887:
592:
40:
31:
2682:
2670:
934:
Roche requested and received approval of
Invirase via the FDA's "Accelerated Approval" program—a process designed to speed drugs to market for the treatment of serious diseases—a decision that was controversial, as AIDS activists disagreed over the benefits of thorough testing versus early access to
930:
by a few months. This new class of antiretrovirals played a critical role in the development of highly active antiretroviral therapy (HAART), which helped significantly lower the risk of death from AIDS-related causes, as seen by a reduction of the annual U.S. HIV-associated death rate, from over
707:
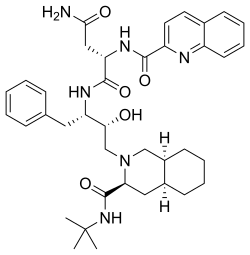
InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1
873:
are enzymes that cleave protein molecules into smaller fragments. HIV protease is vital for both viral replication within the cell and release of mature viral particles from an infected cell. Saquinavir binds to the active site of the viral protease and prevents cleavage of viral polyproteins,
665:
849:
isozyme. Normally, this enzyme metabolizes saquinavir to an inactive form, but with the ritonavir inhibiting this enzyme, the saquinavir blood plasma levels increased considerably. Additionally, ritonavir also inhibits multidrug transporters, although to a much lower extent.
831:
Saquinavir, in the
Invirase formulation, has a low and variable oral bioavailability, when given alone. The Fortovase formulation at the standard dosage delivers approximately eightfold more active drug than Invirase, also at the standard dosage.
1394:
ascribes this to "highly active antiretroviral therapy", without mention of either of these drugs, see the preceding citation. A further citation is needed to make this accurate connection between this drop and the introduction of the protease
2067:
890:
New HIV infections and deaths, before and after the FDA approval of "highly active antiretroviral therapy", of which saquinavir, ritonavir and indinavir were key as the first three protease inhibitors.
2041:
839:. For patients, this has the major benefit that they can take less saquinavir, while maintaining sufficient saquinavir blood plasma levels to efficiently suppress the replication of HIV.
1959:
234:
893:
2061:
2251:
2256:
2202:
1674:
2641:
189:
1838:
835:
In the clinic, it was found that the oral bioavailability of saquinavir in both formulations significantly increases when patients also receive the PI
121:
2208:
2266:
2186:
939:. Roche announced in May 2005 that, given reduced demand, Fortovase would cease being marketed early in 2006, in favor of Invirase boosted with
2271:
2712:
2219:
1620:
70:
2241:
2225:
679:
2594:
2589:
1144:
2236:
1265:
2246:
1462:
1561:
1217:"Effect of omeprazole on the pharmacokinetics of saquinavir-500 mg formulation with ritonavir in healthy male and female volunteers"
2513:
1317:
2157:
1058:
842:
The mechanism behind this welcome observation was not directly known, but later it was determined that ritonavir inhibits the
2717:
2612:
2503:
1105:
764:
Common side effects include nausea, vomiting, diarrhea, and feeling tired. More serious side effects include problems with
1410:
2261:
1598:
1512:
2276:
1185:
815:
The most frequent adverse events with saquinavir in either formulation are mild gastrointestinal symptoms, including
724:
138:
1832:
1613:
1275:
1169:
1138:
866:
777:
699:
2727:
2629:
2286:
2170:
2732:
2296:
2281:
1944:
1932:
1432:
935:
new drugs. It was approved again on
November 7, 1997, as Fortovase, a soft gel capsule reformulated for improved
333:
219:
102:
1365:
1340:
2176:
412:
1606:
1438:
919:
463:
2381:
571:
995:, orally-administered formulation), with trade name Fortovase, which was discontinued worldwide in 2006.
2707:
2660:
512:
1040:
910:
As a result of the new therapies, HIV deaths in the United States fell dramatically within two years.
742:
116:
2453:
2363:
2344:
2231:
2192:
587:
532:
2617:
2213:
843:
175:
52:
2291:
553:
279:
2399:
1215:
Winston A, Back D, Fletcher C, Robinson L, Unsworth J, Tolowinska I, et al. (June 2006).
1159:
1128:
918:. Saquinavir was the sixth antiretroviral and the first protease inhibitor approved by the US
2722:
2702:
2488:
2301:
2181:
2051:
1650:
403:
452:
367:
2371:
2197:
2046:
1804:
1486:
472:
302:
1628:
1587:
1469:
886:
8:
2737:
2306:
2165:
2037:
1955:
1754:
1633:
1569:
1297:
1059:"Invirase- saquinavir mesylate capsule INVIRASE- saquinavir mesylate tablet, film coated"
804:
754:
358:
271:
1233:
1216:
591:
1543:
1246:
961:
853:
Unlike other protease inhibitors, the absorption of saquinavir seems to be improved by
149:
823:, loose stools and abdominal discomfort. Invirase is better tolerated than Fortovase.
2448:
2111:
1535:
1309:
1271:
1238:
1165:
1134:
915:
543:
247:
94:
1547:
1250:
2473:
1527:
1228:
608:
392:
257:
202:
2647:
1101:
1013:
80:
2674:
2549:
2508:
1668:
1531:
985:
936:
875:
765:
289:
265:
1295:
2686:
2623:
1629:
1406:
773:
687:
O=C(N)C(NC(=O)c1nc2c(cc1)cccc2)C(=O)N(Cc3ccccc3)(O)CN5(C(=O)NC(C)(C)C)C4CCCC4C5
2696:
2389:
2139:
1974:
1860:
992:
39:
2443:
2433:
2014:
2004:
1822:
1779:
1774:
1769:
1731:
1711:
1641:
1539:
1513:"Microemulsions for oral administration and their therapeutic applications"
1313:
1242:
1193:
781:
523:
197:
22:
1562:"Roche to discontinue the sale and distribution of Fortovase (saquinavir)"
2569:
2544:
2483:
2468:
2463:
2127:
2094:
2009:
1989:
1784:
1764:
1743:
1690:
1658:
1404:
769:
88:
432:
2574:
2554:
2539:
2534:
2498:
2493:
2407:
2352:
2322:
2134:
2122:
2117:
2089:
2029:
2019:
1994:
1979:
1969:
1918:
1913:
1901:
1875:
1855:
1817:
1727:
1715:
1707:
1407:"Drugs! Drugs! Drugs! An Overview of the Approved Anti-HIV Medications"
943:, owing to the ability of the latter co-formulated drug to inhibit the
854:
795:
Saquinavir is used together with other medications to treat or prevent
776:, and liver problems. It appears to be safe in pregnancy. It is in the
641:
443:
349:--3-hydroxy-1-phenylbutan-2-yl]-2-(quinolin-2-ylformamido)butanediamide
30:
2584:
2564:
2559:
2529:
2458:
2438:
2327:
2084:
1999:
1984:
1907:
1880:
1870:
1865:
1812:
1789:
1702:
981:
940:
927:
923:
836:
816:
800:
750:
378:
315:
74:
2579:
1964:
977:
870:
796:
758:
746:
492:
423:
182:
133:
664:
559:
2478:
2024:
1794:
1296:
Centers for
Disease Control and Prevention (CDC) (3 June 2011).
2428:
2423:
944:
846:
820:
503:
295:
120:
1738:
874:
preventing maturation of the virus. Saquinavir inhibits both
655:
2681:
980:, with trade name Invirase, which requires combination with
576:
2252:
Elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide
1697:
1685:
1214:
483:
2257:
Elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil
1038:
1722:
1637:
745:
used together with other medications to treat or prevent
2203:
Darunavir/cobicistat/emtricitabine/tenofovir alafenamide
826:
787:
Saquinavir was patented in 1988 and first sold in 1995.
1433:"Drug Approval Package: Fortovase/Saquinavir NDA 20828"
914:
Saquinavir was developed by the pharmaceutical company
894:"Protease inhibitors give wings to combination therapy"
2658:
1104:. The American Society of Health-System Pharmacists.
931:50,000 to about 18,000 over a period of two years.
411:
1006:
2694:
2209:Dolutegravir/emtricitabine/tenofovir alafenamide
391:
2267:Emtricitabine/rilpivirine/tenofovir alafenamide
2187:Bictegravir/emtricitabine/tenofovir alafenamide
1405:AIDS Community Research Initiative of America.
1366:"Antiretroviral Drug Discovery and Development"
1157:
1130:Handbook of Assay Development in Drug Discovery
366:
2272:Emtricitabine/rilpivirine/tenofovir disoproxil
1758:(Integrase strand transfer inhibitors (INSTI))
1263:
1039:Roche Products Pty Limited (6 November 2018).
991:a soft-gel capsule formulation of saquinavir (
2220:Dolutegravir/lamivudine/tenofovir alafenamide
1614:
1051:
1047:– via MedAdvisor International Pty Ltd.
2242:Efavirenz/emtricitabine/tenofovir disoproxil
2226:Dolutegravir/lamivudine/tenofovir disoproxil
1510:
1302:Morbidity and Mortality Weekly Report (MMWR)
531:
137:
1298:"HIV Surveillance—United States, 1981-2008"
1291:
1289:
1287:
2237:Doravirine/lamivudine/tenofovir disoproxil
1621:
1607:
1096:
1094:
1092:
1090:
1088:
1086:
1084:
1082:
1080:
1032:
590:
451:
2247:Efavirenz/lamivudine/tenofovir disoproxil
1232:
471:
2514:Zinc finger protein transcription factor
1284:
1264:Dolin R, Masur H, Saag MS, eds. (1999).
885:
1341:"F.D.A. Backs A New Drug To Fight AIDS"
1077:
586:
431:
93:
2695:
1392:Morbidity and Mortality Weekly Report,
1270:. Churchill Livingstone. p. 129.
1164:. John Wiley & Sons. p. 509.
1151:
1120:
976:a hard-gel capsule formulation of the
950:
945:enzyme that metabolizes the AIDS drugs
860:
111:
1602:
1338:
1126:
1108:from the original on 8 September 2015
972:Two formulations have been marketed:
960:As of 2015, it is not available as a
891:
827:Bioavailability and drug interactions
552:
511:
241:
128:
79:
2713:Drugs developed by Hoffmann-La Roche
1594:. U.S. National Library of Medicine.
1413:from the original on 9 November 2013
1320:from the original on 9 November 2013
757:to increase its effect. It is taken
558:
201:
2262:Emtricitabine/tenofovir alafenamide
1234:10.1097/01.aids.0000233573.41597.8a
1147:from the original on 31 March 2016.
1133:. Hoboken: CRC Press. p. 117.
491:
382:
13:
2277:Emtricitabine/tenofovir disoproxil
1511:Gibaud S, Attivi D (August 2012).
14:
2749:
1580:
1041:"Invirase® (Saquinavir mesilate)"
1014:"Saquinavir Use During Pregnancy"
228:
160:
2680:
2668:
2650:. Formerly or rarely used agent.
2287:Lamivudine/nevirapine/zidovudine
2171:Abacavir/dolutegravir/lamivudine
780:class and works by blocking the
625:
619:
38:
29:
2454:Epigallocatechin gallate (EGCG)
2297:Lamivudine/tenofovir disoproxil
2282:Lamivudine/nevirapine/stavudine
1554:
1520:Expert Opinion on Drug Delivery
1504:
1487:"Generic Invirase Availability"
1479:
1463:"Withdrawal of Fortovase (PDF)"
1455:
1425:
1398:
1384:
1358:
1332:
1192:. 22 March 2019. Archived from
1158:Fischer J, Ganellin CR (2006).
967:
810:
790:
712:Key:QWAXKHKRTORLEM-UGJKXSETSA-N
2177:Abacavir/lamivudine/zidovudine
1257:
1208:
1178:
1045:Australian Product Information
631:
613:
1:
1568:. 18 May 2005. Archived from
1161:Analogue-based Drug Discovery
999:
2718:Drugs developed by Genentech
1532:10.1517/17425247.2012.694865
1439:Food and Drug Administration
1339:Hilts PJ (8 December 1995).
955:
920:Food and Drug Administration
892:Cully M (28 November 2018).
799:. Typically it is used with
749:. Typically it is used with
737:, sold under the brand name
7:
2052:Tenofovir alafenamide (TAF)
984:to increase the saquinavir
10:
2754:
2047:Tenofovir disoproxil (TDF)
881:
603:Chemical and physical data
320:feces (81%) and urine (3%)
2607:
2522:
2416:
2398:
2380:
2362:
2343:
2336:
2315:
2156:
2103:
2077:
2068:Discovery and development
2060:
1943:
1931:
1894:
1848:
1839:Discovery and development
1831:
1803:
1753:
1675:Discovery and development
1667:
1649:
743:antiretroviral medication
720:
695:
675:
653:
640:
607:
602:
570:
542:
522:
502:
482:
462:
442:
422:
402:
377:
357:
329:
324:
314:
301:
288:
278:
264:
256:
218:
213:
188:
174:
148:
101:
87:
69:
61:
51:
46:
37:
28:
2364:Transcription inhibitors
2316:Pharmacokinetic boosters
2232:Dolutegravir/rilpivirine
2193:Cabotegravir/rilpivirine
1833:Protease Inhibitors (PI)
807:to increase its effect.
2728:HIV protease inhibitors
2648:initial regimen options
2214:Dolutegravir/lamivudine
1691:Enfuvirtide (ENF, T-20)
1669:Entry/fusion inhibitors
1592:Drug Information Portal
270:~4% (without ritonavir
2733:Decahydroisoquinolines
2489:Portmanteau inhibitors
2382:Translation inhibitors
2292:Lamivudine/raltegravir
2062:Non-nucleoside (NNRTI)
2020:Islatravir (EFdA, ISL)
911:
2504:Synergistic enhancers
2302:Lamivudine/zidovudine
2182:Atazanavir/cobicistat
2158:Combined formulations
1995:Zidovudine (AZT, ZDV)
1933:Reverse-transcriptase
1805:Maturation inhibitors
889:
878:and HIV-2 proteases.
2345:Uncoating inhibitors
2198:Darunavir/cobicistat
2038:Nucleotide analogues
1956:Nucleoside analogues
1755:Integrase inhibitors
1634:antiretroviral drugs
1572:on 22 February 2015.
741:among others, is an
2337:Experimental agents
2307:Lopinavir/ritonavir
2166:Abacavir/lamivudine
2140:Elsulfavirine (ESV)
1975:Emtricitabine (FTC)
1861:Fosamprenavir (FPV)
951:Society and culture
861:Mechanism of action
805:lopinavir/ritonavir
755:lopinavir/ritonavir
237:(Prescription only)
57:Invirase, Fortovase
25:
2634:Never to phase III
1780:Elvitegravir (EVG)
1775:Dolutegravir (DTG)
1770:Cabotegravir (CAB)
1443:. 24 December 1999
1372:. 26 November 2018
1065:. 26 December 2019
962:generic medication
912:
867:protease inhibitor
778:protease inhibitor
21:
2708:CYP3A4 inhibitors
2656:
2655:
2603:
2602:
2449:Diarylpyrimidines
2152:
2151:
2148:
2147:
2128:Rilpivirine (RPV)
2112:diarylpyrimidines
2095:Delavirdine (DLV)
1990:Zalcitabine (ddC)
1947:nucleotide (NRTI)
1927:
1926:
1785:Raltegravir (RAL)
1765:Bictegravir (BIC)
1744:Fostemsavir (FTR)
1659:Lenacapavir (LEN)
1651:Capsid inhibitors
1227:(10): 1401–1406.
1127:Minor LK (2006).
900:. Open Publishing
774:high blood lipids
732:
731:
666:Interactive image
572:CompTox Dashboard
294:Liver, mainly by
245:
232:
164:
131:
114:
16:Chemical compound
2745:
2685:
2684:
2673:
2672:
2671:
2664:
2474:Hydroxycarbamide
2341:
2340:
2135:Doravirine (DOR)
2123:Etravirine (ETR)
2118:Dapivirine (DPV)
2090:Nevirapine (NVP)
2075:
2074:
1980:Lamivudine (3TC)
1970:Didanosine (ddI)
1941:
1940:
1914:Tipranavir (TPV)
1902:Atazanavir (ATV)
1886:Saquinavir (SQV)
1876:Nelfinavir (NFV)
1856:Amprenavir (APV)
1846:
1845:
1728:Ibalizumab (IBA)
1623:
1616:
1609:
1600:
1599:
1595:
1574:
1573:
1566:News-Medical.Net
1558:
1552:
1551:
1517:
1508:
1502:
1501:
1499:
1497:
1483:
1477:
1476:
1474:
1468:. Archived from
1467:
1459:
1453:
1452:
1450:
1448:
1429:
1423:
1422:
1420:
1418:
1402:
1396:
1390:The CDC, in its
1388:
1382:
1381:
1379:
1377:
1362:
1356:
1355:
1353:
1351:
1336:
1330:
1329:
1327:
1325:
1293:
1282:
1281:
1261:
1255:
1254:
1236:
1212:
1206:
1205:
1203:
1201:
1196:on 28 April 2020
1182:
1176:
1175:
1155:
1149:
1148:
1124:
1118:
1117:
1115:
1113:
1098:
1075:
1074:
1072:
1070:
1055:
1049:
1048:
1036:
1030:
1029:
1027:
1025:
1010:
909:
907:
905:
865:Saquinavir is a
728:
727:
668:
648:
633:
627:
621:
615:
595:
594:
580:
578:
562:
556:
535:
515:
495:
475:
455:
435:
415:
395:
385:
384:
370:
306:
243:
240:
230:
227:
205:
162:
159:
141:
130:
127:
124:
113:
110:
97:
83:
42:
33:
26:
24:
20:
2753:
2752:
2748:
2747:
2746:
2744:
2743:
2742:
2693:
2692:
2691:
2679:
2669:
2667:
2659:
2657:
2652:
2651:
2639:
2624:Clinical trials
2599:
2550:Dexelvucitabine
2518:
2509:Tre recombinase
2412:
2394:
2376:
2372:Tat antagonists
2358:
2332:
2311:
2144:
2099:
2085:Efavirenz (EFV)
2064:
2056:
1985:Stavudine (d4T)
1946:
1934:
1923:
1908:Darunavir (DRV)
1890:
1881:Ritonavir (RTV)
1871:Lopinavir (LPV)
1866:Indinavir (IDV)
1835:
1827:
1799:
1757:
1749:
1703:Maraviroc (MVC)
1671:
1663:
1645:
1630:Antiviral drugs
1627:
1586:
1583:
1578:
1577:
1560:
1559:
1555:
1515:
1509:
1505:
1495:
1493:
1485:
1484:
1480:
1475:on 14 May 2006.
1472:
1465:
1461:
1460:
1456:
1446:
1444:
1431:
1430:
1426:
1416:
1414:
1403:
1399:
1389:
1385:
1375:
1373:
1364:
1363:
1359:
1349:
1347:
1337:
1333:
1323:
1321:
1308:(21): 689–693.
1294:
1285:
1278:
1262:
1258:
1213:
1209:
1199:
1197:
1184:
1183:
1179:
1172:
1156:
1152:
1141:
1125:
1121:
1111:
1109:
1100:
1099:
1078:
1068:
1066:
1057:
1056:
1052:
1037:
1033:
1023:
1021:
1020:. 20 March 2018
1012:
1011:
1007:
1002:
986:bioavailability
970:
958:
953:
937:bioavailability
922:(FDA), leading
903:
901:
898:Nature Research
884:
863:
844:cytochrome P450
829:
813:
793:
766:QT prolongation
723:
721:
716:
713:
708:
703:
702:
691:
688:
683:
682:
671:
646:
636:
630:
624:
618:
598:
574:
566:
538:
518:
498:
478:
458:
438:
418:
398:
381:
373:
353:
350:
337:
336:
304:
280:Protein binding
266:Bioavailability
258:Pharmacokinetic
252:
209:
177:
170:
151:
144:
17:
12:
11:
5:
2751:
2741:
2740:
2735:
2730:
2725:
2720:
2715:
2710:
2705:
2690:
2689:
2677:
2654:
2653:
2638:
2637:
2636:
2635:
2632:
2621:
2615:
2609:
2608:
2605:
2604:
2601:
2600:
2598:
2597:
2592:
2587:
2582:
2577:
2572:
2567:
2562:
2557:
2552:
2547:
2542:
2537:
2532:
2526:
2524:
2520:
2519:
2517:
2516:
2511:
2506:
2501:
2496:
2491:
2486:
2481:
2476:
2471:
2466:
2461:
2456:
2451:
2446:
2441:
2436:
2431:
2426:
2420:
2418:
2414:
2413:
2411:
2410:
2404:
2402:
2396:
2395:
2393:
2392:
2386:
2384:
2378:
2377:
2375:
2374:
2368:
2366:
2360:
2359:
2357:
2356:
2349:
2347:
2338:
2334:
2333:
2331:
2330:
2325:
2323:Cobicistat (c)
2319:
2317:
2313:
2312:
2310:
2309:
2304:
2299:
2294:
2289:
2284:
2279:
2274:
2269:
2264:
2259:
2254:
2249:
2244:
2239:
2234:
2229:
2223:
2217:
2211:
2206:
2200:
2195:
2190:
2184:
2179:
2174:
2168:
2162:
2160:
2154:
2153:
2150:
2149:
2146:
2145:
2143:
2142:
2137:
2132:
2131:
2130:
2125:
2120:
2107:
2105:
2101:
2100:
2098:
2097:
2092:
2087:
2081:
2079:
2072:
2058:
2057:
2055:
2054:
2049:
2033:
2032:
2027:
2022:
2017:
2012:
2007:
2002:
1997:
1992:
1987:
1982:
1977:
1972:
1967:
1965:Abacavir (ABC)
1951:
1949:
1945:Nucleoside and
1938:
1929:
1928:
1925:
1924:
1922:
1921:
1916:
1911:
1905:
1898:
1896:
1892:
1891:
1889:
1888:
1883:
1878:
1873:
1868:
1863:
1858:
1852:
1850:
1843:
1829:
1828:
1826:
1825:
1820:
1815:
1809:
1807:
1801:
1800:
1798:
1797:
1792:
1787:
1782:
1777:
1772:
1767:
1761:
1759:
1751:
1750:
1748:
1747:
1735:
1719:
1705:
1694:
1681:
1679:
1665:
1664:
1662:
1661:
1655:
1653:
1647:
1646:
1626:
1625:
1618:
1611:
1603:
1597:
1596:
1582:
1581:External links
1579:
1576:
1575:
1553:
1526:(8): 937–951.
1503:
1478:
1454:
1424:
1397:
1383:
1357:
1345:New York Times
1331:
1283:
1276:
1256:
1207:
1177:
1170:
1150:
1139:
1119:
1076:
1050:
1031:
1004:
1003:
1001:
998:
997:
996:
989:
969:
966:
957:
954:
952:
949:
883:
880:
862:
859:
828:
825:
812:
809:
792:
789:
730:
729:
718:
717:
715:
714:
711:
709:
706:
698:
697:
696:
693:
692:
690:
689:
686:
678:
677:
676:
673:
672:
670:
669:
661:
659:
651:
650:
644:
638:
637:
634:
628:
622:
616:
611:
605:
604:
600:
599:
597:
596:
583:
581:
568:
567:
565:
564:
548:
546:
540:
539:
537:
536:
528:
526:
520:
519:
517:
516:
508:
506:
500:
499:
497:
496:
488:
486:
480:
479:
477:
476:
468:
466:
460:
459:
457:
456:
448:
446:
440:
439:
437:
436:
428:
426:
420:
419:
417:
416:
408:
406:
400:
399:
397:
396:
388:
386:
375:
374:
372:
371:
363:
361:
355:
354:
352:
351:
340:
332:
331:
330:
327:
326:
322:
321:
318:
312:
311:
308:
299:
298:
292:
286:
285:
282:
276:
275:
268:
262:
261:
254:
253:
251:
250:
238:
224:
222:
216:
215:
211:
210:
208:
207:
194:
192:
186:
185:
180:
178:administration
172:
171:
169:
168:
166:
156:
154:
146:
145:
143:
142:
125:
107:
105:
99:
98:
91:
85:
84:
77:
67:
66:
63:
59:
58:
55:
49:
48:
44:
43:
35:
34:
15:
9:
6:
4:
3:
2:
2750:
2739:
2736:
2734:
2731:
2729:
2726:
2724:
2721:
2719:
2716:
2714:
2711:
2709:
2706:
2704:
2701:
2700:
2698:
2688:
2683:
2678:
2676:
2666:
2665:
2662:
2649:
2646:
2643:
2633:
2631:
2628:
2627:
2625:
2622:
2619:
2616:
2614:
2611:
2610:
2606:
2596:
2593:
2591:
2588:
2586:
2583:
2581:
2578:
2576:
2573:
2571:
2568:
2566:
2563:
2561:
2558:
2556:
2553:
2551:
2548:
2546:
2543:
2541:
2538:
2536:
2533:
2531:
2528:
2527:
2525:
2523:Failed agents
2521:
2515:
2512:
2510:
2507:
2505:
2502:
2500:
2497:
2495:
2492:
2490:
2487:
2485:
2482:
2480:
2477:
2475:
2472:
2470:
2467:
2465:
2462:
2460:
2457:
2455:
2452:
2450:
2447:
2445:
2442:
2440:
2437:
2435:
2432:
2430:
2427:
2425:
2422:
2421:
2419:
2415:
2409:
2406:
2405:
2403:
2401:
2397:
2391:
2390:Trichosanthin
2388:
2387:
2385:
2383:
2379:
2373:
2370:
2369:
2367:
2365:
2361:
2354:
2351:
2350:
2348:
2346:
2342:
2339:
2335:
2329:
2328:Ritonavir (r)
2326:
2324:
2321:
2320:
2318:
2314:
2308:
2305:
2303:
2300:
2298:
2295:
2293:
2290:
2288:
2285:
2283:
2280:
2278:
2275:
2273:
2270:
2268:
2265:
2263:
2260:
2258:
2255:
2253:
2250:
2248:
2245:
2243:
2240:
2238:
2235:
2233:
2230:
2227:
2224:
2221:
2218:
2215:
2212:
2210:
2207:
2204:
2201:
2199:
2196:
2194:
2191:
2188:
2185:
2183:
2180:
2178:
2175:
2172:
2169:
2167:
2164:
2163:
2161:
2159:
2155:
2141:
2138:
2136:
2133:
2129:
2126:
2124:
2121:
2119:
2116:
2115:
2114:
2113:
2109:
2108:
2106:
2102:
2096:
2093:
2091:
2088:
2086:
2083:
2082:
2080:
2076:
2073:
2071:
2069:
2063:
2059:
2053:
2050:
2048:
2045:
2043:
2039:
2035:
2034:
2031:
2028:
2026:
2023:
2021:
2018:
2016:
2013:
2011:
2008:
2006:
2003:
2001:
1998:
1996:
1993:
1991:
1988:
1986:
1983:
1981:
1978:
1976:
1973:
1971:
1968:
1966:
1963:
1961:
1957:
1953:
1952:
1950:
1948:
1942:
1939:
1936:
1930:
1920:
1917:
1915:
1912:
1909:
1906:
1903:
1900:
1899:
1897:
1893:
1887:
1884:
1882:
1879:
1877:
1874:
1872:
1869:
1867:
1864:
1862:
1859:
1857:
1854:
1853:
1851:
1847:
1844:
1842:
1840:
1834:
1830:
1824:
1821:
1819:
1816:
1814:
1811:
1810:
1808:
1806:
1802:
1796:
1793:
1791:
1788:
1786:
1783:
1781:
1778:
1776:
1773:
1771:
1768:
1766:
1763:
1762:
1760:
1756:
1752:
1745:
1741:
1740:
1736:
1733:
1729:
1725:
1724:
1720:
1717:
1713:
1709:
1706:
1704:
1700:
1699:
1695:
1692:
1688:
1687:
1683:
1682:
1680:
1678:
1676:
1670:
1666:
1660:
1657:
1656:
1654:
1652:
1648:
1643:
1639:
1636:used against
1635:
1631:
1624:
1619:
1617:
1612:
1610:
1605:
1604:
1601:
1593:
1589:
1585:
1584:
1571:
1567:
1563:
1557:
1549:
1545:
1541:
1537:
1533:
1529:
1525:
1521:
1514:
1507:
1492:
1488:
1482:
1471:
1464:
1458:
1442:
1440:
1434:
1428:
1412:
1408:
1401:
1393:
1387:
1371:
1367:
1361:
1346:
1342:
1335:
1319:
1315:
1311:
1307:
1303:
1299:
1292:
1290:
1288:
1279:
1277:9780443075926
1273:
1269:
1268:
1260:
1252:
1248:
1244:
1240:
1235:
1230:
1226:
1222:
1218:
1211:
1195:
1191:
1187:
1181:
1173:
1171:9783527607495
1167:
1163:
1162:
1154:
1146:
1142:
1140:9781420015706
1136:
1132:
1131:
1123:
1107:
1103:
1097:
1095:
1093:
1091:
1089:
1087:
1085:
1083:
1081:
1064:
1060:
1054:
1046:
1042:
1035:
1019:
1015:
1009:
1005:
994:
993:microemulsion
990:
987:
983:
979:
975:
974:
973:
965:
963:
948:
946:
942:
938:
932:
929:
925:
921:
917:
899:
895:
888:
879:
877:
872:
868:
858:
856:
851:
848:
845:
840:
838:
833:
824:
822:
818:
808:
806:
802:
798:
788:
785:
783:
779:
775:
771:
767:
762:
760:
756:
752:
748:
744:
740:
736:
726:
719:
710:
705:
704:
701:
694:
685:
684:
681:
674:
667:
663:
662:
660:
657:
652:
645:
643:
639:
612:
610:
606:
601:
593:
589:
588:DTXSID6044012
585:
584:
582:
573:
569:
561:
560:RCSB PDB
555:
550:
549:
547:
545:
541:
534:
530:
529:
527:
525:
521:
514:
510:
509:
507:
505:
501:
494:
490:
489:
487:
485:
481:
474:
470:
469:
467:
465:
461:
454:
450:
449:
447:
445:
441:
434:
430:
429:
427:
425:
421:
414:
410:
409:
407:
405:
401:
394:
390:
389:
387:
380:
376:
369:
365:
364:
362:
360:
356:
348:
344:
339:
338:
335:
328:
323:
319:
317:
313:
309:
307:
300:
297:
293:
291:
287:
283:
281:
277:
273:
269:
267:
263:
259:
255:
249:
239:
236:
226:
225:
223:
221:
217:
212:
204:
199:
196:
195:
193:
191:
187:
184:
181:
179:
173:
167:
158:
157:
155:
153:
147:
140:
135:
126:
123:
118:
109:
108:
106:
104:
100:
96:
92:
90:
86:
82:
78:
76:
72:
68:
64:
60:
56:
54:
50:
47:Clinical data
45:
41:
36:
32:
27:
19:
2723:Hepatotoxins
2703:Carboxamides
2644:
2444:Cyanovirin-N
2434:Calanolide A
2110:
2104:2 generation
2078:1 generation
2065:
2036:
2015:Elvucitabine
2005:Apricitabine
1954:
1895:2 generation
1885:
1849:1 generation
1836:
1823:Fipravirimat
1737:
1732:Semzuvolimab
1721:
1712:Cenicriviroc
1696:
1684:
1672:
1591:
1588:"Saquinavir"
1570:the original
1565:
1556:
1523:
1519:
1506:
1494:. Retrieved
1490:
1481:
1470:the original
1457:
1445:. Retrieved
1436:
1427:
1415:. Retrieved
1409:. The Body.
1400:
1391:
1386:
1374:. Retrieved
1369:
1360:
1348:. Retrieved
1344:
1334:
1322:. Retrieved
1305:
1301:
1267:AIDS Therapy
1266:
1259:
1224:
1220:
1210:
1198:. Retrieved
1194:the original
1189:
1180:
1160:
1153:
1129:
1122:
1110:. Retrieved
1102:"Saquinavir"
1067:. Retrieved
1062:
1053:
1044:
1034:
1022:. Retrieved
1017:
1008:
971:
968:Formulations
959:
933:
913:
902:. Retrieved
897:
864:
852:
841:
834:
830:
814:
811:Side effects
794:
791:Medical uses
786:
782:HIV protease
763:
738:
734:
733:
722:
524:NIAID ChemDB
346:
342:
303:Elimination
220:Legal status
214:Legal status
103:License data
18:
2645:recommended
2620:from market
2570:Lersivirine
2545:Capravirine
2484:Miltefosine
2469:Griffithsin
2464:Fosdevirine
2010:Censavudine
1640:(primarily
1417:20 February
1395:inhibitors.
1186:"Fortovase"
1112:5 September
770:heart block
649: g·mol
368:127779-20-8
325:Identifiers
89:MedlinePlus
62:Other names
53:Trade names
2738:Quinolines
2697:Categories
2575:Lodenosine
2555:Droxinavir
2540:Brecanavir
2535:Atevirdine
2499:Seliciclib
2494:Scytovirin
2408:Elipovimab
2353:TRIM5alpha
2030:Stampidine
1935:inhibitors
1919:TMC-310911
1818:BMS-955176
1716:Leronlimab
1708:Vicriviroc
1447:28 January
1376:29 October
1350:28 October
1324:8 November
1200:28 January
1069:28 January
1024:28 January
1000:References
904:28 October
855:omeprazole
735:Saquinavir
654:3D model (
642:Molar mass
544:PDB ligand
473:L3JE09KZ2F
444:ChemSpider
404:IUPHAR/BPS
359:CAS Number
334:IUPAC name
310:9–15 hours
290:Metabolism
139:Saquinavir
23:Saquinavir
2630:Phase III
2618:Withdrawn
2595:Telinavir
2590:Palinavir
2585:Mozenavir
2565:Emivirine
2560:Lasinavir
2530:Aplaviroc
2459:Foscarnet
2439:Ceragenin
2000:Amdoxovir
1813:Bevirimat
1790:BI 224436
1491:Drugs.com
1190:Drugs.com
1018:Drugs.com
982:ritonavir
956:Economics
941:ritonavir
928:indinavir
924:ritonavir
871:Proteases
837:ritonavir
817:diarrhoea
801:ritonavir
751:ritonavir
513:ChEMBL114
316:Excretion
305:half-life
176:Routes of
150:Pregnancy
81:Monograph
75:Drugs.com
2675:Medicine
2580:Loviride
1548:28468973
1540:22663249
1411:Archived
1318:Archived
1314:21637182
1251:44506039
1243:16791014
1145:Archived
1106:Archived
1063:DailyMed
978:mesylate
797:HIV/AIDS
759:by mouth
747:HIV/AIDS
739:Invirase
725:(verify)
424:DrugBank
272:boosting
190:ATC code
183:By mouth
165: B1
152:category
134:DailyMed
2687:Viruses
2661:Portals
2479:KP-1461
2025:Racivir
1795:MK-2048
882:History
647:670.855
609:Formula
433:DB01232
379:PubChem
206:)
200: (
198:J05AE01
136::
119::
95:a696001
2613:WHO-EM
2429:BIT225
2424:Abzyme
2355:(gene)
2042:NtRTIs
1937:(RTIs)
1546:
1538:
1496:9 July
1312:
1274:
1249:
1241:
1168:
1137:
821:nausea
680:SMILES
533:000640
504:ChEMBL
493:D00429
453:390016
393:441243
296:CYP3A4
248:℞-only
246:
233:
132:
122:by INN
115:
2417:Other
2400:BNAbs
1960:NRTIs
1739:gp120
1544:S2CID
1516:(PDF)
1473:(PDF)
1466:(PDF)
1441:(FDA)
1437:U.S.
1247:S2CID
916:Roche
876:HIV-1
700:InChI
656:JSmol
551:ROC (
2642:DHHS
1698:CCR5
1686:gp41
1536:PMID
1498:2020
1449:2020
1419:2013
1378:2020
1352:2020
1326:2013
1310:PMID
1272:ISBN
1239:PMID
1221:AIDS
1202:2020
1166:ISBN
1135:ISBN
1114:2015
1071:2020
1026:2020
926:and
906:2020
554:PDBe
484:KEGG
464:UNII
413:4813
260:data
71:AHFS
1723:CD4
1642:J05
1638:HIV
1528:doi
1370:NIH
1229:doi
847:3A4
803:or
753:or
577:EPA
383:CID
284:98%
203:WHO
117:EMA
65:SQV
2699::
2626::
1730:,
1714:,
1710:,
1632::
1590:.
1564:.
1542:.
1534:.
1522:.
1518:.
1489:.
1435:.
1368:.
1343:.
1316:.
1306:60
1304:.
1300:.
1286:^
1245:.
1237:.
1225:20
1223:.
1219:.
1188:.
1143:.
1079:^
1061:.
1043:.
1016:.
964:.
947:.
896:.
869:.
857:.
819:,
784:.
772:,
768:,
761:.
623:50
617:38
557:,
345:)-
341:(2
242:US
235:S4
229:AU
161:AU
129:US
112:EU
2663::
2640:°
2228:°
2222:°
2216:°
2205:°
2189:°
2173:°
2070:)
2066:(
2044::
2040:/
1962::
1958:/
1910:°
1904:°
1841:)
1837:(
1746:)
1742:(
1734:)
1726:(
1718:)
1701:(
1693:)
1689:(
1677:)
1673:(
1644:)
1622:e
1615:t
1608:v
1550:.
1530::
1524:9
1500:.
1451:.
1421:.
1380:.
1354:.
1328:.
1280:.
1253:.
1231::
1204:.
1174:.
1116:.
1073:.
1028:.
988:;
908:.
658:)
635:5
632:O
629:6
626:N
620:H
614:C
579:)
575:(
563:)
347:N
343:S
274:)
244::
231::
163::
73:/
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.