243:
139:
20:
1208:, analogous to the platinized silver of Mr. Smee. Those who are acquainted with the ingenious device of that gentleman, are aware that the characteristic of his arrangement is, that the negative plate, where hydrogen is released, shall part with this hydrogen very readily. Under ordinary circumstances, the hydrogen adheres very much to the plates of an acid battery, and throws a considerable portion of the plates out of the action, by its presence on their surfaces. To remedy this, he has, as he terms it, "platinized" the surfaces.
1604:
1592:
408:
302:
Goodisman excludes this reaction as being inconsistent with the experiments, and notes that the correct chemistry, which involves the evolution of hydrogen at the copper electrode but also can use silver instead of copper, has been known for many years. Most of the detailed predictions of the model apply to the battery's voltage that is measured directly by a meter
125:
buzzers, and small digital clocks. With the zinc/copper electrodes, at least two lemon cells were needed for any of these devices. Substituting a magnesium electrode for the zinc electrode makes a cell with a larger voltage (1.5−1.6 V), and a single magnesium/copper cell will power some devices. Note that
326:
the acidity of the electrolyte, as measured by its pH; decreasing acidity (and increasing pH) causes the voltage to fall. This effect is also predicted by the Nernst equation; the particular acid that was used (citric, hydrochloric, sulfuric, etc.) does not affect the voltage except through the pH value.
394:. Apparently amalgamated zinc was less prone to degradation by an acidic solution than is pure zinc. Amalgamated zinc and plain zinc electrodes give essentially the same voltage when the zinc is pure. With the imperfectly refined zinc in 19th century laboratories they typically gave different voltages.
1515:
Description of an acid cell with zinc & copper electrodes, including a JAVA-based animation. The animation shows zinc dissolving into the electrolyte, electrons flowing from the zinc to the copper electrodes, and little hydrogen bubbles coming off the copper electrode. The animation also suggests
255:
Most textbooks present the following model for the chemical reactions of a lemon battery. When the cell is providing an electric current through an external circuit, the metallic zinc at the surface of the zinc electrode is dissolving into the solution. Zinc atoms dissolve into the liquid electrolyte
212:
Zinc and copper electrodes are reasonably safe and easy to obtain. Other metals such as lead, iron, magnesium, etc., can be studied as well; they yield different voltages than the zinc/copper pair. In particular, magnesium/copper electrodes can generate voltages as large as 1.6 V in lemon cells. This
124:
to power an LED (see illustration) or other devices. The series connection increases the voltage available to devices. Swartling and Morgan have published a list of low-voltage devices along with the corresponding number of lemon cells that were needed to power them; they included LEDs, piezoelectric
23:
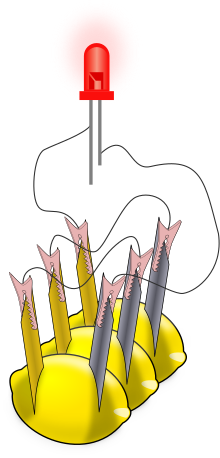
Diagram showing three lemon cells wired together so that they energize the red light-emitting diode (LED) at the top. Each individual lemon has a zinc electrode and a copper electrode inserted into it; the zinc is colored gray in the diagram. The slender lines drawn between the electrodes and the LED
325:
When the battery is hooked up to an external circuit and a significant electric current is flowing, the zinc electrode loses mass, as predicted by the zinc oxidation reaction above. Similarly, hydrogen gas evolves as bubbles from the copper electrode. Finally, the voltage from the cell depended upon
221:
For the youngest pupils, about ages 5–9, the educational goal is utilitarian: batteries are devices that can power other devices, as long as they are connected by a conductive material. Batteries are components in electrical circuits; hooking a single wire between a battery and a light bulb will not
129:
light bulbs from flashlights are not used because the lemon battery is not designed to produce enough electric current to light them. Such a battery typically produces 0.001 A (1 mA) of current at a potential difference of 0.7 V; these values are multiplied together to determine the overall power of
1181:
The first real improvement over the plain zinc-copper in acid cell was due to Dr. Alfred Smee, who noticed that the hydrogen gas liberated at the negative plate was evolved from it much more readily, hence polarization took place much less rapidly if the surface of this plate were roughened instead
301:
This model of the chemical reactions makes several predictions that were examined in experiments published by Jerry
Goodisman in 2001. Goodisman notes that numerous recent authors propose chemical reactions for the lemon battery that involve dissolution of the copper electrode into the electrolyte.
866:
These authors note that hydrogen evolves from the zinc electrode. As described somewhat later by
Goodisman, this effect is unrelated to the evolution of hydrogen that occurs when the cell is providing electric current to an external circuit; the hydrogen associated with these currents evolves from
1247:
In 1840 Smee invented a battery which made electrotyping possible commercially. ... Perhaps one of the greatest forward steps in connection with electrotyping was made when the plating dynamo was invented. The first adoption of a dynamo, in place of the Smee type of battery, was by Leslie, of New
292:
This reaction is called reduction. The electrons used in the copper to form the molecules of hydrogen are transferred from the zinc through an external wire connecting between the copper and the zinc. The hydrogen molecules formed on the surface of the copper by the reduction reaction ultimately
225:
For children in the age range 10−13, batteries are used to illustrate the connection between chemistry and electricity as well as to deepen the circuit concept for electricity. The fact that different chemical elements such as copper and zinc are used can be placed in the larger context that the
1153:
Formerly, a galvanic battery was a stupendous, and an expensive, machine occupying a large space and costing a considerable sum to keep it in its short-lived action. Now, a far more powerful instrument may be made in a snuff box and carried in the pocket. These remarks are forced upon us by the
329:
The Nernst equation prediction failed for strongly acid electrolytes (pH < 3.4), when the zinc electrode dissolves into the electrolyte even when the battery is not providing any current to a circuit. The two oxidation-reduction reactions listed above only occur when electrical charge can be
246:
Cross-section of a copper/zinc cell with a sulfuric acid electrolyte. The drawing illustrates the atomic model for the chemical reactions; lemon cells have essentially the same model. Zinc atoms enter the electrolyte as ions missing two electrons (Zn). Two negatively charged electrons from the
229:
For older pupils and for college students, batteries serve to illustrate the principles of oxidation-reduction reactions. Students can discover that two identical electrodes yield no voltage and that different pairs of metals (beyond copper and zinc) yield different voltages. The voltages and
177:
lighting have been proposed for use in poor countries or by off-grid populations. International research begun in 2010 showed that boiling potatoes for eight minutes improves their electrical output, as does placing slices of potatoes between multiple copper and zinc plates. Boiled and chopped
378:
coating instead of a copper electrode. Hydrogen gas clinging to the surface of a silver or copper electrode reduces the electric current that can be drawn from a cell; the phenomenon is called "polarization". The roughened, "platinized" surface speeds up the bubbling of the hydrogen gas, and
146:
machine screw. The copper electrode is a wire. Note the labels − and + marked on the potato indicating that the copper electrode is the positive terminal of the battery. A short screw and nut connect the electrodes to the copper wires that have black and red insulating plastic coatings.
330:
transported through the external circuit. The additional, open-circuit reaction can be observed by the formation of bubbles at the zinc electrode under open-circuit. This effect ultimately limited the voltage of the cells to 1.0 V near room temperature at the highest levels of acidity.
493:
Season 6 episode "The
Blackout in the Blizzard", Angela and the other "squints" build a massive potato battery array in an attempt to power a cell phone. Illustrating the pathetically low output of such a system, it is successful for only a handful of seconds while using dozens of
233:
The current that is output by the battery through a meter will depend on the size of the electrodes, how far the electrodes are inserted into the fruit, and how close to each other the electrodes are placed; the voltage is fairly independent of these details of the electrodes.
1309:
Some extremely important commissions were made in electrotypes, such as the "bronzes" that adorn the Opera, Paris, and the 320 cm high statue of Prince Albert and four accompanying figures, erected behind the Albert Hall in London as a memorial to the Great
Exhibition of
781:
This webpage describes experiments starting with a single lemon cell that is studied with a multimeter, and then leads to a lemon battery capable of lighting an LED. Hila
Science Camp has also posted a video showing how to build the battery and light an LED; see
814:
This webpage contains instructions for elementary school teachers. The project uses the voltmeter to show that the battery is working. A key element is that several pairs of electrodes are used (iron/zinc, iron/copper, as well as zinc/copper) to yield different
379:
increases the current from the cell. Unlike the zinc electrode, the copper or platinized silver electrodes are not consumed by using the battery, and the details of this electrode do not affect the cell's voltage. The Smee cell was convenient for
119:
from the voltaic cell; a typical voltage is 0.9 V with lemons. Currents are more variable, but range up to about 1 mA (the larger the electrode surfaces, the bigger the current). For a more visible effect, lemon cells can be connected in
213:
voltage is larger than obtainable using zinc/copper cells. It is comparable to that of standard household batteries (1.5 V), which is useful in powering devices with a single cell instead of using cells in series.
584:
Season 2 episode 10, "The
Growing Good of the World," Anne and her classmates use potato batteries to prove to their town how much they've learned from their new teacher's unconventional teaching methods.
357:
reduction of zinc with carbon, which requires an energy input. The energy produced in the lemon battery comes from reversing this reaction, recovering some of the energy input during the zinc production.
151:
Many fruits and liquids can be used for the acidic electrolyte. Fruit is convenient, because it provides both the electrolyte and a simple way to support the electrodes. The acid involved in
1345:
The singular property possessed by amalgamated zinc of not being attacked by sulphuric acid diluted with water is due to the adhesion of hydrogen on the plate in the acid solution
247:
dissolved zinc atom are left in the zinc metal. Two of the dissolved protons (H) in the acidic electrolyte combine with each other and two electrons to form molecular hydrogen H
322:) did not affect the voltage. This result is consistent with the fact that copper atoms from the electrode are not involved in the chemical reaction model for the cell.
251:, which bubbles off of the copper electrode. The electrons lost from the copper are made up by moving two electrons from the zinc through the external wire.
87:. There are many variations of the lemon cell that use different fruits (or liquids) as electrolytes and metals other than zinc and copper as electrodes.
338:
The energy comes from the chemical change in the zinc when it dissolves into the acid. The energy does not come from the lemon or potato. The zinc is
342:
inside the lemon, exchanging some of its electrons with the acid in order to reach a lower energy state, and the energy released provides the power.
318:
for the model. The Nernst equation essentially says how much the voltage drops as more zinc sulfate is added. The addition of copper sulfate (CuSO
973:
Sauerkraut is quite acidic due to the lactic acid produced during fermentation. The sauerkraut clock powers a digital thermometer in this video.
107:) nails and screws. Commercial "potato clock" science kits include electrodes and a low-voltage digital clock. After one cell is assembled, a
1182:
of being quite smooth; and the means he found most efficient was that of coating the silver sheet or sheets with finely divided platinum ...
1091:
549:
Season 7 episode 8, "Power Down", Abby Sciuto uses lemons as a power source for her stereo when she runs out of batteries during a blackout.
1415:
370:
electrolyte were widely used in the printing industry. While copper electrodes like those in lemon batteries were sometimes used, in 1840
1100:
A quantitative study of both the voltages and currents produced by fruit batteries; part of a larger project including "penny batteries".
1172:
Galvanic batteries, their theory, construction and use, comprising primary, single and double fluid cells, secondary and gas batteries
660:
Volta used silver, not copper, in his first cells; the chemical reactions involved in zinc/copper and zinc/silver cells are the same.
278:
ions (H) from the electrolyte combine with two electrons at the copper electrode's surface and form an uncharged hydrogen molecule (H
1503:
1389:
647:
1458:
877:
383:, which produced copper plates for letterpress printing of newspapers and books, and also statues and other metallic objects.
1302:
1065:
709:
962:
1020:
Schmidt, Hans-Jürgen; Marohn, Annette; Harrison, Allan G. (2007). "Factors that prevent learning in electrochemistry".
1533:
1371:
1124:
1073:
906:
679:
447:
209:
program). The sauerkraut had been canned, and became the electrolyte while the can itself was one of the electrodes.
1629:
1080:
Guide to lemon battery experiments for science teachers, including both fabrication notes and educational outcomes.
802:
1278:
The Smee cell is the cell most commonly employed because of its extreme simplicity of construction and management.
429:
425:
1624:
1204:
Of the application of this cell a very important modification can be arranged, by converting it into an ACID
948:
52:
and connected by wires. Power generated by reaction of the metals is used to power a small device such as a
923:
554:
528:
121:
1154:
astonishing platinum batteries of Mr. Grove, and the chemico-mechanical batteries invented by Mr. Smee ...
731:
1251:
Primer for apprentices in the printing industry. Good short introduction to the history of electrotyping.
95:
There are numerous sets of instructions for making lemon batteries and for obtaining components such as
1582:
1574:
987:
789:
619:
1559:. Cool and fun animated video explaining in great detail how a lemon battery works from the inside.
1332:
1195:
1359:
1176:
1095:
769:
575:, the devices used to step from one universe to another appear to be powered by potato batteries.
570:
566:
480:
Season 6 episode "The Proton
Resurgence", Leonard and Sheldon's childhood hero Professor Proton (
418:
1521:
1265:
1221:
896:
699:
1292:
1140:
1114:
1057:
594:
199:
1516:
that a single cell can light an LED, which is not possible for LEDs that emit visible light.
1356:
The standard electrode potential is 0.76 V for both pure zinc and for amalgamated zinc. See
1238:
173:
and work well; they are the basis for commercial "potato clock" kits. Potato batteries with
1029:
843:
743:
604:
558:, Season 3, episode 2, "Easy Money", Magnum uses a lemon battery to charge a walkie-talkie.
508:
303:
96:
53:
33:
8:
1170:
512:
476:
1326:
1033:
847:
747:
306:(nothing else is connected to the battery). When the electrolyte was modified by adding
1562:
655:
614:
536:
tried taking over the world using potato batteries, only for her plans to be ruined by
498:
366:
From 1840 to the late 19th century, large, voltaic cells using a zinc electrode and a
1529:
1367:
1336:
1298:
1120:
1069:
902:
881:
831:
705:
675:
489:
391:
354:
72:
1608:
1441:
1037:
1002:
851:
751:
599:
545:
387:
179:
116:
64:
471:
is embedded into a potato-battery-run computer for a significant part of the game.
1281:
A detailed discussion of the construction and maintenance of Smee cells, c. 1874.
562:
533:
315:
230:
currents from series and parallel combinations of the batteries can be examined.
170:
185:
Instead of fruit, liquids in various containers can be used. Household vinegar (
1484:
580:
537:
346:
142:
Potato battery with zinc (left) and copper electrodes. The zinc electrode is a
71:(salt water) instead of lemon juice. The lemon battery illustrates the type of
1568:
1556:
968:
783:
672:
The
Extraordinary Chemistry of Ordinary Things, with Late Nite Labs: Edition 4
226:
elements do not disappear or break down when they undergo chemical reactions.
1618:
1596:
1056:
Sorey, Timothy; Hunt, Vanessa; Balandova, Evguenia; Palmquist, Bruce (2012).
624:
609:
380:
367:
143:
1200:
The
Chemist; Or, Reporter of Chemical Discoveries and Improvements, Volume 1
390:
instead of pure zinc; the surface of amalgamated zinc has been treated with
19:
1340:
519:
in 23 AD in order to get lemons to build a lemon battery to power up their
350:
307:
126:
60:
1196:"Review: Proceedings of the London Electrical Society, 1841-1842 Session"
520:
481:
371:
205:
194:
186:
156:
84:
1006:
432: in this section. Unsourced material may be challenged and removed.
190:
108:
104:
100:
41:
1041:
855:
755:
374:
invented a refined version of this cell that used silver with a rough
138:
503:
80:
407:
1489:. Season 2. Episode 10. November 18, 2018. 36 minutes in. Netflix.
463:
375:
339:
275:
261:
484:) attempts to show the group a potato battery, which amazes Penny.
242:
1591:
274:. While zinc is entering the electrolyte, two positively charged
112:
314:), the voltage from the cell was reduced as predicted using the
1459:"The Long Earth by Terry Pratchett and Stephen Baxter – review"
986:
Abraham, Ann; Palencsar, Attila; Scherson, Daniel (Fall 2006).
468:
166:
152:
79:) that occurs in batteries. The zinc and copper are called the
45:
36:
often made for the purpose of education. Typically, a piece of
1446:. Season 7. Episode 8. November 17, 2009. 28 minutes in. CBS.
182:(stem) is also suitable, according to Sri Lankan researchers.
516:
271:
76:
68:
49:
1294:
Copper and bronze in art: corrosion, colorants, conservation
1055:
257:
197:) was featured in one episode of the US television program
37:
1223:
A physical treatise on electricity and magnetism, Volume 1
1062:
Fuel for
Thought: Building Energy Awareness in Grades 9-12
1565:. The orange battery powers an inexpensive digital watch.
174:
16:
Simple battery made with a lemon for educational purposes
988:"Electrochemistry for K-12: The Potato Clock and Beyond"
985:
1245:. Chicago: United Typothetae of America. pp. 2–4.
160:
1390:"Portal 2 Science Kit Has Talking, Evil Potato GLaDOS"
1058:"Juan's Dilemma: A New Twist on the Old Lemon Battery"
1580:
1019:
924:"Potato power: the spuds that could light the world"
832:"Lemon Cells Revisited—The Lemon-Powered Calculator"
805:. Burlington, Iowa: How Things Work Science Projects
1328:
The Voltaic Cell: its Construction and its Capacity
894:
1577:. Three potato cells in series power a calculator.
949:"DIY vinegar battery lights LEDs for several days"
159:. The acidity, which is indicated by the measured
654:. Case Western Reserve University. Archived from
1616:
1237:Hatch, Harris B.; Stewart, Alexander A. (1918).
1112:
830:Swartling, Daniel J.; Morgan, Charlotte (1998).
1364:Handbook of Chemistry and Physics: 93rd Edition
829:
155:fruits (lemons, oranges, grapefruits, etc.) is
83:, and the juice inside the lemon is called the
1270:The Telegraphic Journal and Electrical Review
1506:. US National High Magnetic Field Laboratory
1236:
526:In the sixth episode of the final season of
1416:"TV Review: RED DWARF X Episode 3 'Lemons'"
825:
823:
821:
725:
723:
721:
1504:"Maglab - Simple Electrical Cell Tutorial"
1366:. Chemical Rubber Company. pp. 5–80.
1259:
1257:
1164:
1162:
1145:The Magazine of Science and School of Arts
1119:. Tata McGraw-Hill Education. p. 50.
981:
979:
729:
704:. Panpac Education Pte Ltd. p. 236.
701:Chemistry Expression: An Inquiry Approach
448:Learn how and when to remove this message
345:In current practice, zinc is produced by
90:
1194:Watt, Charles; Watt, John, eds. (1840).
1193:
1133:
1108:
1106:
818:
718:
641:
639:
241:
137:
18:
1519:
1413:
1357:
1320:
1318:
1263:
1254:
1226:. D. Appleton and Company. p. 207.
1168:
1159:
1022:Journal of Research in Science Teaching
1013:
976:
967:. The Discovery Channel. Archived from
946:
940:
1617:
1219:
1213:
1116:Introduction to Electrical Engineering
1113:Naidu, M. S.; Kamakshiaih, S. (1995).
895:Lisinska, G.; Leszczynski, W. (1989).
772:. Pembroke, Ontario: Hila Science Camp
669:
645:
48:(such as a penny) are inserted into a
1387:
1290:
1284:
1230:
1103:
1049:
995:The Electrochemical Society Interface
693:
691:
663:
636:
397:
296:
1456:
1388:Noble, Mckinley (19 December 2011).
1324:
1315:
1187:
511:), the crew travel 4,000 miles from
430:adding citations to reliable sources
401:
216:
59:The lemon battery is similar to the
1522:"Does a Lemon Battery Really Work?"
1414:Keeling, Robert (19 October 2012).
1350:
1220:Gordon, James Edward Henry (1880).
762:
697:
260:(Zn), leaving 2 negatively charged
13:
1496:
1297:. Getty Publications. p. 22.
1089:
1083:
795:
688:
14:
1641:
1550:
1483:"The Growing Good of the World".
947:Heeling, Harmjan (May 12, 2012).
921:
1602:
1590:
1528:. Scholastic. pp. 104–108.
406:
333:
1476:
1450:
1433:
1407:
1381:
1362:. In Haynes, William M. (ed.).
1264:Sprague, J. T. (July 1, 1874).
1239:"History of Electrotype Making"
1046:Full text by subscription only.
955:
915:
646:Decker, Franco (January 2005).
417:needs additional citations for
1457:Adam, Roberts (20 June 2012).
1266:"Electro-deposition of Metals"
1243:Electrotyping and stereotyping
1175:. Whittaker & Co. p.
1169:Bottone, Selimo Romeo (1902).
888:
870:
1:
1526:Mythbusters Science Fair Book
1141:"Mr. Smee's Galvanic Battery"
898:Potato Science and Technology
836:Journal of Chemical Education
736:Journal of Chemical Education
732:"Observations on Lemon Cells"
652:Electrochemistry Encyclopedia
630:
293:bubble away as hydrogen gas.
133:
99:, (LEDs), electrical meters (
964:Head Rush - Sauerkraut Clock
529:Mystery Science Theater 3000
501:" of the television program
361:
237:
7:
588:
111:can be used to measure the
10:
1646:
1520:Margles, Samantha (2011).
928:BBC - Future - Technology
901:. Springer. p. 286.
730:Goodisman, Jerry (2001).
674:. John Wiley & Sons.
264:(e) behind in the metal:
1360:"Electrochemical Series"
1291:Scott, David A. (2002).
1060:. In Metz, Steve (ed.).
698:Oon, Hock Leong (2007).
670:Snyder, Carl H. (2004).
270:This reaction is called
256:as electrically charged
163:, varies substantially.
61:first electrical battery
1630:Biodegradable materials
1325:Park, Benjamin (1893).
1202:. London: R. Hastings.
1068:Press. pp. 91–98.
803:"Lemon Battery Project"
509:tenth series (Series X)
1358:Vanýsek, Petr (2012).
1151:: 22. April 18, 1840.
785:Create a Lemon Battery
648:"Volta and the 'Pile'"
252:
148:
91:Use in school projects
25:
1092:"Fruit/veg batteries"
867:the copper electrode.
595:List of battery types
245:
141:
97:light-emitting diodes
44:nail) and a piece of
22:
1625:Disposable batteries
1570:Potato battery video
1563:Orange battery video
1546:Online preview only.
1331:. J. Wiley. p.
605:Electrochemical cell
426:improve this article
203:(an offshoot of the
130:0.0007 W (0.7 mW).
103:), and zinc-coated (
63:invented in 1800 by
54:light-emitting diode
24:represent the wires.
1557:Lemon Battery video
1034:2007JRScT..44..258S
848:1998JChEd..75..181S
748:2001JChEd..78..516G
532:, the main villain
523:'s returner remote.
477:The Big Bang Theory
386:The Smee cell used
77:oxidation-reduction
1090:Du, James (2011).
1007:10.1149/2.F09063IF
884:on April 15, 2009.
615:Galvanic corrosion
461:In the video game
398:In popular culture
297:Experiment results
253:
149:
26:
1304:978-0-89236-638-5
1042:10.1002/tea.20118
922:Kalan, Jonathan.
856:10.1021/ed075p181
756:10.1021/ed078p516
711:978-981-271-162-5
467:, the antagonist
458:
457:
450:
355:pyrometallurgical
217:Learning outcomes
73:chemical reaction
40:metal (such as a
1637:
1607:
1606:
1605:
1595:
1594:
1586:
1571:
1545:
1543:
1542:
1514:
1512:
1511:
1491:
1490:
1480:
1474:
1473:
1471:
1469:
1454:
1448:
1447:
1437:
1431:
1430:
1428:
1426:
1411:
1405:
1404:
1402:
1400:
1385:
1379:
1377:
1354:
1348:
1347:
1322:
1313:
1312:
1288:
1282:
1280:
1261:
1252:
1250:
1234:
1228:
1227:
1217:
1211:
1210:
1191:
1185:
1184:
1166:
1157:
1156:
1137:
1131:
1130:
1110:
1101:
1099:
1094:. Archived from
1087:
1081:
1079:
1053:
1047:
1045:
1017:
1011:
1010:
992:
983:
974:
972:
959:
953:
952:
944:
938:
937:
935:
934:
919:
913:
912:
892:
886:
885:
880:. Archived from
878:"Potato Battery"
874:
868:
865:
863:
862:
827:
816:
813:
811:
810:
799:
793:
786:
780:
778:
777:
766:
760:
759:
727:
716:
715:
695:
686:
685:
667:
661:
659:
643:
600:Alessandro Volta
497:In the episode "
453:
446:
442:
439:
433:
410:
402:
388:amalgamated zinc
222:power the bulb.
117:electric current
65:Alessandro Volta
1645:
1644:
1640:
1639:
1638:
1636:
1635:
1634:
1615:
1614:
1613:
1603:
1601:
1589:
1581:
1569:
1553:
1540:
1538:
1536:
1509:
1507:
1502:
1499:
1497:Further reading
1494:
1482:
1481:
1477:
1467:
1465:
1455:
1451:
1439:
1438:
1434:
1424:
1422:
1412:
1408:
1398:
1396:
1386:
1382:
1374:
1355:
1351:
1323:
1316:
1305:
1289:
1285:
1276:(34): 237–239.
1262:
1255:
1235:
1231:
1218:
1214:
1192:
1188:
1167:
1160:
1139:
1138:
1134:
1127:
1111:
1104:
1088:
1084:
1076:
1054:
1050:
1018:
1014:
990:
984:
977:
961:
960:
956:
945:
941:
932:
930:
920:
916:
909:
893:
889:
876:
875:
871:
860:
858:
828:
819:
808:
806:
801:
800:
796:
784:
775:
773:
770:"Lemon Battery"
768:
767:
763:
728:
719:
712:
696:
689:
682:
668:
664:
644:
637:
633:
591:
563:Terry Pratchett
534:Pearl Forrester
454:
443:
437:
434:
423:
411:
400:
364:
336:
321:
316:Nernst equation
313:
304:at open circuit
299:
290:
288:
281:
268:
250:
240:
219:
171:phosphoric acid
136:
93:
17:
12:
11:
5:
1643:
1633:
1632:
1627:
1612:
1611:
1599:
1579:
1578:
1566:
1560:
1552:
1551:External links
1549:
1548:
1547:
1534:
1517:
1498:
1495:
1493:
1492:
1486:Anne with an E
1475:
1449:
1440:"Power Down".
1432:
1406:
1380:
1372:
1349:
1314:
1303:
1283:
1253:
1248:York, in 1872.
1229:
1212:
1186:
1158:
1132:
1125:
1102:
1098:on 2019-10-30.
1082:
1074:
1048:
1028:(2): 258–283.
1012:
975:
971:on 2011-06-10.
954:
939:
914:
907:
887:
869:
842:(2): 181–182.
817:
794:
761:
742:(4): 516–518.
717:
710:
687:
680:
662:
658:on 2012-07-16.
634:
632:
629:
628:
627:
622:
617:
612:
607:
602:
597:
590:
587:
586:
585:
581:Anne with an E
576:
572:The Long Earth
567:Stephen Baxter
559:
550:
541:
538:Professor Bobo
524:
495:
485:
472:
456:
455:
414:
412:
405:
399:
396:
363:
360:
347:electrowinning
335:
332:
319:
311:
298:
295:
286:
284:
279:
267:Zn → Zn + 2e .
266:
248:
239:
236:
218:
215:
189:) works well.
135:
132:
92:
89:
15:
9:
6:
4:
3:
2:
1642:
1631:
1628:
1626:
1623:
1622:
1620:
1610:
1600:
1598:
1593:
1588:
1587:
1584:
1576:
1572:
1567:
1564:
1561:
1558:
1555:
1554:
1537:
1535:9780545237451
1531:
1527:
1523:
1518:
1505:
1501:
1500:
1488:
1487:
1479:
1464:
1460:
1453:
1445:
1444:
1436:
1421:
1417:
1410:
1395:
1391:
1384:
1375:
1373:9781439880494
1369:
1365:
1361:
1353:
1346:
1342:
1338:
1334:
1330:
1329:
1321:
1319:
1311:
1306:
1300:
1296:
1295:
1287:
1279:
1275:
1271:
1267:
1260:
1258:
1249:
1244:
1240:
1233:
1225:
1224:
1216:
1209:
1207:
1201:
1197:
1190:
1183:
1178:
1174:
1173:
1165:
1163:
1155:
1150:
1146:
1142:
1136:
1128:
1126:9780074622926
1122:
1118:
1117:
1109:
1107:
1097:
1093:
1086:
1077:
1075:9781936137206
1071:
1067:
1063:
1059:
1052:
1043:
1039:
1035:
1031:
1027:
1023:
1016:
1008:
1004:
1000:
996:
989:
982:
980:
970:
966:
965:
958:
950:
943:
929:
925:
918:
910:
908:9781851663071
904:
900:
899:
891:
883:
879:
873:
857:
853:
849:
845:
841:
837:
833:
826:
824:
822:
804:
798:
791:
787:
771:
765:
757:
753:
749:
745:
741:
737:
733:
726:
724:
722:
713:
707:
703:
702:
694:
692:
683:
681:9780471588399
677:
673:
666:
657:
653:
649:
642:
640:
635:
626:
625:Penny battery
623:
621:
618:
616:
613:
611:
610:Galvanic cell
608:
606:
603:
601:
598:
596:
593:
592:
583:
582:
577:
574:
573:
568:
564:
560:
557:
556:
551:
548:
547:
542:
539:
535:
531:
530:
525:
522:
518:
514:
510:
506:
505:
500:
496:
492:
491:
486:
483:
479:
478:
473:
470:
466:
465:
460:
459:
452:
449:
441:
431:
427:
421:
420:
415:This section
413:
409:
404:
403:
395:
393:
389:
384:
382:
381:electrotyping
377:
373:
369:
368:sulfuric acid
359:
356:
352:
348:
343:
341:
334:Energy source
331:
327:
323:
317:
309:
305:
294:
283:
277:
273:
265:
263:
259:
244:
235:
231:
227:
223:
214:
210:
208:
207:
202:
201:
196:
192:
188:
183:
181:
180:plantain pith
176:
172:
168:
164:
162:
158:
154:
145:
140:
131:
128:
123:
118:
114:
110:
106:
102:
98:
88:
86:
82:
78:
74:
70:
66:
62:
57:
55:
51:
47:
43:
39:
35:
31:
30:lemon battery
21:
1539:. Retrieved
1525:
1508:. Retrieved
1485:
1478:
1466:. Retrieved
1463:The Guardian
1462:
1452:
1442:
1435:
1423:. Retrieved
1419:
1409:
1397:. Retrieved
1393:
1383:
1363:
1352:
1344:
1327:
1308:
1293:
1286:
1277:
1273:
1269:
1246:
1242:
1232:
1222:
1215:
1205:
1203:
1199:
1189:
1180:
1171:
1152:
1148:
1144:
1135:
1115:
1096:the original
1085:
1061:
1051:
1025:
1021:
1015:
1001:(3): 43–46.
998:
994:
969:the original
963:
957:
942:
931:. Retrieved
927:
917:
897:
890:
882:the original
872:
859:. Retrieved
839:
835:
807:. Retrieved
797:
774:. Retrieved
764:
739:
735:
700:
671:
665:
656:the original
651:
620:Lasagna cell
579:
571:
553:
544:
527:
521:time machine
502:
488:
475:
462:
444:
438:January 2015
435:
424:Please help
419:verification
416:
385:
365:
351:zinc sulfate
344:
337:
328:
324:
308:zinc sulfate
300:
291:
269:
254:
232:
228:
224:
220:
211:
204:
198:
184:
165:
150:
127:incandescent
94:
58:
32:is a simple
29:
27:
555:Magnum P.I.
482:Bob Newhart
372:Alfred Smee
206:MythBusters
195:lactic acid
187:acetic acid
157:citric acid
101:multimeters
85:electrolyte
67:, who used
1619:Categories
1541:2012-10-07
1510:2012-11-30
1468:8 February
1425:30 January
1399:30 January
933:2014-01-24
861:2020-12-22
809:2012-10-11
776:2012-10-02
631:References
285:2H+ 2e → H
191:Sauerkraut
144:galvanized
134:Variations
109:multimeter
105:galvanized
81:electrodes
42:galvanized
1609:Chemistry
1420:Starburst
815:voltages.
504:Red Dwarf
494:potatoes.
362:Smee cell
272:oxidation
262:electrons
238:Chemistry
200:Head Rush
1394:techhive
589:See also
464:Portal 2
376:platinum
340:oxidized
276:hydrogen
167:Potatoes
1583:Portals
1575:YouTube
1341:7399515
1206:battery
1030:Bibcode
844:Bibcode
790:YouTube
744:Bibcode
513:Britain
487:In the
392:mercury
115:or the
113:voltage
56:(LED).
34:battery
1597:Energy
1532:
1370:
1339:
1301:
1123:
1072:
905:
708:
678:
499:Lemons
469:GLaDOS
153:citrus
122:series
46:copper
1310:1851.
991:(PDF)
517:India
490:Bones
310:(ZnSO
169:have
69:brine
50:lemon
1530:ISBN
1470:2017
1443:NCIS
1427:2015
1401:2015
1368:ISBN
1337:OCLC
1299:ISBN
1121:ISBN
1070:ISBN
1066:NSTA
903:ISBN
706:ISBN
676:ISBN
565:and
546:NCIS
258:ions
38:zinc
1573:on
1333:347
1038:doi
1003:doi
852:doi
788:on
752:doi
578:In
569:'s
561:In
552:In
543:In
515:to
474:In
428:by
353:or
349:of
282:):
175:LED
1621::
1524:.
1461:.
1418:.
1392:.
1343:.
1335:.
1317:^
1307:.
1274:II
1272:.
1268:.
1256:^
1241:.
1198:.
1179:.
1177:88
1161:^
1149:II
1147:.
1143:.
1105:^
1064:.
1036:.
1026:44
1024:.
999:15
997:.
993:.
978:^
926:.
850:.
840:75
838:.
834:.
820:^
750:.
740:78
738:.
734:.
720:^
690:^
650:.
638:^
161:pH
28:A
1585::
1544:.
1513:.
1472:.
1429:.
1403:.
1378:.
1376:.
1129:.
1078:.
1044:.
1040::
1032::
1009:.
1005::
951:.
936:.
911:.
864:.
854::
846::
812:.
792:.
779:.
758:.
754::
746::
714:.
684:.
540:.
507:(
451:)
445:(
440:)
436:(
422:.
320:4
312:4
289:.
287:2
280:2
249:2
193:(
75:(
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.