1057:
881:
68:
59:
902:
1645:
Lee, Donald G.; Chen, Tao (1993), "Reduction of manganate(VI) by mandelic acid and its significance for development of a general mechanism of oxidation of organic compounds by high-valent transition metal oxides",
1722:
Young, R.; Critchley, J. A.; Young, K. K.; Freebairn, R. C.; Reynolds, A. P.; Lolin, Y. I. (1996). "Fatal acute hepatorenal failure following potassium permanganate ingestion".
1162:
is affected if the permanganate is swallowed. Only a limited amount is absorbed by the intestines, but this small amount shows severe effects on the kidneys and on the liver.
1996:
1886:
893:
This is a common and strong disinfectant, used regularly to sanitize baths, toilets, and wash basins. It is a cheap and extremely effective compound for the task.
943:). According to theory, permanganate is strong enough to oxidize water, but this does not actually happen to any extent. Besides this, it is stable.
1623:
1541:
1358:
1362:
488:
media. The exact chemical reaction depends on the carbon-containing reactants present and the oxidant used. For example, trichloroethane (C
285:
1150:
The fatal dose of permanganate is about 10 g, and several fatal intoxications have occurred. The strong oxidative effect leads to
1782:
132:
1227:
Sukalyan Dash, Sabita Patel & Bijay K. Mishra (2009). "Oxidation by permanganate: synthetic and mechanistic aspects".
1056:
2329:
1302:
1277:
260:
363:
477:
80:
1563:
Kovacs KA, Grof P, Burai L, Riedel M (2004). "Revising the
Mechanism of the Permanganate/Oxalate Reaction".
921:
473:
208:
1775:
1760:
229:
246:
1597:
917:
884:
A series of potassium permanganate solutions with varying concentration, increasing to the right.
2324:
1915:
1874:
1768:
1117:
958:
951:
1988:
1949:
1843:
1617:
1535:
1352:
1069:
906:
1518:"Carboxylic acids from the oxidation of terminal alkenes by permanganate: nonadecanoic acid"
2074:
2042:
1923:
1826:
1572:
1108:
1095:
217:
172:
108:
1207:
8:
2082:
2034:
1866:
1135:
1126:
1082:
1009:
962:
763:
98:
1576:
152:
1747:
1699:
1674:
1177:
939:. It is therefore in common use in qualitative analysis that involves redox reactions (
822:
755:
1739:
1704:
1604:
1522:
1489:
1456:
1423:
1390:
1339:
1319:
1298:
1273:
966:
880:
830:
759:
714:
433:
1268:
Cotton, F. Albert; Wilkinson, Geoffrey; Carlos A. Murillo; Manfred
Bochmann (1999).
1807:
1751:
1731:
1694:
1686:
1655:
1580:
1515:
1236:
913:
660:
457:
382:
348:
308:
197:
1212:
1155:
1029:
1025:
1021:
940:
469:
465:
1599:
1517:
1484:
1451:
1418:
1385:
1334:
1267:
905:
Absorption spectrum of an aqueous solution of potassium permanganate, showing a
1735:
1037:
1005:
453:
357:
343:
1332:
1240:
67:
2318:
1188:
45:
1182:
862:
767:
1790:
1743:
1708:
1598:
E. J. Witzemann, Wm. Lloyd Evans, Henry Hass, and E. F. Schroeder (1931).
1254:
1226:
754:
Permanganates can be produced by oxidation of manganese compounds such as
1675:"Potassium permanganate poisoning – a rare cause of fatal self poisoning"
1171:
1033:
936:
1690:
1659:
901:
954:
is used as a disinfectant and water treatment additive in aquaculture.
330:
163:
1584:
1159:
826:
668:
621:
461:
29:
1383:
356:
Except where otherwise noted, data are given for materials in their
1272:(6th ed.). New York: John Wiley & Sons, Inc. p. 770.
1151:
1017:
932:
664:
485:
58:
131:
1449:
1051:
In alkene oxidations one intermediate is a cyclic Mn(V) species:
947:
184:
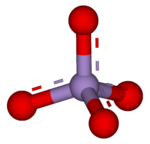
480:. Permanganate solutions are purple in colour and are stable in
1041:
1013:
970:
916:
of permanganic acid. They have a deep purple colour, due to a
388:
1293:
Miessler, Gary L.; Fischer, Paul J.; Tarr, Donald A. (2014).
1001:
957:
Manganates(VII) are not very stable thermally. For instance,
713:
In a neutral solution, however, it gets reduced to the brown
500:) is oxidised by permanganate ions to form carbon dioxide (CO
143:
121:
1721:
1516:
Donald G. Lee, Shannon E. Lamb, and Victor S. Chang (1981).
424:
234:
1045:
865:
or air oxidation of alkaline solutions of manganate salts (
617:
412:
406:
400:
437:
403:
397:
620:
solution, permanganate(VII) is reduced to the pale pink
1333:
A. Calder, A. R. Forrester1, and S. P. Hepburn (1972).
950:, but it is not very selective with organic compounds.
927:
orbitals to empty orbitals derived from manganese(VII)
481:
1562:
1255:"Geo-Cleanse International, INC. | Permanganate"
409:
667:
solution, permanganate(VII) is reduced to the green
421:
415:
385:
1292:
394:
391:
2316:
196:
1416:
107:
1673:Ong, K. L.; Tan, T. H.; Cheung, W. L. (1997).
1482:
508:), hydrogen ions (H), and chloride ions (Cl).
1776:
1384:Nathan Kornblum and Willard J. Jones (1963).
16:"MnO4" redirects here. For the manganate ion
1672:
1622:: CS1 maint: multiple names: authors list (
1540:: CS1 maint: multiple names: authors list (
1357:: CS1 maint: multiple names: authors list (
1320:"Potassium Permanganate Sodium Disinfectant"
1174:, a similar ion with a chlorine(VII) center
778:+ 5 NaClO + 6 NaOH → 2 NaMnO
1783:
1769:
1450:R. L. Shriner and E. C. Kleiderer (1930).
1361:) CS1 maint: numeric names: authors list (
1185:, which is isoelectronic with permanganate
762:by strong oxidizing agents, for instance,
171:
1698:
1638:
1335:"2-methyl-2-nitrosopropane and its dimer"
216:
931:orbitals. Permanganate(VII) is a strong
900:
879:
468:, the permanganate(VII) ion is a strong
1791:Salts and covalent derivatives of the
1644:
245:
2317:
1816:
1297:(5th ed.). Pearson. p. 430.
2205:
1764:
272:Key: NPDODHDPVPPRDJ-UHFFFAOYSA-N
151:
624:(Mn) with an oxidation state of +2.
1724:Human & Experimental Toxicology
187:
13:
1317:
861:They are produced commercially by
14:
2341:
1600:"dl-glyceraldehyde ethyl acetal"
1386:"4-nitro-2,2,4-trimethylpentane"
1055:
381:
66:
57:
1715:
1666:
1591:
1556:
1509:
1476:
1443:
1048:. This list is not exhaustive.
821:It may also be produced by the
721:with an oxidation state of +4.
683:with an oxidation state of +6.
360:(at 25 °C , 100 kPa).
1410:
1377:
1326:
1311:
1286:
1261:
1247:
1220:
1213:Merriam-Webster.com Dictionary
1200:
1000:A permanganate can oxidize an
1:
1194:
961:decomposes at 230 °C to
896:
749:
1270:Advanced Inorganic Chemistry
1063:
32:. For the hypomanganate ion
7:
1165:
10:
2346:
2330:Transition metal oxyanions
2207:
1736:10.1177/096032719601500313
1679:Emergency Medicine Journal
1633:, vol. 11, p. 52
1551:, vol. 60, p. 11
1504:, vol. 16, p. 39
1471:, vol. 10, p. 82
1438:, vol. 31, p. 59
1405:, vol. 43, p. 87
1372:, vol. 52, p. 77
651:+ 5 e → Mn + 4 H
15:
1818:
1800:
1241:10.1016/j.tet.2008.10.038
1145:
504:), manganese dioxide (MnO
354:
301:
281:
256:
91:
79:
74:
65:
56:
1417:J. W. Cornforth (1951).
888:
782:+ 9 NaCl + 3 H
436:with the manganate(VII)
1483:John R. Ruhoff (1936).
912:Permanganates(VII) are
1118:Potassium permanganate
959:potassium permanganate
952:Potassium permanganate
909:
885:
269:InChI=1S/Mn.4O/q;;;;-1
1070:Ammonium permanganate
904:
883:
478:tetrahedral structure
81:Systematic IUPAC name
1109:Lithium permanganate
1096:Calcium permanganate
907:vibronic progression
474:transition metal ion
1691:10.1136/emj.14.1.43
1660:10.1021/ja00077a023
1577:2004JPCA..10811026K
1295:Inorganic Chemistry
1158:. For example, the
1136:Silver permanganate
1127:Sodium permanganate
1083:Barium permanganate
963:potassium manganate
833:as a side-product:
764:sodium hypochlorite
338: g·mol
53:
1485:"n-heptanoic acid"
1452:"Piperonylic acid"
1216:. Merriam-Webster.
1178:Permanganate index
910:
886:
823:disproportionation
756:manganese chloride
364:Infobox references
51:
2312:
2311:
2306:
2305:
1648:J. Am. Chem. Soc.
1631:Collected Volumes
1605:Organic Syntheses
1585:10.1021/jp047061u
1549:Collected Volumes
1523:Organic Syntheses
1502:Collected Volumes
1490:Organic Syntheses
1469:Collected Volumes
1457:Organic Syntheses
1436:Collected Volumes
1424:Organic Syntheses
1403:Collected Volumes
1391:Organic Syntheses
1370:Collected Volumes
1340:Organic Syntheses
967:manganese dioxide
935:, and similar to
831:manganese dioxide
760:manganese sulfate
715:manganese dioxide
434:chemical compound
372:Chemical compound
370:
369:
230:CompTox Dashboard
133:Interactive image
2337:
1804:
1803:
1785:
1778:
1771:
1762:
1761:
1756:
1755:
1719:
1713:
1712:
1702:
1670:
1664:
1662:
1654:(24): 11231–36,
1642:
1636:
1634:
1627:
1621:
1613:
1595:
1589:
1588:
1565:J. Phys. Chem. A
1560:
1554:
1552:
1545:
1539:
1531:
1513:
1507:
1505:
1498:
1480:
1474:
1472:
1465:
1447:
1441:
1439:
1432:
1419:"Ethyl pyruvate"
1414:
1408:
1406:
1399:
1381:
1375:
1373:
1366:
1356:
1348:
1330:
1324:
1323:
1315:
1309:
1308:
1290:
1284:
1283:
1265:
1259:
1258:
1251:
1245:
1244:
1224:
1218:
1217:
1204:
1059:
920:transition from
876:
875:
874:
849:O → 2 NaMnO
741:+ 3 e → MnO
740:
739:
738:
709:
708:
707:
697:
696:
695:
682:
681:
680:
650:
649:
648:
638:
637:
636:
612:
611:
610:
601:
599:
598:
588:
587:
586:
577:
576:
575:
565:
564:
563:
553:
552:
551:
543:
542:
534:
533:
523:
522:
521:
458:permanganic acid
451:
450:
449:
431:
430:
427:
426:
423:
418:
417:
414:
411:
408:
405:
402:
399:
396:
393:
390:
387:
349:Permanganic acid
337:
324:
323:
322:
309:Chemical formula
249:
238:
236:
220:
200:
189:
175:
155:
135:
111:
70:
61:
54:
50:
43:
42:
41:
38:
27:
26:
25:
22:
2345:
2344:
2340:
2339:
2338:
2336:
2335:
2334:
2315:
2314:
2313:
2308:
2307:
2090:
2086:
2078:
2050:
2046:
2038:
2004:
2000:
1992:
1931:
1927:
1919:
1894:
1890:
1882:
1878:
1870:
1851:
1847:
1830:
1811:
1796:
1789:
1759:
1720:
1716:
1671:
1667:
1643:
1639:
1629:
1615:
1614:
1596:
1592:
1561:
1557:
1547:
1533:
1532:
1514:
1510:
1500:
1481:
1477:
1467:
1448:
1444:
1434:
1415:
1411:
1401:
1382:
1378:
1368:
1350:
1349:
1331:
1327:
1316:
1312:
1305:
1291:
1287:
1280:
1266:
1262:
1253:
1252:
1248:
1225:
1221:
1206:
1205:
1201:
1197:
1168:
1156:mucous membrane
1148:
1141:
1132:
1123:
1114:
1105:
1101:
1092:
1088:
1079:
1075:
1066:
1030:carboxylic acid
1026:terminal alkene
1022:carboxylic acid
996:
992:
988:
984:
980:
946:It is a useful
941:permanganometry
918:charge transfer
899:
891:
873:
870:
869:
868:
866:
856:
852:
848:
844:
840:
816:
812:
808:
804:
800:
796:
792:
785:
781:
777:
752:
744:
737:
734:
733:
732:
730:
728:
720:
706:
703:
702:
701:
699:
694:
691:
690:
689:
687:
679:
676:
675:
674:
672:
654:
647:
644:
643:
642:
640:
635:
633:
632:
631:
629:
609:
607:
606:
605:
603:
597:
594:
593:
592:
590:
585:
583:
582:
581:
579:
574:
571:
570:
569:
567:
562:
559:
558:
557:
555:
550:
547:
546:
545:
541:
538:
537:
536:
532:
529:
528:
527:
525:
520:
517:
516:
515:
513:
507:
503:
499:
495:
491:
472:. The ion is a
470:oxidising agent
466:oxidation state
448:
445:
444:
443:
441:
420:
384:
380:
373:
366:
361:
335:
321:
318:
317:
316:
314:
311:
297:
294:
289:
288:
277:
274:
273:
270:
264:
263:
252:
239:
232:
223:
203:
190:
178:
158:
138:
125:
114:
101:
87:
86:
49:
39:
36:
35:
33:
23:
20:
19:
17:
12:
11:
5:
2343:
2333:
2332:
2327:
2310:
2309:
2304:
2303:
2300:
2297:
2294:
2291:
2288:
2285:
2282:
2279:
2276:
2273:
2270:
2267:
2264:
2261:
2257:
2256:
2253:
2250:
2247:
2244:
2241:
2238:
2235:
2232:
2229:
2226:
2223:
2220:
2217:
2214:
2210:
2209:
2206:
2203:
2202:
2199:
2196:
2193:
2190:
2187:
2184:
2181:
2178:
2175:
2172:
2169:
2166:
2163:
2160:
2157:
2154:
2151:
2148:
2144:
2143:
2140:
2137:
2134:
2131:
2128:
2125:
2122:
2119:
2116:
2113:
2110:
2107:
2104:
2101:
2098:
2095:
2092:
2088:
2084:
2080:
2076:
2071:
2070:
2067:
2064:
2061:
2058:
2055:
2052:
2048:
2044:
2040:
2036:
2032:
2029:
2026:
2023:
2020:
2017:
2014:
2011:
2008:
2006:
2002:
1998:
1994:
1990:
1985:
1984:
1981:
1978:
1975:
1972:
1969:
1966:
1963:
1960:
1957:
1954:
1951:
1947:
1944:
1941:
1938:
1935:
1933:
1929:
1925:
1921:
1917:
1912:
1911:
1908:
1905:
1902:
1899:
1896:
1892:
1888:
1884:
1880:
1876:
1872:
1868:
1863:
1862:
1859:
1856:
1853:
1849:
1845:
1841:
1838:
1835:
1832:
1828:
1823:
1822:
1819:
1817:
1815:
1813:
1809:
1802:
1801:
1798:
1797:
1788:
1787:
1780:
1773:
1765:
1758:
1757:
1714:
1665:
1637:
1590:
1555:
1508:
1475:
1442:
1409:
1376:
1325:
1310:
1304:978-0321811059
1303:
1285:
1279:978-0471199571
1278:
1260:
1246:
1235:(4): 707–739.
1219:
1208:"permanganate"
1198:
1196:
1193:
1192:
1191:
1186:
1180:
1175:
1167:
1164:
1147:
1144:
1143:
1142:
1139:
1133:
1130:
1124:
1121:
1115:
1112:
1106:
1103:
1099:
1093:
1090:
1086:
1080:
1077:
1073:
1065:
1062:
1061:
1060:
1038:carbon dioxide
1006:nitro compound
998:
997:
994:
990:
986:
982:
978:
898:
895:
890:
887:
871:
859:
858:
854:
850:
846:
842:
838:
819:
818:
814:
810:
806:
802:
798:
794:
790:
787:
783:
779:
775:
751:
748:
747:
746:
742:
735:
726:
718:
711:
710:
704:
692:
677:
659:In a strongly
657:
656:
652:
645:
634:
614:
613:
608:
595:
584:
572:
560:
548:
539:
530:
518:
505:
501:
497:
493:
489:
464:atom has a +7
460:. Because the
454:conjugate base
446:
371:
368:
367:
362:
358:standard state
355:
352:
351:
346:
344:Conjugate acid
340:
339:
333:
327:
326:
319:
312:
307:
304:
303:
299:
298:
296:
295:
292:
284:
283:
282:
279:
278:
276:
275:
271:
268:
267:
259:
258:
257:
254:
253:
251:
250:
247:DTXSID90912340
242:
240:
228:
225:
224:
222:
221:
213:
211:
205:
204:
202:
201:
193:
191:
183:
180:
179:
177:
176:
168:
166:
160:
159:
157:
156:
148:
146:
140:
139:
137:
136:
128:
126:
119:
116:
115:
113:
112:
104:
102:
97:
94:
93:
89:
88:
84:
83:
77:
76:
72:
71:
63:
62:
9:
6:
4:
3:
2:
2342:
2331:
2328:
2326:
2325:Permanganates
2323:
2322:
2320:
2301:
2298:
2295:
2292:
2289:
2286:
2283:
2280:
2277:
2274:
2271:
2268:
2265:
2262:
2259:
2258:
2254:
2251:
2248:
2245:
2242:
2239:
2236:
2233:
2230:
2227:
2224:
2221:
2218:
2215:
2212:
2211:
2204:
2200:
2197:
2194:
2191:
2188:
2185:
2182:
2179:
2176:
2173:
2170:
2167:
2164:
2161:
2158:
2155:
2152:
2149:
2146:
2145:
2141:
2138:
2135:
2132:
2129:
2126:
2123:
2120:
2117:
2114:
2111:
2108:
2105:
2102:
2099:
2096:
2093:
2091:
2081:
2079:
2073:
2072:
2068:
2065:
2062:
2059:
2056:
2053:
2051:
2041:
2039:
2033:
2030:
2027:
2024:
2021:
2018:
2015:
2012:
2009:
2007:
2005:
1995:
1993:
1987:
1986:
1982:
1979:
1976:
1973:
1970:
1967:
1964:
1961:
1958:
1955:
1952:
1950:
1948:
1945:
1942:
1939:
1936:
1934:
1932:
1922:
1920:
1914:
1913:
1909:
1906:
1903:
1900:
1897:
1895:
1885:
1883:
1873:
1871:
1865:
1864:
1860:
1857:
1854:
1852:
1842:
1839:
1836:
1833:
1831:
1825:
1824:
1820:
1814:
1812:
1806:
1805:
1799:
1794:
1786:
1781:
1779:
1774:
1772:
1767:
1766:
1763:
1753:
1749:
1745:
1741:
1737:
1733:
1730:(3): 259–61.
1729:
1725:
1718:
1710:
1706:
1701:
1696:
1692:
1688:
1684:
1680:
1676:
1669:
1661:
1657:
1653:
1649:
1641:
1632:
1625:
1619:
1611:
1607:
1606:
1601:
1594:
1586:
1582:
1578:
1574:
1571:(50): 11026.
1570:
1566:
1559:
1550:
1543:
1537:
1529:
1525:
1524:
1519:
1512:
1503:
1496:
1492:
1491:
1486:
1479:
1470:
1463:
1459:
1458:
1453:
1446:
1437:
1430:
1426:
1425:
1420:
1413:
1404:
1397:
1393:
1392:
1387:
1380:
1371:
1364:
1360:
1354:
1346:
1342:
1341:
1336:
1329:
1321:
1314:
1306:
1300:
1296:
1289:
1281:
1275:
1271:
1264:
1256:
1250:
1242:
1238:
1234:
1230:
1223:
1215:
1214:
1209:
1203:
1199:
1190:
1189:Pertechnetate
1187:
1184:
1181:
1179:
1176:
1173:
1170:
1169:
1163:
1161:
1157:
1153:
1137:
1134:
1128:
1125:
1119:
1116:
1110:
1107:
1097:
1094:
1084:
1081:
1071:
1068:
1067:
1058:
1054:
1053:
1052:
1049:
1047:
1043:
1039:
1035:
1031:
1027:
1023:
1019:
1015:
1011:
1007:
1003:
976:
975:
974:
972:
968:
964:
960:
955:
953:
949:
944:
942:
938:
934:
930:
926:
923:
919:
915:
908:
903:
894:
882:
878:
864:
857:+ 4 NaOH
836:
835:
834:
832:
828:
824:
809:+ 5 PbSO
805:→ 2 HMnO
788:
773:
772:
771:
769:
765:
761:
757:
724:
723:
722:
716:
686:
685:
684:
670:
666:
662:
627:
626:
625:
623:
622:manganese(II)
619:
511:
510:
509:
487:
483:
479:
475:
471:
467:
463:
459:
455:
439:
435:
429:
378:
365:
359:
353:
350:
347:
345:
342:
341:
334:
332:
329:
328:
313:
310:
306:
305:
300:
291:
290:
287:
280:
266:
265:
262:
255:
248:
244:
243:
241:
231:
227:
226:
219:
215:
214:
212:
210:
207:
206:
199:
195:
194:
192:
186:
182:
181:
174:
170:
169:
167:
165:
162:
161:
154:
150:
149:
147:
145:
142:
141:
134:
130:
129:
127:
123:
118:
117:
110:
106:
105:
103:
100:
96:
95:
90:
82:
78:
73:
69:
64:
60:
55:
52:Permanganate
47:
46:Hypomanganate
31:
1793:permanganate
1792:
1727:
1723:
1717:
1685:(1): 43–45.
1682:
1678:
1668:
1651:
1647:
1640:
1630:
1618:cite journal
1609:
1603:
1593:
1568:
1564:
1558:
1548:
1536:cite journal
1527:
1521:
1511:
1501:
1494:
1488:
1478:
1468:
1461:
1455:
1445:
1435:
1428:
1422:
1412:
1402:
1395:
1389:
1379:
1369:
1353:cite journal
1344:
1338:
1328:
1313:
1294:
1288:
1269:
1263:
1249:
1232:
1228:
1222:
1211:
1202:
1149:
1050:
999:
969:, releasing
956:
945:
928:
924:
911:
892:
863:electrolysis
860:
820:
793:+ 5 PbO
768:lead dioxide
753:
712:
658:
615:
484:or slightly
377:permanganate
376:
374:
92:Identifiers
85:Permanganate
1229:Tetrahedron
1172:Perchlorate
1034:oxalic acid
977:2 KMnO
937:perchlorate
789:2 MnSO
774:2 MnCl
745:+ 4 OH
302:Properties
153:CHEBI:25939
2319:Categories
1195:References
922:oxo ligand
897:Properties
845:+ 2 H
827:manganates
813:+ 2 H
797:+ 3 H
750:Production
331:Molar mass
293:(=O)(=O)=O
218:2BL953CCZ2
164:ChemSpider
120:3D model (
109:14333-13-2
99:CAS Number
1160:esophagus
1064:Compounds
1040:, and an
837:3 Na
669:manganate
462:manganese
30:Manganate
1318:Syndel.
1183:Chromate
1166:See also
1152:necrosis
1098:, Ca(MnO
1085:, Ba(MnO
1018:aldehyde
933:oxidizer
725:2 H
665:alkaline
486:alkaline
2208:
1752:8993404
1744:8839216
1709:9023625
1700:1342846
1573:Bibcode
1154:of the
1138:, AgMnO
1129:, NaMnO
1111:, LiMnO
1010:alcohol
948:reagent
829:, with
628:8
482:neutral
476:with a
432:) is a
336:118.934
325:
185:PubChem
2083:Ba(MnO
2043:Cd(MnO
1997:Sr(MnO
1924:Ca(MnO
1887:Al(MnO
1875:Mg(MnO
1750:
1742:
1707:
1697:
1301:
1276:
1146:Safety
1120:, KMnO
1042:alkene
1014:ketone
971:oxygen
698:+ e →
618:acidic
616:In an
452:, the
286:SMILES
75:Names
44:, see
28:, see
2075:CsMnO
2035:AgMnO
1989:RbMnO
1867:NaMnO
1827:LiMnO
1748:S2CID
1612:: 307
1530:: 397
1497:: 315
1464:: 538
1431:: 467
1398:: 845
1347:: 803
1044:to a
1028:to a
1020:to a
1016:, an
1012:to a
1008:, an
1004:to a
1002:amine
989:+ MnO
973:gas:
914:salts
889:Usage
853:+ MnO
671:ion,
661:basic
261:InChI
198:24401
173:22811
144:ChEBI
122:JSmol
1916:KMnO
1808:HMnO
1740:PMID
1705:PMID
1624:link
1542:link
1363:link
1359:link
1299:ISBN
1274:ISBN
1072:, NH
1046:diol
1024:, a
965:and
729:O +
209:UNII
2302:No
2299:Md
2296:Fm
2293:Es
2290:Cf
2287:Bk
2284:Cm
2281:Am
2278:Pu
2275:Np
2269:Pa
2266:Th
2263:Ac
2260:**
2255:Yb
2252:Tm
2249:Er
2246:Ho
2243:Dy
2240:Tb
2237:Gd
2234:Eu
2231:Sm
2228:Pm
2225:Nd
2222:Pr
2219:Ce
2216:La
2201:Og
2198:Ts
2195:Lv
2192:Mc
2189:Fl
2186:Nh
2183:Cn
2180:Rg
2177:Ds
2174:Mt
2171:Hs
2168:Bh
2165:Sg
2162:Db
2159:Rf
2156:Lr
2153:**
2150:Ra
2147:Fr
2142:Rn
2139:At
2136:Po
2133:Bi
2130:Pb
2127:Tl
2124:Hg
2121:Au
2118:Pt
2115:Ir
2112:Os
2109:Re
2103:Ta
2100:Hf
2097:Lu
2069:Xe
2063:Te
2060:Sb
2057:Sn
2054:In
2031:Pd
2028:Rh
2025:Ru
2022:Tc
2019:Mo
2016:Nb
2013:Zr
1983:Kr
1980:Br
1977:Se
1974:As
1971:Ge
1968:Ga
1965:Zn
1962:Cu
1959:Ni
1956:Co
1953:Fe
1946:Cr
1940:Ti
1937:Sc
1910:Ar
1907:Cl
1898:Si
1861:Ne
1848:MnO
1834:Be
1821:He
1795:ion
1732:doi
1695:PMC
1687:doi
1656:doi
1652:115
1581:doi
1569:108
1237:doi
1076:MnO
1036:to
993:+ O
985:MnO
981:→ K
877:).
867:MnO
841:MnO
825:of
766:or
758:or
731:MnO
717:MnO
700:MnO
688:MnO
673:MnO
663:or
641:MnO
602:+ 9
589:+ 4
568:MnO
566:+ 8
554:→ 6
524:+ 3
514:MnO
456:of
442:MnO
438:ion
425:ɜːr
315:MnO
235:EPA
188:CID
34:MnO
18:MnO
2321::
2272:U
2213:*
2106:W
2094:*
2066:I
2010:Y
1943:V
1904:S
1901:P
1858:F
1855:O
1844:NH
1840:C
1837:B
1746:.
1738:.
1728:15
1726:.
1703:.
1693:.
1683:14
1681:.
1677:.
1650:,
1628:;
1620:}}
1616:{{
1608:.
1602:.
1579:.
1567:.
1546:;
1538:}}
1534:{{
1526:.
1520:.
1499:;
1493:.
1487:.
1466:;
1460:.
1454:.
1433:;
1427:.
1421:.
1400:;
1394:.
1388:.
1367:;
1355:}}
1351:{{
1343:.
1337:.
1233:65
1231:.
1210:.
1032:,
801:SO
770::
639:+
604:Cl
578:+
556:CO
544:Cl
496:Cl
440:,
428:-/
413:eɪ
389:ər
375:A
37:3−
21:2−
2089:2
2087:)
2085:4
2077:4
2049:2
2047:)
2045:4
2037:4
2003:2
2001:)
1999:4
1991:4
1930:2
1928:)
1926:4
1918:4
1893:3
1891:)
1889:4
1881:2
1879:)
1877:4
1869:4
1850:4
1846:4
1829:4
1810:4
1784:e
1777:t
1770:v
1754:.
1734::
1711:.
1689::
1663:.
1658::
1635:.
1626:)
1610:2
1587:.
1583::
1575::
1553:.
1544:)
1528:7
1506:.
1495:2
1473:.
1462:2
1440:.
1429:4
1407:.
1396:5
1374:.
1365:)
1345:6
1322:.
1307:.
1282:.
1257:.
1243:.
1239::
1140:4
1131:4
1122:4
1113:4
1104:2
1102:)
1100:4
1091:2
1089:)
1087:4
1078:4
1074:4
995:2
991:2
987:4
983:2
979:4
929:d
925:p
872:4
855:2
851:4
847:2
843:4
839:2
817:O
815:2
811:4
807:4
803:4
799:2
795:2
791:4
786:O
784:2
780:4
776:2
743:2
736:4
727:2
719:2
705:4
693:4
678:4
655:O
653:2
646:4
630:H
600:O
596:2
591:H
580:H
573:2
561:2
549:3
540:3
535:H
531:2
526:C
519:4
512:8
506:2
502:2
498:3
494:3
492:H
490:2
447:4
422:p
419:,
416:t
410:n
407:ə
404:ɡ
401:ŋ
398:æ
395:m
392:ˈ
386:p
383:/
379:(
320:4
237:)
233:(
124:)
48:.
40:4
24:4
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.