47:
304:
229:
1010:, which are used in a number of coatings applications for protecting against damage caused by UV light, oxidation, and corrosion. One application of malonic acid is in the coatings industry as a crosslinker for low-temperature cure powder coatings, which are becoming increasingly valuable for heat sensitive substrates and a desire to speed up the coatings process. The global coatings market for automobiles was estimated to be $ 18.59 billion in 2014 with projected combined annual growth rate of 5.1% through 2022.
707:
38:
925:
906:
1160:
1961:
1038:
Malonic acid (up to 37.5% w/w) has been used to cross-link corn and potato starches to produce a biodegradable thermoplastic; the process is performed in water using non-toxic catalysts. Starch-based polymers comprised 38% of the global biodegradable polymers market in 2014 with food packaging, foam
1591:
1142:− group required for dehydrogenation. This observation was used to deduce the structure of the active site in succinate dehydrogenase. Inhibition of this enzyme decreases cellular respiration. Since malonic acid is a natural component of many foods, it is present in mammals including humans.
1017:
industry, flavors and fragrances industry, specialty solvents, polymer crosslinking, and pharmaceutical industry. In 2004, annual global production of malonic acid and related diesters was over 20,000 metric tons. Potential growth of these markets could result from advances in industrial
1610:
2367:
2029:
2374:
579:
2153:
Bowman, Caitlyn E.; Rodriguez, Susana; Selen
Alpergin, Ebru S.; Acoba, Michelle G.; Zhao, Liang; Hartung, Thomas; Claypool, Steven M.; Watkins, Paul A.; Wolfgang, Michael J. (2017).
1024:
In food and drug applications, malonic acid can be used to control acidity, either as an excipient in pharmaceutical formulation or natural preservative additive for foods.
2043:
de Sain-van der Velden MG, van der Ham M, Jans JJ, Visser G, Prinsen HC, Verhoeven-Duif NM, et al. (2016). Morava E, Baumgartner M, Patterson M, Rahman S (eds.).
1959:, Netravali AN, Dastidar TG, "Crosslinked native and waxy starch resin compositions and processes for their manufacture.", assigned to Cornell University
1208:
1151:
1848:
Facke T, Subramanian R, Dvorchak M, Feng S (February 2004). "Diethylmalonate blocked isocyanate as crosslinkers for low temperature cure powder coatings.".
1027:
Malonic acid is used as a building block chemical to produce numerous valuable compounds, including the flavor and fragrance compounds gamma-nonalactone,
2360:
1315:
696:
353:
1058:
1392:
1362:
1874:
1544:
2104:"Mammalian ACSF3 Protein Is a Malonyl-CoA Synthetase That Supplies the Chain Extender Units for Mitochondrial Fatty Acid Synthesis"
1975:
Ghosh
Dastidar T, Netravali AN (November 2012). "'Green' crosslinking of native starches with malonic acid and their properties".
1061:(CMAMMA). By calculating the malonic acid to methylmalonic acid ratio in blood plasma, CMAMMA can be distinguished from classic
1508:
Gopalan RS, Kumaradhas P, Kulkarni GU, Rao CN (2000). "An experimental charge density study of aliphatic dicarboxylic acids".
1074:
2068:
1911:
Top Value Added
Chemicals From Biomass. Volume I: Results of Screening for Potential Candidates from Sugars and Synthesis Gas
1339:
749:
to sodium malonate, and acidification affords malonic acid. Industrially, however, malonic acid is produced by hydrolysis of
1813:
Perks HM, Liebman JF (2000). "Paradigms and
Paradoxes: Aspects of the Energetics of Carboxylic Acids and Their Anhydrides".
1021:
In 2004, malonic acid was listed by the US Department of Energy as one of the top 30 chemicals to be produced from biomass.
2245:"Studies on the Mechanism of Hydrogen Transport in Animal Tissues : VI. Inhibitor Studies with Succinic Dehydrogenase"
2383:
2010:
1487:"Note sur un acide obtenu par l'oxydation de l'acide malique"] (Note on an acid obtained by oxidation of malic acid)"
2155:"The Mammalian Malonyl-CoA Synthetase ACSF3 Is Required for Mitochondrial Protein Malonylation and Metabolic Efficiency"
1946:
Hildbrand, S.; Pollak, P. Malonic Acid & Derivatives. March 15, 2001. Ullmann's
Encyclopedia of Industrial Chemistry
1014:
318:
1910:
1589:, Britton EC, Ezra M, "Production of malonic acid", issued 1945-04-03, assigned to Dow Chemical Co
594:
126:
1180:
664:
Malonic acid is a naturally occurring substance found in many fruits and vegetables. There is a suggestion that
1658:
1641:
119:
1608:, Dietrich JA, "Recombinant host cells for the production of malonate.", assigned to Lygos Inc
830:
261:
224:
1605:
2644:
2625:
282:
1323:
1006:
for use in polyesters and polymers (whose usefulness is unclear though). It can also be a component in
845:
Malonic acid is used to prepare a,b-unsaturated carboxylic acids by condensation and decarboxylation.
809:, a versatile intermediate in further transformations. The esters of malonic acid are also used as a CH
464:
166:
2024:
1956:
1586:
1123:
734:
2352:
1042:
1013:
It is used in a number of manufacturing processes as a high value specialty chemical including the
883:
299:
1155:
1119:
818:
695:
and extensive property data including for condensed phase thermochemistry are available from the
493:
1486:
1112:
1101:
1062:
790:
1863:
James S. Global
Automotive Coatings Market. 2015 Grand View Research Market Report (Report).
1386:
1359:
1624:
Pollak P, Romeder G (2005). "Malonic Acid and
Derivatives". In Pollak P, Romeder G (eds.).
1517:
1183:
1105:
1018:
biotechnology that seeks to displace petroleum-based chemicals in industrial applications.
972:
918:
270:
59:
1885:
1412:
8:
2347:
834:
186:
95:
85:
1521:
672:
contain higher levels of malonic acid than fruits produced in conventional agriculture.
303:
228:
146:
2601:
2550:
2269:
2244:
2179:
2154:
2130:
2103:
2079:
2044:
1850:
Proceedings of 31st
International Waterborene, High-Solids and Powder Coating Symposium
1830:
1787:
1687:-Acylamino-1,3-dienes from 2,4-Pentadienoic Acids by the Curtius Rearrangement: benzyl
1176:
1054:
789:
is most useful to obtain diesters or diamides. In a well-known reaction, malonic acid
46:
2220:
2203:
1529:
1438:"Organic Acids Concentration in Citrus Juice from Conventional Versus Organic Farming"
2305:
2274:
2225:
2184:
2135:
2084:
2064:
1992:
1927:
1693:
1637:
1564:
1467:
1437:
1335:
1290:
1285:
1265:
987:
806:
782:
750:
711:
700:
676:
610:
573:
561:
1834:
1749:"Ueber die der Sorbinsäure homologen, ungesättigten Säuren mit zwei Doppelbindungen"
1453:
2301:
2294:
Biochimica et
Biophysica Acta (BBA) - Specialized Section on Enzymological Subjects
2264:
2256:
2215:
2174:
2170:
2166:
2125:
2115:
2074:
2056:
1984:
1917:
1822:
1795:
1760:
1729:
1702:
1629:
1525:
1457:
1449:
1327:
1280:
1269:
913:
When malonic acid is condensed in hot pyridine, the condensation is accompanied by
786:
754:
746:
730:
722:
718:
638:
630:
453:
381:
1988:
1045:
Kodak company and others use malonic acid and derivatives as a surgical adhesive.
1366:
1320:
Nomenclature of
Organic Chemistry: IUPAC Recommendations and Preferred Names 2013
1314:
1003:
942:
914:
899:
798:
692:
669:
557:
527:
1559:
250:
2567:
1633:
938:
726:
649:
588:
537:
1826:
1799:
1764:
2638:
2446:
2428:
1733:
1706:
1471:
1381:
1131:
1028:
846:
778:
541:
442:
432:
217:
2120:
1909:
Werpy TA, Holladay JE, White JF (August 2004). Werpy TA, Petersen G (eds.).
1172:
706:
2584:
2533:
2516:
2499:
2482:
2278:
2229:
2188:
2139:
2088:
2042:
1996:
1683:
Jessup, Peter J.; Petty, C. Bruce; Roos, Jan; Overman, Larry E. (1979). "1-
758:
545:
2319:
2060:
1659:"The Effect of Salicylic Acid on the Briggs-Rauscher Oscillating Reaction"
1331:
2464:
2404:
2260:
2204:"Malonate inhibition of oxidations in the Krebs tricarboxylic acid cycle"
1295:
1127:
1078:
785:
can be used as an intermediate to mono-ester or amide derivatives, while
642:
533:
21:
2292:
Dervartanian DV, Veeger C (1964). "Studies on succinate dehydrogenase".
1462:
1396:. Vol. 17 (11th ed.). Cambridge University Press. p. 495.
437:
135 to 137 °C (275 to 279 °F; 408 to 410 K) (decomposes)
206:
1778:
1097:
1089:
924:
742:
680:
409:
177:
17:
2382:
1932:
1032:
999:
905:
1782:
1748:
587:
Except where otherwise noted, data are given for materials in their
1922:
1385:
1241:
1220:
1130:
of the enzyme without reacting, competing with the usual substrate
891:
887:
518:
197:
2152:
1092:
derivative of malonate, malonyl-CoA, is an important precursor in
125:
118:
37:
1164:
1093:
814:
802:
762:
738:
422:
237:
2027:, Hawkins G, Fassett D, "Surgical Adhesive Compositions"
2348:
Calculator: Water and solute activities in aqueous malonic acid
1248:
1238:
1227:
1223:
1116:
1100:. Malonyl CoA is formed there from acetyl CoA by the action of
895:
665:
157:
675:
Malonic acid was first prepared in 1858 by the French chemist
2102:
Witkowski, Andrzej; Thweatt, Jennifer; Smith, Stuart (2011).
1273:
1082:
1039:
packaging, and compost bags as the largest end-use segments.
1007:
774:
645:
626:
137:
108:
1847:
1507:
1053:
If elevated malonic acid levels are accompanied by elevated
287:
1159:
971:
The transformation is achieved by warming a dry mixture of
794:
2045:"A New Approach for Fast Metabolic Diagnostics in CMAMMA"
1215:
928:
The Doebner modification of the Knoevenagel condensation.
773:
Malonic acid reacts as a typical carboxylic acid forming
622:
512:
1974:
1682:
699:. A classical preparation of malonic acid starts from
2101:
1955:
1720:
Allen, C. F. H.; VanAllan, J. (1944). "Sorbic Acid".
1186:
1436:
Ha CN, Ngoc ND, Ngoc CP, Trung DD, Quang BN (2012).
2011:
Biodegradable Polymers: Chemical Economics Handbook
2291:
1908:
1585:
1542:
1360:pKa Data Compiled by R. Williams (pdf; 77 kB)
1202:
986:) and malonic acid. It reacts in a similar way to
327:InChI=1S/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7)
1316:International Union of Pure and Applied Chemistry
932:
909:Z=COOH (malonic acid) or Z=COOR' (malonate ester)
337:InChI=1/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7)
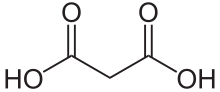
27:Carboxylic acid with chemical formula CH2(COOH)2
2636:
2023:
1057:levels, this may indicate the metabolic disease
249:
1435:
94:
1753:Berichte der Deutschen Chemischen Gesellschaft
697:National Institute of Standards and Technology
2368:
2195:
1719:
1623:
2242:
2201:
1968:
1812:
1150:The fluorinated version of malonic acide is
686:
1059:combined malonic and methylmalonic aciduria
840:
824:
691:The structure has been determined by X-ray
2375:
2361:
1484:
1355:
1353:
1351:
801:. Malonic acid may also be condensed with
302:
227:
185:
2268:
2219:
2178:
2129:
2119:
2078:
1931:
1921:
1777:
1461:
1111:Malonic acid is the classic example of a
998:Malonic acid is a precursor to specialty
269:
1626:Van Nostrand's Encyclopedia of Chemistry
1380:
1376:
1374:
1158:
1104:, and the malonate is transferred to an
923:
904:
705:
2055:. Berlin, Heidelberg: Springer: 15–22.
1746:
1656:
1604:
1348:
1211:is 2.8 and the second is 5.7. Thus the
1077:(mtFASII), in which it is converted to
829:Malonic acid is a key component in the
298:
205:
2637:
937:Malonic acid does not readily form an
218:
2356:
1407:
1405:
1403:
1371:
625:form of malonic acid, as well as its
330:Key: OFOBLEOULBTSOW-UHFFFAOYSA-N
165:
145:
1145:
1124:respiratory electron transport chain
757:. It has also been produced through
2384:Linear saturated dicarboxylic acids
2243:Potter VR, Dubois KP (March 1943).
2208:The Journal of Biological Chemistry
2202:Pardee AB, Potter VR (March 1949).
1916:(Report). US Department of Energy.
1884:. UNEP Publications. Archived from
1163:Chemical structure of the malonate
1108:to be added to a fatty acid chain.
1096:fatty acid biosynthesis along with
1083:acyl-CoA synthetase family member 3
340:Key: OFOBLEOULBTSOW-UHFFFAOYAJ
240:
13:
1557:
1400:
1075:mitochondrial fatty acid synthesis
1031:, and the pharmaceutical compound
886:, malonic acid condenses with the
14:
2656:
2341:
2249:The Journal of General Physiology
1862:
1073:Malonic acid is the precursor in
710:Preparation of malonic acid from
2013:(Report). IHS Markit. June 2021.
393:
45:
36:
2312:
2285:
2236:
2146:
2108:Journal of Biological Chemistry
2095:
2036:
2017:
2003:
1949:
1940:
1902:
1867:
1856:
1841:
1806:
1771:
1740:
1713:
1676:
1650:
1617:
1598:
1579:
1454:10.17660/actahortic.2012.933.78
1068:
993:
648:. The name originates from the
591:(at 25 °C , 100 kPa).
2171:10.1016/j.chembiol.2017.04.009
1551:
1536:
1510:Journal of Molecular Structure
1501:
1478:
1429:
1324:The Royal Society of Chemistry
1308:
933:Preparation of carbon suboxide
729:to provide the sodium salt of
399:
387:
1:
2320:"Metabocard for Malonic acid"
2221:10.1016/S0021-9258(18)56954-4
1989:10.1016/j.carbpol.2012.07.041
1691:-1,3-butadiene-1-carbamate".
1530:10.1016/S0022-2860(99)00293-8
1301:
1175:; that is, it can donate two
835:oscillating chemical reaction
725:, which is then reacted with
2306:10.1016/0926-6569(64)90182-8
1783:"Ueber das Kohlensuboxyd. I"
1048:
833:, the classic example of an
768:
7:
2626:Category:Dicarboxylic acids
1002:. It can be converted into
10:
2661:
1634:10.1002/0471740039.vec1571
1574:, vol. 2, p. 376
849:are prepared in this way:
659:
15:
2623:
2585:Hexadecanedioic acid
2399:
2324:Human Metabolome Database
1800:10.1002/cber.190603901103
1765:10.1002/cber.190203501187
1276:of malonic acid, such as
735:nucleophilic substitution
687:Structure and preparation
585:
567:
504:
374:
349:
314:
78:
70:
58:
53:
44:
35:
2604:(Phelogenic acid) C
2568:Tredecanedioic acid
1734:10.15227/orgsyn.024.0092
1707:10.15227/orgsyn.059.0001
1543:NIST Chemistry WebBook.
1179:per molecule. Its first
884:Knoevenagel condensation
866:+ ArCHO → ArCH=CHCO
841:Knoevenagel condensation
831:Briggs–Rauscher reaction
825:Briggs–Rauscher reaction
73:Methanedicarboxylic acid
16:Not to be confused with
2551:Dodecanedioic acid
2121:10.1074/jbc.M111.291591
1875:"Malonic acid diesters"
1827:10.1023/A:1009270411806
1417:The Good Scents Company
1393:Encyclopædia Britannica
1120:succinate dehydrogenase
882:In this, the so-called
819:malonic ester synthesis
494:Magnetic susceptibility
2483:Heptanedioic acid
2449:(Glutaric acid) C
2447:Pentanedioic acid
2431:(Succinic acid) C
2417:(Malonic acid) CH
2415:Propanedioic acid
2407:(Oxalic acid) (CO
1204:
1203:{\displaystyle pK_{a}}
1168:
1102:acetyl-CoA carboxylase
1063:methylmalonic acidemia
929:
910:
715:
2587:(Thapsic acid) C
2536:(Sebacic acid) C
2534:Decanedioic acid
2519:(Azelaic acid) C
2517:Nonanedioic acid
2502:(Suberic acid) C
2500:Octanedioic acid
2485:(Pimelic acid) C
2465:Hexanedioic acid
2429:Butanedioic acid
2405:Ethanedioic acid
2159:Cell Chemical Biology
2061:10.1007/8904_2016_531
1977:Carbohydrate Polymers
1587:US patent 2373011
1485:Dessaignes V (1858).
1332:10.1039/9781849733069
1205:
1162:
1122:(complex II), in the
1113:competitive inhibitor
990:, forming malonates.
962:→ O=C=C=C=O + 2 H
927:
908:
721:generates the sodium
709:
679:via the oxidation of
2467:(Adipic acid) C
2261:10.1085/jgp.26.4.391
1815:Structural Chemistry
1657:Csepei LI, Bolla C.
1387:"Malonic Acid"
1184:
1152:difluoromalonic acid
1106:acyl carrier protein
973:phosphorus pentoxide
941:, dehydration gives
919:Doebner modification
60:Preferred IUPAC name
2114:(39): 33729–33736.
1545:"Propanedioic acid"
1522:2000JMoSt.521...97S
1413:"Propanedioic acid"
1134:but lacking the −CH
668:fruits produced in
656:) meaning 'apple'.
454:Solubility in water
417: g·mol
32:
2645:Dicarboxylic acids
2602:Docosanedioic acid
2570:(Brassylic acid) C
1747:Doebner O (1902).
1442:Acta Horticulturae
1365:2010-06-02 at the
1200:
1169:
1126:. It binds to the
1088:Additionally, the
1055:methylmalonic acid
930:
911:
716:
641:is malonic acid's
595:Infobox references
505:Related compounds
30:
2632:
2631:
2165:(6): 673–684.e4.
2070:978-3-662-53681-0
1781:, Wolf B (1906).
1722:Organic Syntheses
1694:Organic Syntheses
1666:Studia UBB Chemia
1572:Collected Volumes
1565:Organic Syntheses
1341:978-0-85404-182-4
1291:Disodium malonate
1286:Dimethyl malonate
1146:Related Chemicals
988:malonic anhydride
783:Malonic anhydride
751:dimethyl malonate
712:chloroacetic acid
701:chloroacetic acid
677:Victor Dessaignes
613:with structure CH
611:dicarboxylic acid
603:Chemical compound
601:
600:
574:Safety data sheet
562:Dimethyl malonate
553:Related compounds
283:CompTox Dashboard
127:Interactive image
120:Interactive image
64:Propanedioic acid
2652:
2377:
2370:
2363:
2354:
2353:
2335:
2334:
2332:
2331:
2316:
2310:
2309:
2289:
2283:
2282:
2272:
2240:
2234:
2233:
2223:
2199:
2193:
2192:
2182:
2150:
2144:
2143:
2133:
2123:
2099:
2093:
2092:
2082:
2040:
2034:
2033:
2032:
2028:
2021:
2015:
2014:
2007:
2001:
2000:
1972:
1966:
1965:
1964:
1960:
1953:
1947:
1944:
1938:
1937:
1935:
1925:
1915:
1906:
1900:
1899:
1897:
1896:
1890:
1879:
1871:
1865:
1864:
1860:
1854:
1853:
1845:
1839:
1838:
1810:
1804:
1803:
1775:
1769:
1768:
1744:
1738:
1737:
1717:
1711:
1709:
1680:
1674:
1673:
1663:
1654:
1648:
1647:
1621:
1615:
1614:
1613:
1609:
1602:
1596:
1595:
1594:
1590:
1583:
1577:
1575:
1568:
1555:
1549:
1548:
1540:
1534:
1533:
1505:
1499:
1498:
1482:
1476:
1475:
1465:
1448:(933): 601–606.
1433:
1427:
1426:
1424:
1423:
1409:
1398:
1397:
1389:
1378:
1369:
1357:
1346:
1345:
1312:
1281:Diethyl malonate
1260:
1259:
1258:
1255:
1235:
1209:
1207:
1206:
1201:
1199:
1198:
1171:Malonic acid is
985:
967:
917:, the so-called
898:, followed by a
878:
787:malonyl chloride
755:diethyl malonate
747:sodium hydroxide
731:cyanoacetic acid
719:Sodium carbonate
639:diethyl malonate
528:carboxylic acids
500:-46.3·10 cm/mol
416:
401:
395:
389:
382:Chemical formula
307:
306:
291:
289:
273:
253:
242:
231:
220:
209:
189:
169:
149:
129:
122:
98:
49:
40:
33:
29:
2660:
2659:
2655:
2654:
2653:
2651:
2650:
2649:
2635:
2634:
2633:
2628:
2619:
2618:
2615:
2611:
2607:
2598:
2594:
2590:
2581:
2577:
2573:
2564:
2560:
2556:
2547:
2543:
2539:
2530:
2526:
2522:
2513:
2509:
2505:
2496:
2492:
2488:
2478:
2474:
2470:
2460:
2456:
2452:
2442:
2438:
2434:
2424:
2420:
2410:
2395:
2393:
2389:
2381:
2344:
2339:
2338:
2329:
2327:
2318:
2317:
2313:
2290:
2286:
2241:
2237:
2200:
2196:
2151:
2147:
2100:
2096:
2071:
2041:
2037:
2030:
2022:
2018:
2009:
2008:
2004:
1973:
1969:
1962:
1954:
1950:
1945:
1941:
1913:
1907:
1903:
1894:
1892:
1888:
1877:
1873:
1872:
1868:
1861:
1857:
1846:
1842:
1811:
1807:
1776:
1772:
1745:
1741:
1718:
1714:
1681:
1677:
1661:
1655:
1651:
1644:
1622:
1618:
1611:
1603:
1599:
1592:
1584:
1580:
1570:
1556:
1552:
1541:
1537:
1516:(1–3): 97–106.
1506:
1502:
1483:
1479:
1434:
1430:
1421:
1419:
1411:
1410:
1401:
1379:
1372:
1367:Wayback Machine
1358:
1349:
1342:
1326:. p. 746.
1313:
1309:
1304:
1256:
1253:
1252:
1246:
1237:
1233:
1219:
1194:
1190:
1185:
1182:
1181:
1148:
1141:
1137:
1071:
1051:
1004:1,3-propanediol
996:
984:
980:
976:
965:
961:
957:
953:
949:
943:carbon suboxide
935:
915:decarboxylation
900:decarboxylation
877:
873:
869:
865:
861:
857:
853:
843:
827:
812:
799:barbituric acid
771:
693:crystallography
689:
670:organic farming
662:
637:. For example,
633:, are known as
620:
616:
604:
597:
592:
560:
558:Malondialdehyde
554:
544:
540:
536:
530:
515:
497:
486:
482:
480:
473:
456:
414:
404:
398:
392:
384:
370:
367:
366:C(C(=O)O)C(=O)O
362:
357:
356:
345:
342:
341:
338:
332:
331:
328:
322:
321:
310:
292:
285:
276:
256:
243:
212:
192:
172:
152:
132:
112:
101:
88:
74:
66:
65:
28:
25:
12:
11:
5:
2658:
2648:
2647:
2630:
2629:
2624:
2621:
2620:
2617:
2616:
2613:
2609:
2605:
2599:
2596:
2592:
2588:
2582:
2579:
2575:
2571:
2565:
2562:
2558:
2554:
2548:
2545:
2541:
2537:
2531:
2528:
2524:
2520:
2514:
2511:
2507:
2503:
2497:
2494:
2490:
2486:
2480:
2476:
2472:
2468:
2462:
2458:
2454:
2450:
2444:
2440:
2436:
2432:
2426:
2422:
2418:
2412:
2408:
2401:
2400:
2397:
2396:
2391:
2387:
2380:
2379:
2372:
2365:
2357:
2351:
2350:
2343:
2342:External links
2340:
2337:
2336:
2311:
2300:(2): 233–247.
2284:
2255:(4): 391–404.
2235:
2214:(1): 241–250.
2194:
2145:
2094:
2069:
2035:
2016:
2002:
1967:
1948:
1939:
1923:10.2172/926125
1901:
1866:
1855:
1840:
1821:(4): 265–269.
1805:
1770:
1739:
1712:
1675:
1649:
1642:
1616:
1606:US 20200172941
1597:
1578:
1560:"Malonic acid"
1550:
1535:
1500:
1491:Comptes rendus
1477:
1428:
1399:
1384:, ed. (1911).
1382:Chisholm, Hugh
1370:
1347:
1340:
1306:
1305:
1303:
1300:
1299:
1298:
1293:
1288:
1283:
1261:. Malonate or
1244:
1231:
1197:
1193:
1189:
1147:
1144:
1139:
1135:
1070:
1067:
1050:
1047:
995:
992:
982:
978:
969:
968:
963:
959:
955:
951:
934:
931:
880:
879:
875:
871:
867:
863:
859:
855:
847:Cinnamic acids
842:
839:
826:
823:
810:
807:Meldrum's acid
777:, ester, and
770:
767:
727:sodium cyanide
688:
685:
661:
658:
618:
614:
602:
599:
598:
593:
589:standard state
586:
583:
582:
577:
570:
569:
565:
564:
555:
552:
549:
548:
538:Propionic acid
531:
525:
522:
521:
516:
510:
507:
506:
502:
501:
498:
492:
489:
488:
484:
478:
475:
471:
461:
460:
457:
452:
449:
448:
445:
439:
438:
435:
429:
428:
425:
419:
418:
412:
406:
405:
402:
396:
390:
385:
380:
377:
376:
372:
371:
369:
368:
365:
363:
360:
352:
351:
350:
347:
346:
344:
343:
339:
336:
335:
333:
329:
326:
325:
317:
316:
315:
312:
311:
309:
308:
295:
293:
281:
278:
277:
275:
274:
266:
264:
258:
257:
255:
254:
246:
244:
236:
233:
232:
222:
214:
213:
211:
210:
202:
200:
194:
193:
191:
190:
182:
180:
174:
173:
171:
170:
162:
160:
154:
153:
151:
150:
142:
140:
134:
133:
131:
130:
123:
115:
113:
106:
103:
102:
100:
99:
91:
89:
84:
81:
80:
76:
75:
72:
68:
67:
63:
62:
56:
55:
51:
50:
42:
41:
26:
9:
6:
4:
3:
2:
2657:
2646:
2643:
2642:
2640:
2627:
2622:
2603:
2600:
2586:
2583:
2569:
2566:
2552:
2549:
2535:
2532:
2518:
2515:
2501:
2498:
2484:
2481:
2466:
2463:
2448:
2445:
2430:
2427:
2416:
2413:
2406:
2403:
2402:
2398:
2385:
2378:
2373:
2371:
2366:
2364:
2359:
2358:
2355:
2349:
2346:
2345:
2325:
2321:
2315:
2307:
2303:
2299:
2295:
2288:
2280:
2276:
2271:
2266:
2262:
2258:
2254:
2250:
2246:
2239:
2231:
2227:
2222:
2217:
2213:
2209:
2205:
2198:
2190:
2186:
2181:
2176:
2172:
2168:
2164:
2160:
2156:
2149:
2141:
2137:
2132:
2127:
2122:
2117:
2113:
2109:
2105:
2098:
2090:
2086:
2081:
2076:
2072:
2066:
2062:
2058:
2054:
2050:
2046:
2039:
2026:
2020:
2012:
2006:
1998:
1994:
1990:
1986:
1983:(4): 1620–8.
1982:
1978:
1971:
1958:
1952:
1943:
1934:
1929:
1924:
1919:
1912:
1905:
1891:on 2017-11-18
1887:
1883:
1876:
1870:
1859:
1851:
1844:
1836:
1832:
1828:
1824:
1820:
1816:
1809:
1801:
1797:
1793:
1790:
1789:
1784:
1780:
1774:
1766:
1762:
1758:
1754:
1750:
1743:
1735:
1731:
1727:
1723:
1716:
1708:
1704:
1700:
1696:
1695:
1690:
1686:
1679:
1671:
1667:
1660:
1653:
1645:
1639:
1635:
1631:
1627:
1620:
1607:
1601:
1588:
1582:
1573:
1567:
1566:
1561:
1554:
1546:
1539:
1531:
1527:
1523:
1519:
1515:
1511:
1504:
1496:
1492:
1488:
1481:
1473:
1469:
1464:
1459:
1455:
1451:
1447:
1443:
1439:
1432:
1418:
1414:
1408:
1406:
1404:
1395:
1394:
1388:
1383:
1377:
1375:
1368:
1364:
1361:
1356:
1354:
1352:
1343:
1337:
1333:
1329:
1325:
1321:
1317:
1311:
1307:
1297:
1294:
1292:
1289:
1287:
1284:
1282:
1279:
1278:
1277:
1275:
1271:
1267:
1264:
1263:propanedioate
1250:
1243:
1240:
1229:
1225:
1222:
1217:
1214:
1210:
1195:
1191:
1187:
1178:
1174:
1166:
1161:
1157:
1156:
1153:
1143:
1133:
1129:
1125:
1121:
1118:
1114:
1109:
1107:
1103:
1099:
1095:
1091:
1086:
1084:
1080:
1076:
1066:
1064:
1060:
1056:
1046:
1044:
1040:
1036:
1034:
1030:
1029:cinnamic acid
1025:
1022:
1019:
1016:
1011:
1009:
1005:
1001:
991:
989:
974:
948:
947:
946:
944:
940:
926:
922:
920:
916:
907:
903:
901:
897:
893:
889:
885:
852:
851:
850:
848:
838:
836:
832:
822:
820:
816:
808:
804:
800:
796:
792:
788:
784:
781:derivatives.
780:
776:
766:
764:
760:
756:
752:
748:
744:
741:group can be
740:
736:
732:
728:
724:
720:
713:
708:
704:
702:
698:
694:
684:
682:
678:
673:
671:
667:
657:
655:
651:
647:
644:
640:
636:
632:
628:
624:
612:
608:
596:
590:
584:
581:
580:External MSDS
578:
575:
572:
571:
566:
563:
559:
556:
551:
550:
547:
543:
542:Succinic acid
539:
535:
532:
529:
524:
523:
520:
517:
514:
509:
508:
503:
499:
495:
491:
490:
476:
470:
466:
463:
462:
458:
455:
451:
450:
446:
444:
443:Boiling point
441:
440:
436:
434:
433:Melting point
431:
430:
426:
424:
421:
420:
413:
411:
408:
407:
386:
383:
379:
378:
373:
364:
361:O=C(O)CC(O)=O
359:
358:
355:
348:
334:
324:
323:
320:
313:
305:
301:
300:DTXSID7021659
297:
296:
294:
284:
280:
279:
272:
268:
267:
265:
263:
260:
259:
252:
248:
247:
245:
239:
235:
234:
230:
226:
223:
221:
219:ECHA InfoCard
216:
215:
208:
204:
203:
201:
199:
196:
195:
188:
184:
183:
181:
179:
176:
175:
168:
164:
163:
161:
159:
156:
155:
148:
144:
143:
141:
139:
136:
135:
128:
124:
121:
117:
116:
114:
110:
105:
104:
97:
93:
92:
90:
87:
83:
82:
77:
69:
61:
57:
52:
48:
43:
39:
34:
31:Malonic acid
23:
19:
2414:
2328:. Retrieved
2326:. 2020-03-13
2323:
2314:
2297:
2293:
2287:
2252:
2248:
2238:
2211:
2207:
2197:
2162:
2158:
2148:
2111:
2107:
2097:
2052:
2049:JIMD Reports
2048:
2038:
2019:
2005:
1980:
1976:
1970:
1951:
1942:
1904:
1893:. Retrieved
1886:the original
1881:
1869:
1858:
1849:
1843:
1818:
1814:
1808:
1791:
1786:
1773:
1756:
1752:
1742:
1725:
1721:
1715:
1698:
1692:
1688:
1684:
1678:
1669:
1665:
1652:
1625:
1619:
1600:
1581:
1571:
1563:
1553:
1538:
1513:
1509:
1503:
1494:
1490:
1480:
1463:10400.1/2790
1445:
1441:
1431:
1420:. Retrieved
1416:
1391:
1319:
1310:
1262:
1212:
1170:
1149:
1110:
1087:
1072:
1069:Biochemistry
1052:
1041:
1037:
1026:
1023:
1020:
1012:
1008:alkyd resins
997:
994:Applications
970:
936:
912:
890:group of an
881:
844:
828:
772:
759:fermentation
717:
690:
674:
663:
653:
652:word μᾶλον (
634:
607:Malonic acid
606:
605:
546:Fumaric acid
468:
79:Identifiers
71:Other names
1794:: 689–697.
1759:: 1136–36.
1296:Malonyl-CoA
1128:active site
1079:malonyl-CoA
1015:electronics
534:Oxalic acid
447:decomposes
427:1.619 g/cm
375:Properties
225:100.005.003
147:CHEBI:30794
22:maleic acid
2330:2020-10-06
2025:US 3591676
1957:US 9790350
1895:2015-12-11
1788:Chem. Ber.
1672:: 285–300.
1643:0471740039
1558:Weiner N.
1422:2020-10-07
1302:References
1098:acetyl CoA
1090:coenzyme A
1085:(ACSF3).
1000:polyesters
743:hydrolyzed
681:malic acid
410:Molar mass
271:9KX7ZMG0MK
178:ChemSpider
167:ChEMBL7942
107:3D model (
86:CAS Number
18:malic acid
1472:0567-7572
1266:compounds
1132:succinate
1094:cytosolic
1049:Pathology
1033:valproate
945:instead:
939:anhydride
791:condenses
769:Reactions
635:malonates
2639:Category
2279:19873352
2230:18112108
2189:28479296
2140:21846720
2089:26915364
1997:22944425
1835:92816468
1497:: 76–79.
1363:Archived
1318:(2014).
1268:include
1213:malonate
1173:diprotic
892:aldehyde
888:carbonyl
874:O + CO
805:to form
797:to form
779:chloride
568:Hazards
526:Related
519:Malonate
496:(χ)
459:763 g/L
198:DrugBank
96:141-82-2
2270:2142566
2180:5482780
2131:3190830
2080:5110436
1779:Diels O
1518:Bibcode
1218:can be
1177:protons
1165:dianion
1115:of the
1043:Eastman
870:H + H
817:in the
815:synthon
803:acetone
763:glucose
739:nitrile
660:History
643:diethyl
623:ionized
487:= 5.69
481:= 2.83
465:Acidity
423:Density
415:104.061
238:PubChem
207:DB02175
2390:C-R-CO
2277:
2267:
2228:
2187:
2177:
2138:
2128:
2087:
2077:
2067:
2031:
1995:
1963:
1933:926125
1930:
1882:Inchem
1833:
1728:: 92.
1640:
1612:
1593:
1470:
1338:
1274:esters
1117:enzyme
896:ketone
737:. The
733:via a
666:citrus
627:esters
621:. The
617:(COOH)
576:(SDS)
513:anions
511:Other
354:SMILES
158:ChEMBL
54:Names
1914:(PDF)
1889:(PDF)
1878:(PDF)
1831:S2CID
1701:: 1.
1689:trans
1662:(PDF)
1270:salts
813:COOH
793:with
775:amide
745:with
654:malon
650:Greek
646:ester
631:salts
609:is a
319:InChI
138:ChEBI
109:JSmol
2275:PMID
2226:PMID
2185:PMID
2136:PMID
2085:PMID
2065:ISBN
1993:PMID
1928:OSTI
1638:ISBN
1468:ISSN
1336:ISBN
1272:and
795:urea
723:salt
629:and
262:UNII
2475:(CO
2457:(CO
2439:(CO
2421:(CO
2386:(HO
2302:doi
2265:PMC
2257:doi
2216:doi
2212:178
2175:PMC
2167:doi
2126:PMC
2116:doi
2112:286
2075:PMC
2057:doi
1985:doi
1918:doi
1823:doi
1796:doi
1761:doi
1730:doi
1703:doi
1630:doi
1526:doi
1514:521
1458:hdl
1450:doi
1446:933
1328:doi
1236:or
1234:COO
1216:ion
1081:by
954:(CO
894:or
858:(CO
761:of
753:or
288:EPA
251:867
241:CID
187:844
20:or
2641::
2610:42
2606:22
2593:30
2589:16
2576:24
2572:13
2559:22
2555:12
2542:18
2538:10
2525:16
2508:14
2491:12
2479:H)
2461:H)
2443:H)
2425:H)
2411:H)
2394:H)
2322:.
2298:92
2296:.
2273:.
2263:.
2253:26
2251:.
2247:.
2224:.
2210:.
2206:.
2183:.
2173:.
2163:24
2161:.
2157:.
2134:.
2124:.
2110:.
2106:.
2083:.
2073:.
2063:.
2053:30
2051:.
2047:.
1991:.
1981:90
1979:.
1926:.
1880:.
1829:.
1819:11
1817:.
1792:39
1785:.
1757:35
1755:.
1751:.
1726:24
1724:.
1699:59
1697:.
1668:.
1664:.
1636:.
1628:.
1569:;
1562:.
1524:.
1512:.
1495:47
1493:.
1489:.
1466:.
1456:.
1444:.
1440:.
1415:.
1402:^
1390:.
1373:^
1350:^
1334:.
1322:.
1254:2−
1251:O)
1247:(C
1230:CH
1138:CH
1065:.
1035:.
983:10
958:H)
950:CH
921:.
902:.
862:H)
854:CH
837:.
821:.
765:.
703::
683:.
485:a2
483:pK
479:a1
477:pK
474:)
467:(p
2614:4
2612:O
2608:H
2597:4
2595:O
2591:H
2580:4
2578:O
2574:H
2563:4
2561:O
2557:H
2553:C
2546:4
2544:O
2540:H
2529:4
2527:O
2523:H
2521:9
2512:4
2510:O
2506:H
2504:8
2495:4
2493:O
2489:H
2487:7
2477:2
2473:8
2471:H
2469:4
2459:2
2455:6
2453:H
2451:3
2441:2
2437:4
2435:H
2433:2
2423:2
2419:2
2409:2
2392:2
2388:2
2376:e
2369:t
2362:v
2333:.
2308:.
2304::
2281:.
2259::
2232:.
2218::
2191:.
2169::
2142:.
2118::
2091:.
2059::
1999:.
1987::
1936:.
1920::
1898:.
1852:.
1837:.
1825::
1802:.
1798::
1767:.
1763::
1736:.
1732::
1710:.
1705::
1685:N
1670:1
1646:.
1632::
1576:.
1547:.
1532:.
1528::
1520::
1474:.
1460::
1452::
1425:.
1344:.
1330::
1257:2
1249:O
1245:2
1242:H
1239:C
1232:2
1228:C
1226:O
1224:O
1221:H
1196:a
1192:K
1188:p
1167:.
1154:.
1140:2
1136:2
981:O
979:4
977:P
975:(
966:O
964:2
960:2
956:2
952:2
876:2
872:2
868:2
864:2
860:2
856:2
811:2
714:.
619:2
615:2
472:a
469:K
403:4
400:O
397:4
394:H
391:3
388:C
290:)
286:(
111:)
24:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.