
4451:
3484:
Knowles, William S. (2002). "Asymmetric Hydrogenations (Nobel Lecture)".
3135:
3069:
1475:
1357:
1350:
1320:
1282:
1134:
1122:
1058:
945:
925:
4274:
3744:
3709:
3682:
3650:
3623:
3596:
3470:
3178:"Atropine and the hyoscyamines-a study of the action of optical isomers"
3082:
1736:
5077:
4131:
3663:
2728:
2340:
2010:
1811:
1700:
1578:
1540:
1381:
1228:
1192:
1154:
933:
307:
271:
206:
202:
166:
143:
66:
3798:
3730:
3540:
2990:
2973:
2702:
Selected Papers on Natural Optical Activity (SPIE Milestone Volume 15)
2583:
2480:
2463:
1858:
1353:
disaster the development and licensing of drugs changed dramatically.
1296:
but also by the development of new techniques. The first of these was
825:
Biocatalysis makes use of biological compounds, ranging from isolated
5139:
4441:
4306:
3808:
3553:
3104:
2618:
2362:
2081:"Asymmetric Photocatalysis with Bis-cyclometalated Rhodium Complexes"
1594:
1113:
proposed that it had a molecular basis originating from some form of
856:
703:
249:
224:
3053:
2942:
2911:
2880:
2397:"Chemoenzymatic Synthesis of Building Blocks for Statin Side Chains"
1575:, for the use of a chiral ionic liquid in enantioselective synthesis
5161:
3784:
3274:
McBride, W. G. (1961). "Thalidomide and Congenital Abnormalities".
1529:
1248:
1185:
1025:
872:
343:
108:
2568:
1109:. The nature of this property remained a mystery until 1848, when
833:
and reagent specificity, as well as mild operating conditions and
282:)-(+) enantiomer is responsible for the drug's beneficial effects.
5181:
2642:
Chromatographic enantioseparation : methods and applications
1598:
1483:
1212:
884:
762:
715:
339:
323:
319:
275:
257:
245:
2644:. Chichester, West Sussex, England: E. Horwood. pp. 64–66.
1380:
The Cahn–Ingold–Prelog priority rules (often abbreviated as the
745:", are effective in a wide range of reactions; examples include
40:
reaction the chirality of the product can be controlled by the "
1560:
1436:
1432:
1000:
876:
868:
838:
826:
788:
of the product. It is typically then recovered for future use.
738:
79:
44:" used. This is an example of enantioselective synthesis using
41:
27:
Chemical reaction(s) which favor one chiral isomer over another
3397:
2158:
2134:
N. Jacobsen, Eric; Pfaltz, Andreas; Yamamoto, Hisashi (1999).
2858:(Second ed.). Hoboken, N.J.: Wiley. pp. 17, 28–29.
1029:
746:
198:
127:
3757:
1391:
Metal-catalysed enantioselective synthesis was pioneered by
1169:; in which he correctly ascribed selective the formation of
134:(or reaction sequence) in which one or more new elements of
5156:
1959:
1314:
1180:
1007:, which can be used on a small scale to allow analysis via
807:, which enables their facile separation by methods such as
4058:
3582:
3303:"Teratogenic effects of thalidomide: molecular mechanisms"
2909:
197:
Many of the building blocks of biological systems such as
138:
are formed in a substrate molecule and which produces the
5166:
2829:
Archives Néerlandaises des Sciences Exactes et Naturelles
2133:
1892:"Chiral Toxicology: It's the Same Thing...Only Different"
1840:
1521:
985:
951:
879:
and other non-metal elements. When the organocatalyst is
2435:
2320:(6th rev. and corr. ed.). Berlin: Springer-Verlag.
2972:
Campbell, Chantel D.; Vederas, John C. (23 June 2010).
209:. As a result, living systems possess a high degree of
3301:
Ito, Takumi; Ando, Hideki; Handa, Hiroshi (May 2011).
2454:
2910:
Fischer, Emil; Hirschberger, Josef (1 January 1889).
2499:(1. ed., 2. reprint. ed.). Weinheim: Wiley-VCH.
1581:, a chiral ligand widely used in asymmetric synthesis
1273:(where enantiomers separate upon crystallisation) or
390:
306:‑penicillamine is toxic as it inhibits the action of
2664:
278:
mixture. However, studies have shown that only the (
2533:
2261:
2232:
1843:"The Antipyridoxine Effect of Penicillamine in Man"
1553:
asymmetric ester hydrolysis with pig-liver esterase
1524:; either using chiral derivatizing agents, such as
1407:. Knowles and Noyori began with the development of
844:
2604:
2318:Biotransformations in organic chemistry a textbook
1841:JAFFE, IA; ALTMAN, K; MERRYMAN, P (October 1964).
1749:
1231:formed by its diketide synthase (LovF) during its
464:
4190:Ultraviolet–visible spectroscopy of stereoisomers
3224:
2228:
2226:
2079:Huang, Xiaoqiang; Meggers, Eric (19 March 2019).
1468:Noyori: Enantioselective cyclopropanation (1968)
1067:ultraviolet-visible spectroscopy of stereoisomers
1024:, these typically involve coordination to chiral
5255:
3369:(1966). "Specification of Molecular Chirality".
3148:
2266:. Wiley-VCH Verlag GmbH & Co. pp. 3–9.
1601:which originates from enantioselective synthesis
1543:in 1978 and featured prominently in the work of
2971:
2849:
2847:
2845:
2843:
2841:
2032:Zhang, Lilu; Meggers, Eric (21 February 2017).
1474:Noyori devised a copper complex using a chiral
895:as chiral catalysts; these are inexpensive and
3227:"The action of optical isomers: II. Hyoscines"
3012:
2947:Berichte der Deutschen Chemischen Gesellschaft
2916:Berichte der Deutschen Chemischen Gesellschaft
2885:Berichte der Deutschen Chemischen Gesellschaft
2223:
1285:, a method which provides no structural data.
1251:. At the time many prominent chemists such as
4290:
4044:
3824:
2940:
2825:"Sur les formules de structure dans l'espace"
2699:
697:
3300:
2838:
2765:
2519:: CS1 maint: multiple names: authors list (
2255:
2078:
2031:
723:, which is used to reduce a wide variety of
3019:, Universitat de València, pp. 72–73,
2749:
2495:Gröger, Albrecht Berkessel; Harald (2005).
2207:Compendium of chiral auxiliary applications
1743:
4297:
4283:
4051:
4037:
3831:
3817:
3787:Organic Process Research & Development
1549:Hajos–Parrish–Eder–Sauer–Wiechert reaction
1375:
369:, and is temperature-dependent. Using the
4304:
3326:
3250:
3201:
3128:Journal of the Chemical Society (Resumed)
3125:
2989:
2639:
2479:
2412:
2390:
2388:
1975:(1st ed.). Oxford University Press.
1907:
1866:
1585:Spontaneous absolute asymmetric synthesis
1463:Knowles: Asymmetric hydrogenation (1968)
1263:
1157:form of the 2-methylbutyric acid product.
741:. Certain ligands, often referred to as "
346:, or environment and works by making the
193:of an enantioselective addition reaction.
3698:Journal of the American Chemical Society
3695:
3639:Journal of the American Chemical Society
3612:Journal of the American Chemical Society
3585:Journal of the American Chemical Society
3459:Journal of the American Chemical Society
3225:Cushny, AR; Peebles, AR (13 July 1905).
3051:
2856:Asymmetric synthesis of natural products
2853:
1492:
1403:; for which they would receive the 2001
1319:
1179:
1144:
1096:
902:
184:
65:
31:
3518:
3486:Angewandte Chemie International Edition
3483:
3404:Angewandte Chemie International Edition
3372:Angewandte Chemie International Edition
3273:
2878:
2768:Angewandte Chemie International Edition
2536:Angewandte Chemie International Edition
2401:Angewandte Chemie International Edition
2162:Angewandte Chemie International Edition
334:and can involve chiral features in the
14:
5256:
3838:
3175:
3052:McKenzie, Alexander (1 January 1904).
2800:
2494:
2394:
2385:
2136:Comprehensive asymmetric catalysis 1-3
1539:Chiral auxiliaries were introduced by
1532:based shift reagents, of which Eu(DPM)
1153:, resulting in a slight excess of the
976:and have identical retention times in
952:Separation and analysis of enantiomers
365:of the reaction, sometimes called the
4278:
4032:
3812:
2315:
2264:Asymmetric Synthesis – The Essentials
1996:
1889:
1847:The Journal of Clinical Investigation
772:
5212:
3999:
3307:Cellular and Molecular Life Sciences
2827:(On structural formulas in space),
2461:
2204:
1507:Sharpless asymmetric dihydroxylation
855:Organocatalysis refers to a form of
361:, for a reaction is function of the
5236:
3013:Cornish-Bawden, Athel, ed. (1997),
2714:
2436:Berkessel, A.; Groeger, H. (2005).
1686:
1324:The two enantiomers of thalidomide:
798:Chiral auxiliaries must be used in
173:of a molecule often have different
24:
2881:"Synthesen in der Zuckergruppe II"
2395:Müller, Michael (7 January 2005).
2292:Compendium of Chemical Terminology
1936:Compendium of Chemical Terminology
1663:Compendium of Chemical Terminology
1634:Compendium of Chemical Terminology
1300:, which was used to determine the
924:
790:
729:
428:
425:
25:
5285:
4185:NMR spectroscopy of stereoisomers
2668:Practical HPLC Method Development
1127:Jacobus Henricus van 't Hoff
1022:NMR spectroscopy of stereoisomers
940:). This approach also requires a
5235:
5223:
5211:
5200:
5199:
4223:Diastereomeric recrystallization
4010:
3998:
3987:
3986:
3733:The Journal of Organic Chemistry
2879:Fischer, Emil (1 October 1894).
1765:10.1097/00004850-200405000-00005
1480:metal–carbenoid cyclopropanation
1454:
1445:
845:Enantioselective organocatalysis
231:-aspartame tastes sweet whereas
205:are produced exclusively as one
3778:
3760:Enzyme and Microbial Technology
3751:
3724:
3689:
3657:
3630:
3603:
3576:
3547:
3512:
3477:
3450:
3423:
3391:
3351:
3294:
3267:
3218:
3169:
3142:
3119:
3076:
3045:
3032:
3006:
2965:
2934:
2903:
2872:
2817:
2794:
2759:
2743:
2708:
2693:
2658:
2633:
2598:
2562:
2527:
2488:
2429:
2334:
2309:
2280:
2198:
2189:
2152:
2127:
2072:
2025:
1990:
1953:
1488:Noyori asymmetric hydrogenation
1441:
814:
759:Noyori asymmetric hydrogenation
3243:10.1113/jphysiol.1905.sp001097
3194:10.1113/jphysiol.1903.sp000988
3176:Cushny, AR (2 November 1903).
2943:"Ueber asymmetrische Synthese"
2209:. San Diego, CA: Acad. Press.
1924:
1883:
1834:
1800:Journal of Neural Transmission
1787:
1715:
1680:
1651:
1622:
1478:ligand, which he used for the
1340:
13:
1:
4564:Interface and colloid science
4318:Glossary of chemical formulae
3570:10.1016/S0040-4020(01)91998-2
3521:Journal of Chemical Education
3444:10.1016/S0040-4039(00)70231-0
3288:10.1016/S0140-6736(61)90927-8
3163:10.1016/S0021-9673(01)97011-6
2572:Accounts of Chemical Research
2085:Accounts of Chemical Research
2038:Accounts of Chemical Research
1890:Smith, Silas W. (July 2009).
1615:
1555:. The emerging technology of
1101:In 1815 the French physicist
899:, as no metals are involved.
706:, which are typically chiral
692:
70:Two enantiomers of a generic
58:= Medium-sized substituent; R
4218:Chiral column chromatography
3772:10.1016/0141-0229(84)90125-X
2097:10.1021/acs.accounts.9b00028
2050:10.1021/acs.accounts.6b00586
1467:
1462:
1215:-catalyzed enantioselective
1005:chiral column chromatography
7:
4841:Bioorganometallic chemistry
4328:List of inorganic compounds
3974:Volume combustion synthesis
3664:J. A. Dale, D. L. Dull and
3151:Journal of Chromatography A
2854:Koskinen, Ari M.P. (2013).
2700:Lakhtakia, A., ed. (1990).
2640:Allenmark, Stig G. (1988).
1573:Aza-Baylis–Hillman reaction
1566:
1465:
1451:
1133:independently proposed the
938:Oseltamivir total synthesis
330:. This biasing is known as
180:
10:
5290:
4767:Dynamic covalent chemistry
4738:Enantioselective synthesis
4718:Physical organic chemistry
4671:Organolanthanide chemistry
4180:Chiral derivatizing agents
4061:enantioselective synthesis
3904:Enantioselective synthesis
2438:Asymmetric Organocatalysis
1497:The Sharpless oxyamination
1304:of an organic compound by
1227:—e.g., as a side chain of
1195:, used successfully as an
1091:
906:
848:
818:
776:
698:Enantioselective catalysis
116:Enantioselective synthesis
5195:
5098:
4859:
4775:
4696:
4646:
4522:
4465:
4356:Electroanalytical methods
4341:
4313:
4231:
4198:
4167:
4119:
4067:
3982:
3909:Fully automated synthesis
3854:Artificial gene synthesis
3846:
3319:10.1007/s00018-010-0619-9
3231:The Journal of Physiology
3182:The Journal of Physiology
2897:10.1002/cber.189402703109
2823:van 't Hoff, J.H. (1874)
1668:stereoselective synthesis
1610:Enantioselective analysis
1362:Kefauver-Harris Amendment
1175:Kiliani–Fischer synthesis
1048:enantioselective analysis
1017:chiral derivatizing agent
974:thin layer chromatography
546:
518:
490:
310:, an essential B vitamin.
298:and for the treatment of
256:-(+)-carvone smells like
38:Sharpless dihydroxylation
5111:Nobel Prize in Chemistry
5027:Supramolecular chemistry
4666:Organometallic chemistry
4106:Supramolecular chirality
3884:Custom peptide synthesis
2959:10.1002/cber.19040370165
2928:10.1002/cber.18890220183
2607:Chemical Society Reviews
1999:Chemical Society Reviews
1753:Int Clin Psychopharmacol
1409:asymmetric hydrogenation
1405:Nobel Prize in Chemistry
1165:outlined the concept of
1061:to compare the level of
897:environmentally friendly
835:low environmental impact
721:asymmetric hydrogenation
373:of the energy barrier, Δ
235:-aspartame is tasteless.
54:= Largest substituent; R
5049:Combinatorial chemistry
4960:Food physical chemistry
4923:Environmental chemistry
4807:Bioorthogonal chemistry
4733:Retrosynthetic analysis
4554:Chemical thermodynamics
4537:Spectroelectrochemistry
4480:Computational chemistry
3363:Christopher Kelk Ingold
2801:Le Bel, Joseph (1874).
2462:List, Benjamin (2007).
2440:. Weinheim: Wiley-VCH.
2305:10.1351/goldbook.B00652
1949:10.1351/goldbook.A00483
1676:10.1351/goldbook.S05990
1647:10.1351/goldbook.A00484
1376:Modern age (since 1965)
1347:Arthur Robertson Cushny
1140:Le Bel–van 't Hoff rule
992:spectra are identical.
5121:of element discoveries
4967:Agricultural chemistry
4955:Carbohydrate chemistry
4846:Bioinorganic chemistry
4711:Alkane stereochemistry
4656:Coordination chemistry
4485:Mathematical chemistry
4351:Instrumental chemistry
3964:Solvothermal synthesis
3914:Hydrothermal synthesis
3417:10.1002/anie.198205671
3385:10.1002/anie.196603851
2941:Marckwald, W. (1904).
2780:10.1002/anie.200603714
2414:10.1002/anie.200460852
2205:Roos, Gregory (2002).
2175:10.1002/anie.200504212
1896:Toxicological Sciences
1517:, being the earliest.
1511:Sharpless oxyamination
1498:
1337:
1313:silica gel for chiral
1302:absolute configuration
1271:spontaneous resolution
1264:Early work (1905–1965)
1241:enantiotopic selection
1200:
1158:
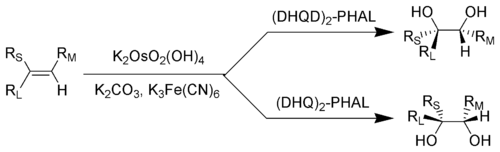
1078:absolute configuration
929:
859:, where the rate of a
795:
734:
708:coordination complexes
466:
194:
112:
63:
62:= Smallest substituent
5116:Timeline of chemistry
5013:Post-mortem chemistry
4998:Clandestine chemistry
4928:Atmospheric chemistry
4851:Biophysical chemistry
4683:Solid-state chemistry
4633:Equilibrium chemistry
4542:Photoelectrochemistry
4244:Chiral pool synthesis
4158:Diastereomeric excess
3959:Solid-phase synthesis
2677:10.1002/9781118592014
2249:10.1055/s-2006-942399
1909:10.1093/toxsci/kfp097
1503:Sharpless epoxidation
1496:
1323:
1298:X-ray crystallography
1209:Universität zu Berlin
1199:by Marckwald in 1904.
1183:
1148:
1097:Inception (1815–1905)
1082:X-ray crystallography
928:
909:Chiral pool synthesis
903:Chiral pool synthesis
809:column chromatography
794:
733:
467:
227:has two enantiomers.
188:
69:
35:
5106:History of chemistry
5061:Chemical engineering
4836:Bioorganic chemistry
4586:Structural chemistry
4323:List of biomolecules
4254:Asymmetric catalysis
4239:Asymmetric induction
3879:Convergent synthesis
3859:Biomimetic synthesis
3136:10.1039/JR9520003940
3070:10.1039/CT9048501249
2750:Pasteur, L. (1848).
2316:Faber, Kurt (2011).
2138:. Berlin: Springer.
1941:asymmetric induction
1725:J. Agric. Food Chem.
1639:asymmetric synthesis
1417:Wilkinson's catalyst
1366:Directive 65/65/EEC1
1310:paper chromatography
1253:Jöns Jacob Berzelius
1237:asymmetric catalysis
1167:asymmetric induction
1163:Hermann Emil Fischer
811:or crystallization.
388:
332:asymmetric induction
300:rheumatoid arthritis
222:artificial sweetener
120:asymmetric synthesis
46:asymmetric induction
5129:The central science
5083:Ceramic engineering
5008:Forensic toxicology
4981:Chemistry education
4879:Radiation chemistry
4861:Interdisciplinarity
4814:Medicinal chemistry
4752:Fullerene chemistry
4628:Microwave chemistry
4497:Molecular mechanics
4492:Molecular modelling
4152:Enantiomeric excess
3894:Divergent synthesis
3745:10.1021/jo00402a037
3710:10.1021/ja01046a038
3683:10.1021/jo01261a013
3651:10.1021/ja00841a071
3624:10.1021/ja00214a053
3597:10.1021/ja00538a077
3533:1986JChEd..63..222K
3471:10.1021/ja00460a018
3432:Tetrahedron Letters
3097:1951Natur.168..271B
3058:J. Chem. Soc. Trans
2912:"Ueber Mannose. II"
2807:Bull. Soc. Chim. Fr
2355:2001Natur.409..258S
1798:)-(+)-enantiomer".
1737:10.1021/jf60176a035
1557:genetic engineering
1425:enantiomeric excess
1219:of 2-ethyl-2-methyl
1055:enantiomeric excess
863:is increased by an
264:Drug effectiveness:
175:biological activity
165:, as the different
126:. It is defined by
18:Asymmetric reaction
5264:Chemical synthesis
5172:Chemical substance
5034:Chemical synthesis
5003:Forensic chemistry
4884:Actinide chemistry
4826:Clinical chemistry
4507:Molecular geometry
4502:Molecular dynamics
4457:Elemental analysis
4410:Separation process
4249:Chiral auxiliaries
4213:Kinetic resolution
4111:Inherent chirality
4096:-symmetric ligands
3840:Chemical synthesis
3359:Robert Sidney Cahn
3040:Alexander McKenzie
2729:10.1002/chir.20866
2011:10.1039/C2CS15234G
1812:10.1007/BF01244820
1701:10.1002/chir.22071
1536:was the earliest.
1499:
1413:triphenylphosphine
1401:K. Barry Sharpless
1393:William S. Knowles
1386:gas chromatography
1338:
1275:kinetic resolution
1201:
1159:
1103:Jean-Baptiste Biot
1069:by exploiting the
1028:complexes such as
930:
796:
773:Chiral auxiliaries
767:aromatic chemicals
743:privileged ligands
735:
462:
211:chemical chirality
195:
124:chemical synthesis
113:
64:
5251:
5250:
5187:Quantum mechanics
5152:Chemical compound
5135:Chemical reaction
5073:Materials science
4991:General chemistry
4986:Amateur chemistry
4914:Photogeochemistry
4899:Stellar chemistry
4869:Nuclear chemistry
4790:Molecular biology
4757:Polymer chemistry
4728:Organic synthesis
4723:Organic reactions
4688:Ceramic chemistry
4678:Cluster chemistry
4608:Chemical kinetics
4596:Molecular physics
4475:Quantum chemistry
4388:Mass spectrometry
4272:
4271:
4208:Recrystallization
4200:Chiral resolution
4026:
4025:
3939:Peptide synthesis
3934:Organic synthesis
3929:One-pot synthesis
3864:Bioretrosynthesis
3799:10.1021/op990101l
3704:(18): 5160–5162.
3591:(18): 5974–5976.
3541:10.1021/ed063p222
3492:(12): 1999–2007.
3465:(18): 5946–5952.
3438:(10): 1009–1015.
3091:(4268): 271–272.
3026:978-84-370-3328-0
2991:10.1002/bip.21428
2865:978-1-118-34733-1
2835: : 445–454.
2774:(22): 4016–4024.
2686:978-1-118-59201-4
2584:10.1021/ar0300468
2542:(20): 3726–3748.
2481:10.1021/cr078412e
2474:(12): 5413–5883.
2464:"Organocatalysis"
2349:(6817): 258–268.
2327:978-3-642-17393-6
2273:978-3-527-31399-0
2243:(12): 1899–1930.
2216:978-0-12-595344-3
2169:(29): 4732–4762.
2145:978-3-540-64337-1
1982:978-0-19-850346-0
1973:Organic Chemistry
1963:; Greeves, Nick;
1961:Clayden, Jonathan
1859:10.1172/JCI105060
1472:
1471:
1421:phosphine ligands
1172:
861:chemical reaction
725:functional groups
688:
687:
459:
413:
371:Gibbs free energy
363:activation energy
348:activation energy
305:
296:chelation therapy
291:
234:
230:
132:chemical reaction
16:(Redirected from
5281:
5239:
5238:
5227:
5215:
5214:
5203:
5202:
5147:Chemical element
4802:Chemical biology
4661:Magnetochemistry
4638:Mechanochemistry
4591:Chemical physics
4532:Electrochemistry
4437:Characterization
4299:
4292:
4285:
4276:
4275:
4175:Optical rotation
4120:Chiral molecules
4085:Planar chirality
4053:
4046:
4039:
4030:
4029:
4014:
4002:
4001:
3990:
3989:
3924:Mechanosynthesis
3899:Electrosynthesis
3833:
3826:
3819:
3810:
3809:
3803:
3802:
3782:
3776:
3775:
3755:
3749:
3748:
3739:(8): 1610–1612.
3728:
3722:
3721:
3693:
3687:
3686:
3677:(9): 2543–2549.
3661:
3655:
3654:
3645:(8): 2305–2307.
3634:
3628:
3627:
3618:(6): 1968–1970.
3607:
3601:
3600:
3580:
3574:
3573:
3564:(9): 3655–3669.
3551:
3545:
3544:
3516:
3510:
3509:
3481:
3475:
3474:
3454:
3448:
3447:
3427:
3421:
3420:
3395:
3389:
3388:
3355:
3349:
3348:
3330:
3313:(9): 1569–1579.
3298:
3292:
3291:
3271:
3265:
3264:
3254:
3222:
3216:
3215:
3205:
3173:
3167:
3166:
3146:
3140:
3139:
3123:
3117:
3116:
3105:10.1038/168271a0
3080:
3074:
3073:
3049:
3043:
3036:
3030:
3029:
3010:
3004:
3003:
2993:
2969:
2963:
2962:
2938:
2932:
2931:
2907:
2901:
2900:
2891:(3): 3189–3232.
2876:
2870:
2869:
2851:
2836:
2821:
2815:
2814:
2798:
2792:
2791:
2763:
2757:
2756:
2747:
2741:
2740:
2712:
2706:
2705:
2697:
2691:
2690:
2662:
2656:
2655:
2637:
2631:
2630:
2619:10.1039/b903816g
2602:
2596:
2595:
2566:
2560:
2559:
2531:
2525:
2524:
2518:
2510:
2492:
2486:
2485:
2483:
2458:
2452:
2451:
2433:
2427:
2426:
2416:
2392:
2383:
2382:
2363:10.1038/35051736
2338:
2332:
2331:
2313:
2307:
2284:
2278:
2277:
2259:
2253:
2252:
2230:
2221:
2220:
2202:
2196:
2193:
2187:
2186:
2156:
2150:
2149:
2131:
2125:
2124:
2076:
2070:
2069:
2029:
2023:
2022:
1994:
1988:
1986:
1957:
1951:
1928:
1922:
1921:
1911:
1887:
1881:
1880:
1870:
1838:
1832:
1831:
1791:
1785:
1784:
1747:
1741:
1740:
1719:
1713:
1712:
1684:
1678:
1655:
1649:
1626:
1597:, a property of
1515:osmium tetroxide
1458:
1449:
1442:
1435:, utilising the
1429:Monsanto Company
1306:Johannes Bijvoet
1279:optical activity
1170:
1121:being coined by
1117:, with the term
1107:optical activity
1063:optical rotation
893:secondary amines
865:organic compound
779:Chiral auxiliary
571:
569:
568:
560:
557:
543:
541:
540:
532:
529:
515:
513:
512:
504:
501:
481:
480:
471:
469:
468:
463:
461:
460:
458:
441:
440:
439:
423:
414:
412:
411:
402:
401:
392:
328:transition state
303:
289:
232:
228:
106:
100:
90:
77:
72:alpha amino acid
21:
5289:
5288:
5284:
5283:
5282:
5280:
5279:
5278:
5269:Stereochemistry
5254:
5253:
5252:
5247:
5191:
5094:
5088:Polymer science
5044:Click chemistry
5039:Green chemistry
4933:Ocean chemistry
4909:Biogeochemistry
4855:
4771:
4743:Total synthesis
4706:Stereochemistry
4692:
4642:
4559:Surface science
4549:Thermochemistry
4518:
4461:
4432:Crystallography
4337:
4309:
4303:
4273:
4268:
4259:Organocatalysis
4227:
4194:
4163:
4147:Racemic mixture
4115:
4101:Axial chirality
4095:
4068:Chirality types
4063:
4057:
4027:
4022:
3978:
3969:Total synthesis
3842:
3837:
3807:
3806:
3783:
3779:
3756:
3752:
3729:
3725:
3694:
3690:
3662:
3658:
3635:
3631:
3608:
3604:
3581:
3577:
3552:
3548:
3517:
3513:
3482:
3478:
3455:
3451:
3428:
3424:
3399:Vladimir Prelog
3396:
3392:
3367:Vladimir Prelog
3356:
3352:
3299:
3295:
3272:
3268:
3237:(5–6): 501–10.
3223:
3219:
3174:
3170:
3147:
3143:
3124:
3120:
3081:
3077:
3050:
3046:
3037:
3033:
3027:
3011:
3007:
2970:
2966:
2939:
2935:
2908:
2904:
2877:
2873:
2866:
2852:
2839:
2822:
2818:
2799:
2795:
2764:
2760:
2748:
2744:
2713:
2709:
2698:
2694:
2687:
2663:
2659:
2652:
2638:
2634:
2603:
2599:
2567:
2563:
2532:
2528:
2512:
2511:
2507:
2493:
2489:
2460:Special Issue:
2459:
2455:
2448:
2434:
2430:
2393:
2386:
2339:
2335:
2328:
2314:
2310:
2285:
2281:
2274:
2260:
2256:
2231:
2224:
2217:
2203:
2199:
2194:
2190:
2157:
2153:
2146:
2132:
2128:
2077:
2073:
2030:
2026:
1995:
1991:
1983:
1958:
1954:
1929:
1925:
1888:
1884:
1853:(10): 1869–73.
1839:
1835:
1792:
1788:
1748:
1744:
1720:
1716:
1695:(12): 959–976.
1685:
1681:
1656:
1652:
1627:
1623:
1618:
1605:Chiral analysis
1569:
1535:
1378:
1343:
1331:
1325:
1294:Vladimir Prelog
1266:
1245:organocatalysis
1225:natural product
1217:decarboxylation
1205:Willy Marckwald
1189:natural product
1099:
1094:
1044:chiral analysis
1038:
1033:
971:
954:
919:
911:
905:
853:
851:Organocatalysis
847:
823:
817:
781:
775:
700:
695:
621:
614:
607:
595:
588:
581:
572:at 323 K)
567:
561:
558:
556:
550:
549:
547:
539:
533:
530:
528:
522:
521:
519:
511:
505:
502:
500:
494:
493:
491:
442:
435:
431:
424:
422:
418:
407:
403:
397:
393:
391:
389:
386:
385:
183:
163:pharmaceuticals
152:unequal amounts
122:, is a form of
111:
104:
102:
98:
96:
93:Carboxylic acid
88:
86:
75:
61:
57:
53:
49:
48:
28:
23:
22:
15:
12:
11:
5:
5287:
5277:
5276:
5271:
5266:
5249:
5248:
5246:
5245:
5233:
5221:
5209:
5196:
5193:
5192:
5190:
5189:
5184:
5179:
5174:
5169:
5164:
5159:
5154:
5149:
5144:
5143:
5142:
5132:
5125:
5124:
5123:
5113:
5108:
5102:
5100:
5096:
5095:
5093:
5092:
5091:
5090:
5085:
5080:
5070:
5069:
5068:
5058:
5057:
5056:
5051:
5046:
5041:
5031:
5030:
5029:
5018:
5017:
5016:
5015:
5010:
5000:
4995:
4994:
4993:
4988:
4977:
4976:
4975:
4974:
4972:Soil chemistry
4964:
4963:
4962:
4957:
4950:Food chemistry
4947:
4945:Carbochemistry
4942:
4940:Clay chemistry
4937:
4936:
4935:
4930:
4919:
4918:
4917:
4916:
4911:
4901:
4895:Astrochemistry
4891:Cosmochemistry
4888:
4887:
4886:
4881:
4876:
4874:Radiochemistry
4865:
4863:
4857:
4856:
4854:
4853:
4848:
4843:
4838:
4833:
4831:Neurochemistry
4828:
4823:
4822:
4821:
4811:
4810:
4809:
4799:
4798:
4797:
4792:
4781:
4779:
4773:
4772:
4770:
4769:
4764:
4762:Petrochemistry
4759:
4754:
4749:
4740:
4735:
4730:
4725:
4720:
4715:
4714:
4713:
4702:
4700:
4694:
4693:
4691:
4690:
4685:
4680:
4675:
4674:
4673:
4663:
4658:
4652:
4650:
4644:
4643:
4641:
4640:
4635:
4630:
4625:
4623:Spin chemistry
4620:
4618:Photochemistry
4615:
4610:
4605:
4603:Femtochemistry
4600:
4599:
4598:
4588:
4583:
4578:
4573:
4572:
4571:
4561:
4556:
4551:
4546:
4545:
4544:
4539:
4528:
4526:
4520:
4519:
4517:
4516:
4515:
4514:
4504:
4499:
4494:
4489:
4488:
4487:
4477:
4471:
4469:
4463:
4462:
4460:
4459:
4454:
4449:
4444:
4439:
4434:
4429:
4428:
4427:
4422:
4415:Chromatography
4412:
4407:
4406:
4405:
4400:
4395:
4385:
4384:
4383:
4378:
4373:
4368:
4358:
4353:
4347:
4345:
4339:
4338:
4336:
4335:
4333:Periodic table
4330:
4325:
4320:
4314:
4311:
4310:
4302:
4301:
4294:
4287:
4279:
4270:
4269:
4267:
4266:
4261:
4256:
4251:
4246:
4241:
4235:
4233:
4229:
4228:
4226:
4225:
4220:
4215:
4210:
4204:
4202:
4196:
4195:
4193:
4192:
4187:
4182:
4177:
4171:
4169:
4165:
4164:
4162:
4161:
4155:
4149:
4144:
4139:
4134:
4129:
4123:
4121:
4117:
4116:
4114:
4113:
4108:
4103:
4098:
4093:
4087:
4082:
4077:
4071:
4069:
4065:
4064:
4056:
4055:
4048:
4041:
4033:
4024:
4023:
4021:
4020:
4008:
3996:
3983:
3980:
3979:
3977:
3976:
3971:
3966:
3961:
3956:
3951:
3949:Retrosynthesis
3946:
3944:Radiosynthesis
3941:
3936:
3931:
3926:
3921:
3916:
3911:
3906:
3901:
3896:
3891:
3889:Direct process
3886:
3881:
3876:
3874:Chemosynthesis
3871:
3866:
3861:
3856:
3850:
3848:
3844:
3843:
3836:
3835:
3828:
3821:
3813:
3805:
3804:
3793:(4): 286–290.
3777:
3766:(6): 242–253.
3750:
3723:
3688:
3656:
3629:
3602:
3575:
3546:
3511:
3476:
3449:
3422:
3411:(8): 567–583.
3390:
3379:(4): 385–415.
3350:
3293:
3282:(7216): 1358.
3266:
3217:
3168:
3141:
3118:
3075:
3044:
3031:
3025:
3005:
2984:(9): 755–763.
2964:
2933:
2922:(1): 365–376.
2902:
2871:
2864:
2837:
2816:
2793:
2758:
2742:
2707:
2692:
2685:
2657:
2650:
2632:
2613:(8): 2178–89.
2597:
2578:(8): 580–591.
2561:
2526:
2505:
2487:
2453:
2446:
2428:
2407:(3): 362–365.
2384:
2333:
2326:
2308:
2279:
2272:
2254:
2222:
2215:
2197:
2188:
2151:
2144:
2126:
2091:(3): 833–847.
2071:
2044:(2): 320–330.
2024:
2005:(8): 3153–67.
1989:
1981:
1969:Wothers, Peter
1965:Warren, Stuart
1952:
1923:
1882:
1833:
1806:(2): 157–160.
1786:
1742:
1731:(4): 785–787.
1714:
1679:
1650:
1620:
1619:
1617:
1614:
1613:
1612:
1607:
1602:
1592:
1582:
1576:
1568:
1565:
1533:
1470:
1469:
1466:
1464:
1460:
1459:
1452:
1450:
1377:
1374:
1342:
1339:
1290:R. B. Woodward
1265:
1262:
1197:organocatalyst
1098:
1095:
1093:
1090:
1086:single crystal
1036:
1031:
969:
953:
950:
944:amount of the
942:stoichiometric
917:
907:Main article:
904:
901:
889:aldol reaction
867:consisting of
849:Main article:
846:
843:
819:Main article:
816:
813:
800:stoichiometric
777:Main article:
774:
771:
712:chiral ligands
699:
696:
694:
691:
690:
689:
686:
685:
683:
680:
678:
675:
673:
670:
666:
665:
663:
660:
658:
655:
653:
650:
646:
645:
643:
640:
638:
635:
633:
630:
626:
625:
622:
618:
615:
611:
608:
604:
600:
599:
596:
592:
589:
585:
582:
578:
574:
573:
565:
554:
545:
544:at 298 K
537:
526:
517:
516:at 273 K
509:
498:
489:
473:
472:
457:
454:
451:
448:
445:
438:
434:
430:
427:
421:
417:
410:
406:
400:
396:
367:energy barrier
312:
311:
292:‑penicillamine
283:
268:antidepressant
261:
236:
191:energy profile
182:
179:
150:) products in
148:diastereomeric
140:stereoisomeric
118:, also called
103:
97:
87:
74:
59:
55:
51:
26:
9:
6:
4:
3:
2:
5286:
5275:
5272:
5270:
5267:
5265:
5262:
5261:
5259:
5244:
5243:
5234:
5232:
5231:
5226:
5222:
5220:
5219:
5210:
5208:
5207:
5198:
5197:
5194:
5188:
5185:
5183:
5180:
5178:
5177:Chemical bond
5175:
5173:
5170:
5168:
5165:
5163:
5160:
5158:
5155:
5153:
5150:
5148:
5145:
5141:
5138:
5137:
5136:
5133:
5130:
5126:
5122:
5119:
5118:
5117:
5114:
5112:
5109:
5107:
5104:
5103:
5101:
5097:
5089:
5086:
5084:
5081:
5079:
5076:
5075:
5074:
5071:
5067:
5066:Stoichiometry
5064:
5063:
5062:
5059:
5055:
5052:
5050:
5047:
5045:
5042:
5040:
5037:
5036:
5035:
5032:
5028:
5025:
5024:
5023:
5022:Nanochemistry
5020:
5019:
5014:
5011:
5009:
5006:
5005:
5004:
5001:
4999:
4996:
4992:
4989:
4987:
4984:
4983:
4982:
4979:
4978:
4973:
4970:
4969:
4968:
4965:
4961:
4958:
4956:
4953:
4952:
4951:
4948:
4946:
4943:
4941:
4938:
4934:
4931:
4929:
4926:
4925:
4924:
4921:
4920:
4915:
4912:
4910:
4907:
4906:
4905:
4902:
4900:
4896:
4892:
4889:
4885:
4882:
4880:
4877:
4875:
4872:
4871:
4870:
4867:
4866:
4864:
4862:
4858:
4852:
4849:
4847:
4844:
4842:
4839:
4837:
4834:
4832:
4829:
4827:
4824:
4820:
4817:
4816:
4815:
4812:
4808:
4805:
4804:
4803:
4800:
4796:
4793:
4791:
4788:
4787:
4786:
4783:
4782:
4780:
4778:
4774:
4768:
4765:
4763:
4760:
4758:
4755:
4753:
4750:
4748:
4747:Semisynthesis
4744:
4741:
4739:
4736:
4734:
4731:
4729:
4726:
4724:
4721:
4719:
4716:
4712:
4709:
4708:
4707:
4704:
4703:
4701:
4699:
4695:
4689:
4686:
4684:
4681:
4679:
4676:
4672:
4669:
4668:
4667:
4664:
4662:
4659:
4657:
4654:
4653:
4651:
4649:
4645:
4639:
4636:
4634:
4631:
4629:
4626:
4624:
4621:
4619:
4616:
4614:
4611:
4609:
4606:
4604:
4601:
4597:
4594:
4593:
4592:
4589:
4587:
4584:
4582:
4581:Sonochemistry
4579:
4577:
4576:Cryochemistry
4574:
4570:
4569:Micromeritics
4567:
4566:
4565:
4562:
4560:
4557:
4555:
4552:
4550:
4547:
4543:
4540:
4538:
4535:
4534:
4533:
4530:
4529:
4527:
4525:
4521:
4513:
4510:
4509:
4508:
4505:
4503:
4500:
4498:
4495:
4493:
4490:
4486:
4483:
4482:
4481:
4478:
4476:
4473:
4472:
4470:
4468:
4464:
4458:
4455:
4453:
4450:
4448:
4447:Wet chemistry
4445:
4443:
4440:
4438:
4435:
4433:
4430:
4426:
4423:
4421:
4418:
4417:
4416:
4413:
4411:
4408:
4404:
4401:
4399:
4396:
4394:
4391:
4390:
4389:
4386:
4382:
4379:
4377:
4374:
4372:
4369:
4367:
4364:
4363:
4362:
4359:
4357:
4354:
4352:
4349:
4348:
4346:
4344:
4340:
4334:
4331:
4329:
4326:
4324:
4321:
4319:
4316:
4315:
4312:
4308:
4300:
4295:
4293:
4288:
4286:
4281:
4280:
4277:
4265:
4262:
4260:
4257:
4255:
4252:
4250:
4247:
4245:
4242:
4240:
4237:
4236:
4234:
4230:
4224:
4221:
4219:
4216:
4214:
4211:
4209:
4206:
4205:
4203:
4201:
4197:
4191:
4188:
4186:
4183:
4181:
4178:
4176:
4173:
4172:
4170:
4166:
4159:
4156:
4153:
4150:
4148:
4145:
4143:
4142:Meso compound
4140:
4138:
4135:
4133:
4130:
4128:
4125:
4124:
4122:
4118:
4112:
4109:
4107:
4104:
4102:
4099:
4097:
4092:
4088:
4086:
4083:
4081:
4078:
4076:
4073:
4072:
4070:
4066:
4062:
4054:
4049:
4047:
4042:
4040:
4035:
4034:
4031:
4019:
4018:
4013:
4009:
4007:
4006:
3997:
3995:
3994:
3985:
3984:
3981:
3975:
3972:
3970:
3967:
3965:
3962:
3960:
3957:
3955:
3954:Semisynthesis
3952:
3950:
3947:
3945:
3942:
3940:
3937:
3935:
3932:
3930:
3927:
3925:
3922:
3920:
3917:
3915:
3912:
3910:
3907:
3905:
3902:
3900:
3897:
3895:
3892:
3890:
3887:
3885:
3882:
3880:
3877:
3875:
3872:
3870:
3867:
3865:
3862:
3860:
3857:
3855:
3852:
3851:
3849:
3845:
3841:
3834:
3829:
3827:
3822:
3820:
3815:
3814:
3811:
3800:
3796:
3792:
3788:
3781:
3773:
3769:
3765:
3761:
3754:
3746:
3742:
3738:
3734:
3727:
3719:
3715:
3711:
3707:
3703:
3699:
3692:
3684:
3680:
3676:
3673:
3672:
3671:J. Org. Chem.
3667:
3660:
3652:
3648:
3644:
3640:
3633:
3625:
3621:
3617:
3613:
3606:
3598:
3594:
3590:
3586:
3579:
3571:
3567:
3563:
3559:
3558:
3550:
3542:
3538:
3534:
3530:
3526:
3522:
3515:
3507:
3503:
3499:
3495:
3491:
3487:
3480:
3472:
3468:
3464:
3460:
3453:
3445:
3441:
3437:
3433:
3426:
3418:
3414:
3410:
3406:
3405:
3400:
3394:
3386:
3382:
3378:
3374:
3373:
3368:
3364:
3360:
3354:
3346:
3342:
3338:
3334:
3329:
3324:
3320:
3316:
3312:
3308:
3304:
3297:
3289:
3285:
3281:
3277:
3270:
3262:
3258:
3253:
3248:
3244:
3240:
3236:
3232:
3228:
3221:
3213:
3209:
3204:
3199:
3195:
3191:
3188:(2): 176–94.
3187:
3183:
3179:
3172:
3164:
3160:
3156:
3152:
3145:
3137:
3133:
3129:
3122:
3114:
3110:
3106:
3102:
3098:
3094:
3090:
3086:
3079:
3071:
3067:
3064:: 1249–1262.
3063:
3059:
3055:
3048:
3041:
3035:
3028:
3022:
3018:
3017:
3009:
3001:
2997:
2992:
2987:
2983:
2979:
2975:
2968:
2960:
2956:
2952:
2948:
2944:
2937:
2929:
2925:
2921:
2917:
2913:
2906:
2898:
2894:
2890:
2886:
2882:
2875:
2867:
2861:
2857:
2850:
2848:
2846:
2844:
2842:
2834:
2830:
2826:
2820:
2812:
2808:
2804:
2797:
2789:
2785:
2781:
2777:
2773:
2769:
2762:
2754:
2746:
2738:
2734:
2730:
2726:
2722:
2718:
2711:
2703:
2696:
2688:
2682:
2678:
2674:
2670:
2669:
2661:
2653:
2651:0-85312-988-6
2647:
2643:
2636:
2628:
2624:
2620:
2616:
2612:
2608:
2601:
2593:
2589:
2585:
2581:
2577:
2573:
2565:
2557:
2553:
2549:
2545:
2541:
2537:
2530:
2522:
2516:
2508:
2506:3-527-30517-3
2502:
2498:
2491:
2482:
2477:
2473:
2469:
2465:
2457:
2449:
2447:3-527-30517-3
2443:
2439:
2432:
2424:
2420:
2415:
2410:
2406:
2402:
2398:
2391:
2389:
2380:
2376:
2372:
2368:
2364:
2360:
2356:
2352:
2348:
2344:
2337:
2329:
2323:
2319:
2312:
2306:
2302:
2298:
2294:
2293:
2288:
2283:
2275:
2269:
2265:
2258:
2250:
2246:
2242:
2238:
2237:
2229:
2227:
2218:
2212:
2208:
2201:
2192:
2184:
2180:
2176:
2172:
2168:
2164:
2163:
2155:
2147:
2141:
2137:
2130:
2122:
2118:
2114:
2110:
2106:
2102:
2098:
2094:
2090:
2086:
2082:
2075:
2067:
2063:
2059:
2055:
2051:
2047:
2043:
2039:
2035:
2028:
2020:
2016:
2012:
2008:
2004:
2000:
1993:
1984:
1978:
1974:
1970:
1966:
1962:
1956:
1950:
1946:
1942:
1938:
1937:
1932:
1927:
1919:
1915:
1910:
1905:
1901:
1897:
1893:
1886:
1878:
1874:
1869:
1864:
1860:
1856:
1852:
1848:
1844:
1837:
1829:
1825:
1821:
1817:
1813:
1809:
1805:
1801:
1797:
1790:
1782:
1778:
1774:
1770:
1766:
1762:
1759:(3): 149–55.
1758:
1754:
1746:
1738:
1734:
1730:
1727:
1726:
1718:
1710:
1706:
1702:
1698:
1694:
1690:
1683:
1677:
1673:
1669:
1665:
1664:
1659:
1654:
1648:
1644:
1640:
1636:
1635:
1630:
1625:
1621:
1611:
1608:
1606:
1603:
1600:
1596:
1593:
1590:
1589:homochirality
1586:
1583:
1580:
1577:
1574:
1571:
1570:
1564:
1562:
1558:
1554:
1550:
1546:
1545:Dieter Enders
1542:
1537:
1531:
1527:
1526:Mosher's acid
1523:
1518:
1516:
1512:
1508:
1504:
1495:
1491:
1489:
1485:
1481:
1477:
1461:
1457:
1453:
1448:
1444:
1443:
1440:
1438:
1434:
1430:
1426:
1422:
1418:
1414:
1410:
1406:
1402:
1398:
1394:
1389:
1387:
1383:
1373:
1369:
1367:
1363:
1359:
1354:
1352:
1348:
1336:)-thalidomide
1335:
1330:)-thalidomide
1329:
1322:
1318:
1316:
1311:
1307:
1303:
1299:
1295:
1291:
1286:
1284:
1280:
1276:
1272:
1269:separated by
1261:
1259:
1254:
1250:
1246:
1242:
1238:
1234:
1230:
1226:
1222:
1218:
1214:
1210:
1206:
1198:
1194:
1190:
1187:
1182:
1178:
1176:
1168:
1164:
1156:
1152:
1147:
1143:
1141:
1136:
1132:
1131:Joseph Le Bel
1128:
1124:
1120:
1116:
1112:
1111:Louis Pasteur
1108:
1104:
1089:
1087:
1083:
1079:
1074:
1072:
1071:Cotton effect
1068:
1064:
1060:
1056:
1051:
1049:
1045:
1040:
1034:
1027:
1023:
1018:
1014:
1010:
1006:
1002:
997:
993:
991:
987:
983:
979:
975:
967:
963:
962:boiling point
959:
958:melting point
949:
947:
943:
939:
935:
927:
923:
921:
910:
900:
898:
894:
890:
886:
882:
878:
874:
870:
866:
862:
858:
852:
842:
840:
836:
832:
828:
822:
812:
810:
806:
805:diastereomers
801:
793:
789:
787:
780:
770:
768:
764:
760:
756:
752:
748:
744:
740:
732:
728:
726:
722:
717:
713:
709:
705:
684:
681:
679:
676:
674:
671:
668:
667:
664:
661:
659:
656:
654:
651:
648:
647:
644:
641:
639:
636:
634:
631:
628:
627:
623:
619:
616:
612:
609:
605:
602:
601:
597:
593:
590:
586:
583:
579:
576:
575:
564:
553:
536:
525:
508:
497:
487:
483:
482:
479:
478:
477:
455:
452:
449:
446:
443:
436:
432:
419:
415:
408:
404:
398:
394:
384:
383:
382:
380:
376:
372:
368:
364:
360:
356:
355:rate constant
351:
349:
345:
341:
337:
333:
329:
325:
321:
317:
309:
301:
297:
293:
287:
284:
281:
277:
274:is sold as a
273:
269:
265:
262:
259:
255:
251:
247:
243:
240:
237:
226:
223:
219:
216:
215:
214:
212:
208:
204:
200:
192:
187:
178:
176:
172:
171:diastereomers
168:
164:
159:
155:
153:
149:
145:
141:
137:
133:
129:
125:
121:
117:
110:
101: R group
94:
85:
84:chiral center
81:
73:
68:
47:
43:
39:
34:
30:
19:
5240:
5228:
5216:
5204:
5054:Biosynthesis
4904:Geochemistry
4819:Pharmacology
4795:Cell biology
4785:Biochemistry
4737:
4613:Spectroscopy
4512:VSEPR theory
4361:Spectroscopy
4305:Branches of
4264:Biocatalysis
4137:Diastereomer
4127:Stereoisomer
4090:
4080:Stereocenter
4060:
4059:Concepts in
4015:
4003:
3991:
3903:
3869:Biosynthesis
3790:
3786:
3780:
3763:
3759:
3753:
3736:
3732:
3726:
3701:
3697:
3691:
3674:
3669:
3666:H. S. Mosher
3659:
3642:
3638:
3632:
3615:
3611:
3605:
3588:
3584:
3578:
3561:
3555:
3549:
3524:
3520:
3514:
3489:
3485:
3479:
3462:
3458:
3452:
3435:
3431:
3425:
3408:
3402:
3393:
3376:
3370:
3353:
3310:
3306:
3296:
3279:
3275:
3269:
3234:
3230:
3220:
3185:
3181:
3171:
3154:
3150:
3144:
3127:
3121:
3088:
3084:
3078:
3061:
3057:
3047:
3034:
3015:
3008:
2981:
2977:
2967:
2950:
2946:
2936:
2919:
2915:
2905:
2888:
2884:
2874:
2855:
2832:
2828:
2819:
2810:
2806:
2796:
2771:
2767:
2761:
2751:
2745:
2720:
2716:
2710:
2701:
2695:
2667:
2660:
2641:
2635:
2610:
2606:
2600:
2575:
2571:
2564:
2539:
2535:
2529:
2496:
2490:
2471:
2467:
2456:
2437:
2431:
2404:
2400:
2346:
2342:
2336:
2317:
2311:
2297:Biocatalysis
2290:
2282:
2263:
2257:
2240:
2234:
2206:
2200:
2191:
2166:
2160:
2154:
2135:
2129:
2088:
2084:
2074:
2041:
2037:
2027:
2002:
1998:
1992:
1972:
1955:
1934:
1926:
1899:
1895:
1885:
1850:
1846:
1836:
1803:
1799:
1795:
1789:
1756:
1752:
1745:
1728:
1723:
1717:
1692:
1688:
1682:
1661:
1653:
1632:
1624:
1563:precursors.
1538:
1519:
1500:
1473:
1419:with chiral
1397:Ryōji Noyori
1390:
1379:
1370:
1355:
1344:
1333:
1327:
1317:separation.
1287:
1267:
1257:
1233:biosynthesis
1221:malonic acid
1202:
1184:Brucine, an
1160:
1151:malonic acid
1118:
1114:
1100:
1075:
1052:
1041:
998:
994:
955:
931:
912:
854:
824:
821:Biocatalysis
815:Biocatalysis
797:
786:racemization
782:
736:
701:
562:
551:
534:
523:
506:
495:
485:
474:
378:
374:
366:
358:
352:
313:
286:Drug safety:
285:
279:
263:
253:
248:smells like
241:
238:
217:
196:
160:
156:
144:enantiomeric
119:
115:
114:
29:
5242:WikiProject
4467:Theoretical
4452:Calorimetry
3557:Tetrahedron
3157:: 364–368.
2978:Biopolymers
2953:: 349–354.
2723:(1): 1–16.
1902:(1): 4–30.
1476:Schiff base
1415:ligands in
1358:teratogenic
1351:thalidomide
1341:Thalidomide
1283:polarimeter
1191:related to
1135:tetrahedral
1123:Lord Kelvin
1115:dissymmetry
1059:polarimeter
1035:and Eu(hfc)
946:enantiopure
294:is used in
203:amino acids
167:enantiomers
5258:Categories
5078:Metallurgy
4777:Biological
4343:Analytical
4132:Enantiomer
3527:(3): 222.
3276:The Lancet
2813:: 337–347.
1616:References
1591:in nature.
1579:Kelliphite
1541:E.J. Corey
1490:reaction.
1382:CIP system
1258:un-natural
1229:lovastatin
1193:strychnine
1155:levorotary
1088:be grown.
934:amino acid
920:2 reaction
693:Approaches
316:enthalpies
308:pyridoxine
272:Citalopram
207:enantiomer
5274:Asymmetry
5140:Catalysis
4648:Inorganic
4442:Titration
4307:chemistry
4232:Reactions
4075:Chirality
2717:Chirality
2515:cite book
2468:Chem. Rev
2236:Synthesis
2105:0001-4842
2058:0001-4842
1987:Page 1226
1689:Chirality
1595:Tacticity
1364:(US) and
1119:chirality
887:with the
857:catalysis
704:catalysts
488:* (kcal)
453:×
447:×
437:∗
429:Δ
426:Δ
336:substrate
320:entropies
250:spearmint
225:aspartame
136:chirality
5206:Category
5162:Molecule
5099:See also
4524:Physical
4168:Analysis
3993:Category
3506:19746594
3345:12391084
3337:21207098
3328:11114848
3261:16992790
3212:16992694
3130:: 3940.
3000:20577995
2788:17328087
2737:20589938
2627:19623342
2592:15311957
2556:11668532
2423:15593081
2371:11196655
2183:16802397
2121:73503362
2113:30840435
2066:28128920
2019:22306968
1971:(2001).
1918:19414517
1877:14236210
1828:20110906
1781:36768144
1773:15107657
1709:23034823
1599:polymers
1567:See also
1530:europium
1439:ligand.
1332:Right: (
1281:using a
1249:vitalism
1211:, for a
1186:alkaloid
1161:In 1894
1026:europium
984:. Their
966:polarity
873:hydrogen
344:catalyst
302:whereas
252:whereas
218:Flavour:
181:Overview
109:Hydrogen
5218:Commons
5182:Alchemy
4698:Organic
4005:Commons
3718:5798101
3529:Bibcode
3252:1465734
3203:1540678
3113:4264310
3093:Bibcode
2704:. SPIE.
2379:4340563
2351:Bibcode
1820:1632943
1484:styrene
1326:Left: (
1213:brucine
1092:History
1030:Eu(fod)
885:proline
839:statins
827:enzymes
763:alkenes
739:ligands
716:achiral
570:
548:
542:
520:
514:
492:
340:reagent
324:racemic
276:racemic
258:caraway
246:carvone
82:at the
36:In the
5230:Portal
4376:UV-Vis
4017:Portal
3716:
3504:
3343:
3335:
3325:
3259:
3249:
3210:
3200:
3111:
3085:Nature
3023:
2998:
2862:
2786:
2735:
2683:
2648:
2625:
2590:
2554:
2503:
2444:
2421:
2377:
2369:
2343:Nature
2324:
2270:
2213:
2181:
2142:
2119:
2111:
2103:
2064:
2056:
2017:
1979:
1916:
1875:
1868:289631
1865:
1826:
1818:
1779:
1771:
1707:
1561:statin
1437:DIPAMP
1433:L-DOPA
1368:(EU).
1243:, and
1001:quartz
881:chiral
877:sulfur
869:carbon
753:, and
672:10500
381:, is:
199:sugars
130:as "a
107:
105:
99:
91:
89:
80:Carbon
78:
76:
50:Key: R
42:AD-mix
4403:MALDI
4371:Raman
3919:LASiS
3847:Types
3341:S2CID
3109:S2CID
2375:S2CID
2287:IUPAC
2117:S2CID
1931:IUPAC
1824:S2CID
1777:S2CID
1658:IUPAC
1629:IUPAC
1528:, or
1046:. or
831:e.e.s
751:Salen
747:BINOL
682:2510
677:4830
652:1650
270:drug
244:-(–)-
239:Odor:
128:IUPAC
95:group
5157:Atom
4425:HPLC
4160:(de)
4154:(ee)
3714:PMID
3502:PMID
3333:PMID
3257:PMID
3208:PMID
3021:ISBN
2996:PMID
2860:ISBN
2784:PMID
2753:book
2733:PMID
2681:ISBN
2646:ISBN
2623:PMID
2588:PMID
2552:PMID
2521:link
2501:ISBN
2442:ISBN
2419:PMID
2367:PMID
2322:ISBN
2268:ISBN
2241:2006
2211:ISBN
2179:PMID
2140:ISBN
2109:PMID
2101:ISSN
2062:PMID
2054:ISSN
2015:PMID
1977:ISBN
1914:PMID
1873:PMID
1816:PMID
1769:PMID
1705:PMID
1399:and
1315:HPLC
1292:and
1129:and
1053:The
1013:HPLC
1011:and
988:and
980:and
978:HPLC
765:and
669:5.0
662:524
657:886
649:4.0
642:109
637:162
632:259
629:3.0
603:2.0
598:.78
591:.46
584:.37
577:1.0
450:1.98
318:and
266:the
220:the
201:and
5167:Ion
4398:ICP
4381:NMR
3795:doi
3768:doi
3741:doi
3706:doi
3679:doi
3647:doi
3620:doi
3616:110
3593:doi
3589:102
3566:doi
3537:doi
3494:doi
3467:doi
3440:doi
3413:doi
3381:doi
3323:PMC
3315:doi
3284:doi
3280:278
3247:PMC
3239:doi
3198:PMC
3190:doi
3159:doi
3132:doi
3101:doi
3089:168
3066:doi
2986:doi
2955:doi
2924:doi
2893:doi
2776:doi
2725:doi
2673:doi
2615:doi
2580:doi
2544:doi
2476:doi
2472:107
2409:doi
2359:doi
2347:409
2301:doi
2299:".
2245:doi
2171:doi
2093:doi
2046:doi
2007:doi
1945:doi
1943:".
1904:doi
1900:110
1863:PMC
1855:doi
1808:doi
1761:doi
1733:doi
1697:doi
1672:doi
1670:".
1643:doi
1641:".
1522:NMR
1482:of
1080:by
986:NMR
972:in
755:BOX
624:.9
617:.8
610:.6
456:2.3
189:An
169:or
154:."
146:or
5260::
4897:/
4893:/
4745:/
4420:GC
4393:EI
4366:IR
3789:.
3762:.
3737:43
3735:.
3712:.
3702:91
3700:.
3675:34
3643:97
3641:.
3614:.
3587:.
3562:24
3560:.
3535:.
3525:63
3523:.
3500:.
3490:41
3488:.
3463:99
3461:.
3434:.
3409:21
3407:.
3375:.
3365:;
3361:;
3339:.
3331:.
3321:.
3311:68
3309:.
3305:.
3278:.
3255:.
3245:.
3235:32
3233:.
3229:.
3206:.
3196:.
3186:30
3184:.
3180:.
3153:.
3107:.
3099:.
3087:.
3062:85
3060:.
3056:.
2994:.
2982:93
2980:.
2976:.
2951:37
2949:.
2945:.
2920:22
2918:.
2914:.
2889:27
2887:.
2883:.
2840:^
2831:,
2811:22
2809:.
2782:.
2772:46
2770:.
2731:.
2721:23
2719:.
2679:.
2671:.
2621:.
2611:38
2609:.
2586:.
2576:37
2574:.
2550:.
2540:40
2538:.
2517:}}
2513:{{
2470:.
2466:.
2417:.
2405:44
2403:.
2399:.
2387:^
2373:.
2365:.
2357:.
2345:.
2289:,
2239:.
2225:^
2177:.
2167:45
2165:.
2115:.
2107:.
2099:.
2089:52
2087:.
2083:.
2060:.
2052:.
2042:50
2040:.
2036:.
2013:.
2003:41
2001:.
1967:;
1933:,
1912:.
1898:.
1894:.
1871:.
1861:.
1851:43
1849:.
1845:.
1822:.
1814:.
1804:88
1802:.
1775:.
1767:.
1757:19
1755:.
1729:19
1703:.
1693:24
1691:.
1660:,
1631:,
1509:,
1505:,
1395:,
1239:,
1207:,
1177:.
1142:.
1073:.
1039:.
1009:GC
990:IR
982:GC
964:,
960:,
922:.
875:,
871:,
769:.
749:,
727:.
620:22
613:29
606:40
484:ΔΔ
420:10
357:,
342:,
338:,
177:.
5131:"
5127:"
4298:e
4291:t
4284:v
4094:2
4091:C
4052:e
4045:t
4038:v
3832:e
3825:t
3818:v
3801:.
3797::
3791:4
3774:.
3770::
3764:6
3747:.
3743::
3720:.
3708::
3685:.
3681::
3653:.
3649::
3626:.
3622::
3599:.
3595::
3572:.
3568::
3543:.
3539::
3531::
3508:.
3496::
3473:.
3469::
3446:.
3442::
3436:7
3419:.
3415::
3387:.
3383::
3377:5
3347:.
3317::
3290:.
3286::
3263:.
3241::
3214:.
3192::
3165:.
3161::
3155:3
3138:.
3134::
3115:.
3103::
3095::
3072:.
3068::
3002:.
2988::
2961:.
2957::
2930:.
2926::
2899:.
2895::
2868:.
2833:9
2790:.
2778::
2755:.
2739:.
2727::
2689:.
2675::
2654:.
2629:.
2617::
2594:.
2582::
2558:.
2546::
2523:)
2509:.
2484:.
2478::
2450:.
2425:.
2411::
2381:.
2361::
2353::
2330:.
2303::
2276:.
2251:.
2247::
2219:.
2185:.
2173::
2148:.
2123:.
2095::
2068:.
2048::
2021:.
2009::
1985:.
1947::
1920:.
1906::
1879:.
1857::
1830:.
1810::
1796:S
1783:.
1763::
1739:.
1735::
1711:.
1699::
1674::
1645::
1534:3
1334:R
1328:S
1171:D
1037:3
1032:3
970:f
918:N
916:S
594:4
587:5
580:6
566:2
563:k
559:/
555:1
552:k
538:2
535:k
531:/
527:1
524:k
510:2
507:k
503:/
499:1
496:k
486:G
444:T
433:G
416:=
409:2
405:k
399:1
395:k
379:T
375:G
359:k
304:L
290:D
280:S
260:.
254:S
242:R
233:D
229:L
142:(
60:S
56:M
52:L
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.