729:, it forms the basis of all modern computational approaches to modeling electron transfer. Its essential feature is that electron transfer can never be regarded as an “instantaneous transition”; instead, the electron is partially transferred at all molecular geometries, with the extent of the transfer being a critical quantum descriptor of all thermal, tunneling, and spectroscopic processes. It also leads seamlessly to understanding electron-transfer transition-state spectroscopy pioneered by
1144:. In that work, he also derived the standard expression for the solvent contribution to the reorganization energy, making the theory more applicable to practical problems. Use of this solvation description (instead of the form that Hush originally proposed) in approaches spanning the adiabatic and non-adiabatic limits is often termed “Marcus-Hush Theory”. These and other contributions, including the widespread demonstration of the usefulness of Eqn. (3), led to the award of the
277:
105:
78:
777:
is of central importance. In the very strong coupling limit when Eqn. (2) is satisfied, intrinsically quantum molecules like the Creutz-Taube ion result. Most intervalence spectroscopy occurs in the weak-coupling limit described by Eqn. (1), however. In both natural photosynthesis and in artificial
95:
apart, either through collisions, covalent bonding, location in a material, protein or polymer structure, etc. A and D have different chemical environments. Each polarizes their surrounding condensed media. Electron-transfer theories describe the influence of a variety of parameters on the rate of
1073:
This approach is widely applicable to long-range ground-state intramolecular electron transfer, electron transfer in biology, and electron transfer in conducting materials. It also typically controls the rate of charge separation in the excited-state photochemical application described in Figure 2
100:
occur by this mechanism. Adiabatic electron-transfer theory stresses that intricately coupled to such charge transfer is the ability of any D-A system to absorb or emit light. Hence fundamental understanding of any electrochemical process demands simultaneous understanding of the optical processes
85:
that may be found in many forms in both condensed phases and the gas phase. Internal structure, external structure, or chance collisions provide interconnection between the species. Upon electron transfer, the structure of the local chemical environments involving D and A change, as does the
1067:
720:
approach to charge-transfer and indeed general chemical reactions applied by Hush using parabolic potential-energy surfaces. Hush himself has carried out many theoretical and experimental studies of mixed valence complexes and long range electron transfer in biological systems. Hush's
873:
at which charge transfer occurs- the coupling typically decreases exponentially with distance. When electron transfer occurs during collisions of the D and A species, the coupling is typically large and the “adiabatic” limit applies in which rate constants are given by
193:
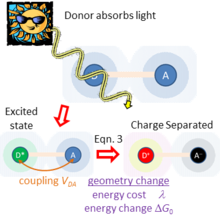
Figure 2 sketches what happens if light is absorbed by just one of the chemical species, taken to be the charge donor. This produces an excited state of the donor. As the donor and acceptor are close to each other and surrounding matter, they experience a coupling
108:
Fig. 2. When the donor species absorbs light energy, it goes into a high-energy excited state, generating significant changes to its local chemical environment and the polarization of its external environment. These environments facilitate coupling
650:
is not small: charge is not localized on just one chemical species but is shared quantum mechanically between two Ru centers, presenting classically forbidden half-integral valence states. that the critical requirement for this phenomenon is
903:
365:
Adiabatic electron-transfer is also relevant to the area of solar energy harvesting. Here, light absorption directly leads to charge separation D-A. Hush's theory for this process considers the donor-acceptor coupling
591:
90:
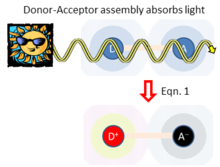
Figure 1 sketches the basic elements of adiabatic electron-transfer theory. Two chemical species (ions, molecules, polymers, protein cofactors, etc.) labelled D (for “donor”) and A (for “acceptor”) become a distance
1262:
280:
Fig. 3. Light energy is absorbed by the donor and acceptor, initiating intervalence charge transfer to directly convert solar energy into electrical energy as D-A. In the weak-coupling limit, the coupling
2180:
Reimers, J. R.; McKemmish, L.; McKenzie, R. H.; Hush, N. S. (2015). "A unified diabatic description for electron transfer reactions, isomerization reactions, proton transfer reactions, and aromaticity".
710:
1316:
Warman, J. M.; Haas, M. P. d.; Paddon-Row, M. N.; Cotsaris, E.; Hush, N. S.; Oevering, H.; Verhoeven, J. W. (1986). "Light-induced giant dipoles in simple model compounds for photosynthesis".
503:
1483:
Nelsen, S. F.; Weaver, M. N.; Luo, Y.; Lockard, J. V.; Zink, J. I. (2006). "Use of the neighboring orbital model for analysis of electronic coupling in Class III intervalence compounds".
139:
between the donor and acceptor, which drives photochemical charge separation with a rate given by Eqn. (3) in the weak-coupling limit. This rate is also dependent on the energy
817:
775:
416:
required to rearrange the atoms from their initial geometry to the preferred local geometry and environment polarization of the charge-separated state, and the energy change
1142:
1446:"Intervalence (Charge-Resonance) Transitions in Organic Mixed-Valence Systems. Through-Space versus Through-Bond Electron Transfer between Bridged Aromatic (Redox) Centers"
444:
359:
252:
187:
1106:
837:
414:
329:
157:
867:
648:
394:
309:
222:
137:
1158:. In particular, this reconnects adiabatic electron-transfer theory with its roots in proton-transfer theory and hydrogen-atom transfer, leading back to
1518:
Rosokha, S. V.; Kochi, J. K. (2008). "Fresh Look at
Electron-Transfer Mechanisms via the Donor/Acceptor Bindings in the Critical Encounter Complex".
159:
required to rearrange the atoms to the preferred local geometry and environment polarization of the charge-separated state D-A and the energy change
1062:{\displaystyle k={\frac {2\pi V_{DA}^{2}}{\hbar (4\pi \lambda k_{\beta }T)^{1/2}}}\exp {\frac {-(\Delta G_{0}+\lambda )^{2}}{4\lambda k_{\beta }T}}}
515:
885:
In the weak-coupling (“non-adiabatic”) limit, the activation energy for electron transfer is given by the expression derived independently by
1885:
Cave, R. J.; Newton, M. D. (1996). "Generalization of the
Mulliken-Hush treatment for the calculation of electron transfer matrix elements".
882:
is externally constrained and so the coupling set at low or high values. In these situations, weak-coupling scenarios often become critical.
612:, which codifies types of mixed valence compounds. An iconic system for understanding Inner sphere electron transfer is the mixed-valence
1179:
Piechota, Eric J.; Meyer, Gerald J. (2019). "Introduction to
Electron Transfer: Theoretical Foundations and Pedagogical Examples".
51:. Adiabatic electron-transfer can occur by either optical or thermal mechanisms. Electron transfer during a collision between an
1967:"Application of the Method of Generating Function to Radiative and Non-Radiative Transitions of a Trapped Electron in a Crystal"
257:
producing charged species. In this way, solar energy is captured and converted to electrical energy. This process is typical of
1823:
1692:
1920:
Reimers, J. R.; Hush, N. S. (2017). "Relating transition-state spectroscopy to standard chemical spectroscopic processes".
657:
1795:
1409:
German, E. D. (1979). "Intramolecular intervalence charge transfer in bimolecular mixed-valence complexes of metals".
1570:
1246:
449:
1145:
2025:
Levich, V. G.; Dogonadze, R. R. (1959). "Theory of rediationless electron transitions between ions in solution".
2114:
Efrima, S.; Bixon, M. (1976). "Vibrational effects in outer-sphere electron-transfer reactions in polar media".
609:
269:
solar-energy capture devices. The inverse of this process is also used to make organic light-emitting diodes (
1711:
Hush, N. S. (1953). "Quantum-mechanical discussion of the gas phase formation of quinonedimethide monomers".
1637:
Hush, N. S. (1975). "Inequivalent XPS binding energies in symmetrical delocalized mixed-valence complexes".
1360:
2262:
602:
897:
then determined the electron-tunneling probability to express the rate constant for thermal reactions as
2257:
2006:
Levich, V. G.; Dogonadze, R. R. (1960). "Adiabatic theory for electron-transfer processes in solution".
1152:
878:. In biological applications, however, as well as some organic conductors and other device materials,
781:
739:
266:
1111:
839:
through use of large molecules like chlorophylls, pentacenes, and conjugated polymers. The coupling
721:
quantum-electronic adiabatic approach to electron transfer was unique; directly connecting with the
613:
60:
1588:
Philosophical
Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences
419:
334:
227:
162:
81:
Fig. 1. Electron transfer occurs between donor (D) and acceptor (A) species separated by distance
875:
505:), Hush showed that the rate of light absorption (and hence charge separation) is given from the
1289:
Hush, N. S. (1961). "Adiabatic theory of outer sphere electron-transfer reactions in solution".
1083:
822:
399:
314:
142:
1155:
506:
2095:
2045:
842:
623:
369:
284:
197:
112:
2190:
2123:
2060:
1978:
1929:
1894:
1760:
1720:
1646:
1595:
1492:
1418:
1375:
1325:
1188:
8:
1586:
Day, P.; Hush, N. S.; Clark, R. J. H. (2008). "Mixed valence: origins and developments".
36:
2194:
2127:
2064:
1982:
1933:
1898:
1764:
1724:
1650:
1599:
1496:
1422:
1379:
1329:
1315:
1192:
2076:
1867:
1619:
1341:
1231:
Intervalence-transfer absorption. II. Theoretical considerations and spectroscopic data
1204:
726:
262:
32:
2237:
1562:
1557:. Advances in Inorganic Chemistry and Radiochemistry. Vol. 10. pp. 247–422.
2206:
2179:
2166:
2135:
1906:
1859:
1819:
1791:
1688:
1658:
1611:
1566:
1535:
1465:
1430:
1391:
1242:
1208:
1077:
722:
44:
2080:
1871:
2233:
2198:
2162:
2131:
2068:
1986:
1945:
1937:
1902:
1851:
1768:
1728:
1680:
1679:. World Scientific Series in 20th Century Chemistry. Vol. 74. pp. 32–60.
1654:
1623:
1603:
1558:
1527:
1504:
1500:
1457:
1445:
1443:
1426:
1383:
1345:
1333:
1298:
1234:
1196:
97:
889:
and
Toyozawa and by Hush. Using adiabatic electron-transfer theory, in this limit
254:
is favorable, this coupling facilitates primary charge separation to produce D-A ,
74:
is often credited with formulation of the theory of adiabatic electron-transfer.
1941:
1387:
894:
890:
52:
1200:
361:
control the rate of light absorption (and hence charge separation) via Eqn. (1).
2046:"On the Theory of Oxidation-Reduction Reactions Involving Electron Transfer. 1"
1732:
1684:
1482:
258:
56:
1855:
1238:
2251:
598:
48:
2224:
Horiuti, J.; Polanyi, M. (2003). "Outlines of a theory of proton transfer".
1361:"Adiabatic Electron Transfer: Comparison of Modified Theory with Experiment"
2210:
1842:
Devault, D. (1980). "Quantum mechanical tunnelling in biological systems".
1615:
1607:
1539:
1469:
1358:
1159:
730:
717:
40:
1863:
1395:
586:{\displaystyle k\propto {\frac {V_{DA}^{2}R^{2}}{\lambda +\Delta G_{0}}}.}
1816:
Electron
Transfer in Chemistry and Biology: An introduction to the theory
1302:
1950:
1675:
London, F. (1932). "On the theory of non-adiabatic chemical reactions".
255:
2202:
1991:
1966:
886:
2072:
1772:
1531:
1461:
1337:
71:
20:
2150:
1785:
617:
1444:
Sun, D. L.; Rosokha, S. V.; Lindeman, S. V.; Kochi, J. K. (2003).
1233:. Progress in Inorganic Chemistry. Vol. 8. pp. 391–444.
616:, wherein otherwise equivalent Ru(III) and Ru(II) are linked by a
276:
104:
77:
1585:
446:
associated with charge separation. In the weak-coupling limit (
2155:
Biochimica et
Biophysica Acta (BBA) - Reviews on Bioenergetics
1751:
Hush, N. S. (1958). "Adiabatic rate processes at electrodes".
2024:
2005:
28:
270:
86:
polarization these species induce on any surrounding media.
1517:
1151:
Adiabatic electron-transfer theory is also widely applied
1919:
1080:
showed that the activation energy in Eqn. (3) reduces to
1884:
1813:
1359:
Nelsen, S. F.; Ismagilov, R. F.; Trieber, D. A. (1997).
2148:
47:
electron-transfers proceed without making or breaking
1114:
1086:
906:
845:
825:
784:
742:
660:
626:
518:
452:
422:
402:
372:
337:
317:
287:
230:
200:
165:
145:
115:
1964:
705:{\displaystyle {\frac {2|J_{DA}|}{\lambda }}\geq 1.}
608:
Adiabatic electron transfer is also relevant to the
2113:
1555:
Mixed
Valence Chemistry-A Survey and Classification
1552:
1136:
1100:
1061:
861:
831:
811:
769:
704:
642:
585:
497:
438:
408:
388:
353:
323:
303:
246:
216:
181:
151:
131:
736:In adiabatic electron-transfer theory, the ratio
2249:
1786:Kornyshev, A. A.; Tosi, M.; Ulstrup, J. (1997).
16:Chemical reaction mechanism for redox reactions
2223:
869:can be controlled by controlling the distance
716:Adiabatic electron-transfer theory stems from
2151:"Electron transfers in chemistry and biology"
2043:
1408:
1178:
498:{\displaystyle 4V_{DA}^{2}/\lambda ^{2}\ll 1}
2097:Electron transfer and single molecule events
2018:
1999:
1788:Electron and Ion Transfer in Condensed Media
1841:
1750:
1710:
1636:
2226:Journal of Molecular Catalysis A: Chemical
2093:
2087:
1674:
1990:
1949:
603:intervalence charge transfer spectroscopy
1450:Journal of the American Chemical Society
1309:
1108:in the case of symmetric reactions with
275:
103:
76:
2173:
1809:
1807:
601:absorbes light, creating the field of
2250:
1837:
1835:
1746:
1744:
1742:
1706:
1704:
1670:
1668:
1476:
1284:
1282:
1280:
1162:theory of general chemical reactions.
1779:
1579:
1437:
1224:
1222:
1220:
1218:
59:occurs adiabatically on a continuous
2217:
2142:
2107:
2037:
1958:
1913:
1878:
1804:
1739:
1546:
1511:
1352:
1288:
1228:
35:is ubiquitous in nature in both the
2183:Physical Chemistry Chemical Physics
1832:
1814:Kuznetsov, A.; Ulstrup, J. (1998).
1701:
1665:
1630:
1291:Transactions of the Faraday Society
1277:
1215:
13:
1402:
1115:
1006:
564:
423:
338:
231:
189:associated with charge separation.
166:
14:
2274:
2149:Marcus, R. A.; Sutin, N. (1985).
812:{\displaystyle 2V_{DA}/\lambda }
770:{\displaystyle 2V_{DA}/\lambda }
2053:The Journal of Chemical Physics
1971:Progress of Theoretical Physics
1965:Kubo, R.; Toyozawa, Y. (1955).
1844:Quarterly Reviews of Biophysics
1790:. Singapore: World Scientific.
1753:The Journal of Chemical Physics
610:Robin-Day classification system
1553:Robin, M. B.; Day, P. (1967).
1505:10.1016/j.chemphys.2006.01.023
1255:
1172:
1137:{\displaystyle \Delta G_{0}=0}
1026:
1003:
971:
945:
778:solar-energy capture devices,
686:
668:
1:
2238:10.1016/s1381-1169(03)00034-7
1563:10.1016/S0065-2792(08)60179-X
1520:Accounts of Chemical Research
1181:Journal of Chemical Education
1165:
1146:1992 Nobel Prize in Chemistry
331:, and the free energy change
101:that the system can undergo.
2167:10.1016/0304-4173(85)90014-x
2136:10.1016/0301-0104(76)87014-0
1942:10.1016/j.cplett.2017.04.070
1907:10.1016/0009-2614(95)01310-5
1659:10.1016/0301-0104(75)87049-2
1431:10.1016/0009-2614(79)80516-3
1388:10.1126/science.278.5339.846
439:{\displaystyle \Delta G_{0}}
354:{\displaystyle \Delta G_{0}}
247:{\displaystyle \Delta G_{0}}
224:. If the free energy change
182:{\displaystyle \Delta G_{0}}
7:
1201:10.1021/acs.jchemed.9b00489
819:is maximized by minimizing
29:oxidation-reduction process
25:adiabatic electron-transfer
10:
2279:
2103:. Paris: Eolss Publishers.
1733:10.1002/pol.1953.120110401
1713:Journal of Polymer Science
1685:10.1142/9789812795762_0003
1101:{\displaystyle \lambda /4}
597:This theory explained how
66:
1856:10.1017/S003358350000175X
1239:10.1002/9780470166093.ch7
267:artificial photosynthesis
98:electrochemical reactions
1922:Chemical Physics Letters
1887:Chemical Physics Letters
1411:Chemical Physics Letters
832:{\displaystyle \lambda }
409:{\displaystyle \lambda }
324:{\displaystyle \lambda }
311:, reorganization energy
152:{\displaystyle \lambda }
61:potential energy surface
2027:Proc. Akad. Naukl. SSSR
2008:Proc. Akad. Naukl. SSSR
876:transition state theory
96:electron-transfer. All
2044:Marcus, R. A. (1956).
1608:10.1098/rsta.2007.2135
1138:
1102:
1074:and related problems.
1063:
863:
862:{\displaystyle V_{DA}}
833:
813:
771:
706:
644:
643:{\displaystyle V_{DA}}
587:
499:
440:
410:
390:
389:{\displaystyle V_{DA}}
362:
355:
325:
305:
304:{\displaystyle V_{DA}}
259:natural photosynthesis
248:
218:
217:{\displaystyle V_{DA}}
190:
183:
153:
133:
132:{\displaystyle V_{DA}}
87:
1818:. Chichester: Wiley.
1156:Molecular Electronics
1139:
1103:
1064:
864:
834:
814:
772:
707:
645:
588:
500:
441:
411:
391:
356:
326:
306:
279:
249:
219:
184:
154:
134:
107:
80:
1303:10.1039/TF9615700557
1229:Hush, N. S. (1967).
1112:
1084:
904:
843:
823:
782:
740:
658:
624:
516:
450:
420:
400:
370:
335:
315:
285:
263:organic photovoltaic
228:
198:
163:
143:
113:
2263:Reaction mechanisms
2195:2015PCCP...1724598R
2189:(38): 24598–24617.
2128:1976CP.....13..447E
2065:1956JChPh..24..966M
1983:1955PThPh..13..160K
1934:2017CPL...683..467R
1899:1996CPL...249...15C
1765:1958JChPh..28..962H
1725:1953JPoSc..11..289H
1651:1975CP.....10..361H
1600:2008RSPTA.366....5D
1497:2006CP....324..195N
1456:(51): 15950–15963.
1423:1979CPL....64..295G
1380:1997Sci...278..846N
1330:1986Natur.320..615W
1193:2019JChEd..96.2450P
939:
545:
473:
2258:Physical chemistry
2203:10.1039/C5CP02236C
1992:10.1143/PTP.13.160
1134:
1098:
1059:
922:
859:
829:
809:
767:
702:
640:
583:
528:
495:
456:
436:
406:
386:
363:
351:
321:
301:
261:as well as modern
244:
214:
191:
179:
149:
129:
88:
2073:10.1063/1.1742723
1825:978-0-471-96749-1
1773:10.1063/1.1744305
1694:978-981-02-2771-5
1677:Quantum Chemistry
1532:10.1021/ar700256a
1462:10.1021/ja037867s
1374:(5339): 846–849.
1324:(6063): 615–616.
1263:"Fellows Details"
1187:(11): 2450–2466.
1057:
989:
723:Quantum Chemistry
694:
578:
507:Einstein equation
2270:
2242:
2241:
2232:(1–2): 185–197.
2221:
2215:
2214:
2177:
2171:
2170:
2146:
2140:
2139:
2116:Chemical Physics
2111:
2105:
2104:
2102:
2091:
2085:
2084:
2050:
2041:
2035:
2034:
2022:
2016:
2015:
2003:
1997:
1996:
1994:
1962:
1956:
1955:
1953:
1917:
1911:
1910:
1882:
1876:
1875:
1839:
1830:
1829:
1811:
1802:
1801:
1783:
1777:
1776:
1748:
1737:
1736:
1708:
1699:
1698:
1672:
1663:
1662:
1645:(2–3): 361–366.
1639:Chemical Physics
1634:
1628:
1627:
1583:
1577:
1576:
1550:
1544:
1543:
1515:
1509:
1508:
1485:Chemical Physics
1480:
1474:
1473:
1441:
1435:
1434:
1406:
1400:
1399:
1365:
1356:
1350:
1349:
1338:10.1038/320615a0
1313:
1307:
1306:
1286:
1275:
1274:
1272:
1270:
1259:
1253:
1252:
1226:
1213:
1212:
1176:
1143:
1141:
1140:
1135:
1127:
1126:
1107:
1105:
1104:
1099:
1094:
1068:
1066:
1065:
1060:
1058:
1056:
1052:
1051:
1035:
1034:
1033:
1018:
1017:
998:
990:
988:
987:
986:
982:
966:
965:
940:
938:
933:
914:
868:
866:
865:
860:
858:
857:
838:
836:
835:
830:
818:
816:
815:
810:
805:
800:
799:
776:
774:
773:
768:
763:
758:
757:
711:
709:
708:
703:
695:
690:
689:
684:
683:
671:
662:
649:
647:
646:
641:
639:
638:
614:Creutz-Taube ion
592:
590:
589:
584:
579:
577:
576:
575:
556:
555:
554:
544:
539:
526:
504:
502:
501:
496:
488:
487:
478:
472:
467:
445:
443:
442:
437:
435:
434:
415:
413:
412:
407:
395:
393:
392:
387:
385:
384:
360:
358:
357:
352:
350:
349:
330:
328:
327:
322:
310:
308:
307:
302:
300:
299:
253:
251:
250:
245:
243:
242:
223:
221:
220:
215:
213:
212:
188:
186:
185:
180:
178:
177:
158:
156:
155:
150:
138:
136:
135:
130:
128:
127:
2278:
2277:
2273:
2272:
2271:
2269:
2268:
2267:
2248:
2247:
2246:
2245:
2222:
2218:
2178:
2174:
2147:
2143:
2112:
2108:
2100:
2094:Schmickler, W.
2092:
2088:
2048:
2042:
2038:
2023:
2019:
2004:
2000:
1963:
1959:
1918:
1914:
1883:
1879:
1840:
1833:
1826:
1812:
1805:
1798:
1784:
1780:
1749:
1740:
1709:
1702:
1695:
1673:
1666:
1635:
1631:
1584:
1580:
1573:
1551:
1547:
1516:
1512:
1481:
1477:
1442:
1438:
1407:
1403:
1363:
1357:
1353:
1314:
1310:
1287:
1278:
1268:
1266:
1265:. Royal Society
1261:
1260:
1256:
1249:
1227:
1216:
1177:
1173:
1168:
1122:
1118:
1113:
1110:
1109:
1090:
1085:
1082:
1081:
1047:
1043:
1036:
1029:
1025:
1013:
1009:
999:
997:
978:
974:
970:
961:
957:
941:
934:
926:
915:
913:
905:
902:
901:
850:
846:
844:
841:
840:
824:
821:
820:
801:
792:
788:
783:
780:
779:
759:
750:
746:
741:
738:
737:
685:
676:
672:
667:
663:
661:
659:
656:
655:
631:
627:
625:
622:
621:
620:. The coupling
571:
567:
557:
550:
546:
540:
532:
527:
525:
517:
514:
513:
483:
479:
474:
468:
460:
451:
448:
447:
430:
426:
421:
418:
417:
401:
398:
397:
377:
373:
371:
368:
367:
345:
341:
336:
333:
332:
316:
313:
312:
292:
288:
286:
283:
282:
238:
234:
229:
226:
225:
205:
201:
199:
196:
195:
173:
169:
164:
161:
160:
144:
141:
140:
120:
116:
114:
111:
110:
69:
17:
12:
11:
5:
2276:
2266:
2265:
2260:
2244:
2243:
2216:
2172:
2161:(3): 265–322.
2141:
2122:(4): 447–460.
2106:
2086:
2059:(5): 966–978.
2036:
2017:
1998:
1977:(2): 160–182.
1957:
1912:
1893:(1–2): 15–19.
1877:
1850:(4): 387–564.
1831:
1824:
1803:
1797:978-9810229290
1796:
1778:
1759:(5): 962–972.
1738:
1719:(4): 289–298.
1700:
1693:
1664:
1629:
1594:(1862): 5–14.
1578:
1571:
1545:
1526:(5): 641–653.
1510:
1491:(1): 195–201.
1475:
1436:
1417:(2): 295–298.
1401:
1351:
1308:
1276:
1254:
1247:
1214:
1170:
1169:
1167:
1164:
1133:
1130:
1125:
1121:
1117:
1097:
1093:
1089:
1071:
1070:
1055:
1050:
1046:
1042:
1039:
1032:
1028:
1024:
1021:
1016:
1012:
1008:
1005:
1002:
996:
993:
985:
981:
977:
973:
969:
964:
960:
956:
953:
950:
947:
944:
937:
932:
929:
925:
921:
918:
912:
909:
856:
853:
849:
828:
808:
804:
798:
795:
791:
787:
766:
762:
756:
753:
749:
745:
714:
713:
701:
698:
693:
688:
682:
679:
675:
670:
666:
637:
634:
630:
595:
594:
582:
574:
570:
566:
563:
560:
553:
549:
543:
538:
535:
531:
524:
521:
494:
491:
486:
482:
477:
471:
466:
463:
459:
455:
433:
429:
425:
405:
383:
380:
376:
348:
344:
340:
320:
298:
295:
291:
241:
237:
233:
211:
208:
204:
176:
172:
168:
148:
126:
123:
119:
68:
65:
49:chemical bonds
15:
9:
6:
4:
3:
2:
2275:
2264:
2261:
2259:
2256:
2255:
2253:
2239:
2235:
2231:
2227:
2220:
2212:
2208:
2204:
2200:
2196:
2192:
2188:
2184:
2176:
2168:
2164:
2160:
2156:
2152:
2145:
2137:
2133:
2129:
2125:
2121:
2117:
2110:
2099:
2098:
2090:
2082:
2078:
2074:
2070:
2066:
2062:
2058:
2054:
2047:
2040:
2032:
2028:
2021:
2013:
2009:
2002:
1993:
1988:
1984:
1980:
1976:
1972:
1968:
1961:
1952:
1947:
1943:
1939:
1935:
1931:
1927:
1923:
1916:
1908:
1904:
1900:
1896:
1892:
1888:
1881:
1873:
1869:
1865:
1861:
1857:
1853:
1849:
1845:
1838:
1836:
1827:
1821:
1817:
1810:
1808:
1799:
1793:
1789:
1782:
1774:
1770:
1766:
1762:
1758:
1754:
1747:
1745:
1743:
1734:
1730:
1726:
1722:
1718:
1714:
1707:
1705:
1696:
1690:
1686:
1682:
1678:
1671:
1669:
1660:
1656:
1652:
1648:
1644:
1640:
1633:
1625:
1621:
1617:
1613:
1609:
1605:
1601:
1597:
1593:
1589:
1582:
1574:
1572:9780120236107
1568:
1564:
1560:
1556:
1549:
1541:
1537:
1533:
1529:
1525:
1521:
1514:
1506:
1502:
1498:
1494:
1490:
1486:
1479:
1471:
1467:
1463:
1459:
1455:
1451:
1447:
1440:
1432:
1428:
1424:
1420:
1416:
1412:
1405:
1397:
1393:
1389:
1385:
1381:
1377:
1373:
1369:
1362:
1355:
1347:
1343:
1339:
1335:
1331:
1327:
1323:
1319:
1312:
1304:
1300:
1296:
1292:
1285:
1283:
1281:
1264:
1258:
1250:
1248:9780470166093
1244:
1240:
1236:
1232:
1225:
1223:
1221:
1219:
1210:
1206:
1202:
1198:
1194:
1190:
1186:
1182:
1175:
1171:
1163:
1161:
1157:
1153:
1149:
1147:
1131:
1128:
1123:
1119:
1095:
1091:
1087:
1079:
1075:
1053:
1048:
1044:
1040:
1037:
1030:
1022:
1019:
1014:
1010:
1000:
994:
991:
983:
979:
975:
967:
962:
958:
954:
951:
948:
942:
935:
930:
927:
923:
919:
916:
910:
907:
900:
899:
898:
896:
892:
888:
883:
881:
877:
872:
854:
851:
847:
826:
806:
802:
796:
793:
789:
785:
764:
760:
754:
751:
747:
743:
734:
732:
728:
724:
719:
699:
696:
691:
680:
677:
673:
664:
654:
653:
652:
635:
632:
628:
619:
615:
611:
606:
604:
600:
599:Prussian blue
580:
572:
568:
561:
558:
551:
547:
541:
536:
533:
529:
522:
519:
512:
511:
510:
508:
492:
489:
484:
480:
475:
469:
464:
461:
457:
453:
431:
427:
403:
396:, the energy
381:
378:
374:
346:
342:
318:
296:
293:
289:
278:
274:
272:
268:
264:
260:
256:
239:
235:
209:
206:
202:
174:
170:
146:
124:
121:
117:
106:
102:
99:
94:
84:
79:
75:
73:
64:
62:
58:
54:
50:
46:
42:
38:
34:
30:
27:is a type of
26:
22:
2229:
2225:
2219:
2186:
2182:
2175:
2158:
2154:
2144:
2119:
2115:
2109:
2096:
2089:
2056:
2052:
2039:
2030:
2026:
2020:
2011:
2007:
2001:
1974:
1970:
1960:
1951:10453/125251
1925:
1921:
1915:
1890:
1886:
1880:
1847:
1843:
1815:
1787:
1781:
1756:
1752:
1716:
1712:
1676:
1642:
1638:
1632:
1591:
1587:
1581:
1554:
1548:
1523:
1519:
1513:
1488:
1484:
1478:
1453:
1449:
1439:
1414:
1410:
1404:
1371:
1367:
1354:
1321:
1317:
1311:
1294:
1290:
1269:18 September
1267:. Retrieved
1257:
1230:
1184:
1180:
1174:
1150:
1076:
1072:
884:
879:
870:
735:
725:concepts of
715:
607:
596:
364:
192:
92:
89:
82:
70:
24:
18:
1928:: 467–477.
1148:to Marcus.
2252:Categories
1166:References
41:biological
1209:208754569
1116:Δ
1088:λ
1049:β
1041:λ
1023:λ
1007:Δ
1001:−
995:
963:β
955:λ
952:π
943:ℏ
920:π
895:Dogonadze
827:λ
807:λ
765:λ
697:≥
692:λ
565:Δ
559:λ
523:∝
490:≪
481:λ
424:Δ
404:λ
339:Δ
319:λ
232:Δ
167:Δ
147:λ
72:Noel Hush
57:reductant
45:Adiabatic
43:spheres.
37:inorganic
33:mechanism
21:chemistry
2211:26193994
2081:16579694
1872:26771752
1616:17827130
1540:18380446
1470:14677987
1160:London's
727:Mulliken
718:London's
618:pyrazine
2191:Bibcode
2124:Bibcode
2061:Bibcode
1979:Bibcode
1930:Bibcode
1895:Bibcode
1864:7015406
1761:Bibcode
1721:Bibcode
1647:Bibcode
1624:5912503
1596:Bibcode
1493:Bibcode
1419:Bibcode
1396:9346480
1376:Bibcode
1368:Science
1346:4346663
1326:Bibcode
1297:: 577.
1189:Bibcode
1069:. … (3)
67:History
53:oxidant
31:. The
2209:
2079:
2014:: 591.
1870:
1862:
1822:
1794:
1691:
1622:
1614:
1569:
1538:
1468:
1394:
1344:
1318:Nature
1245:
1207:
1078:Marcus
891:Levich
731:Zewail
55:and a
2101:(PDF)
2077:S2CID
2049:(PDF)
1868:S2CID
1620:S2CID
1364:(PDF)
1342:S2CID
1205:S2CID
712:… (2)
593:… (1)
271:OLEDs
2207:PMID
2033:: 9.
1860:PMID
1820:ISBN
1792:ISBN
1689:ISBN
1612:PMID
1567:ISBN
1536:PMID
1466:PMID
1392:PMID
1271:2015
1243:ISBN
893:and
887:Kubo
265:and
39:and
2234:doi
2230:199
2199:doi
2163:doi
2159:811
2132:doi
2069:doi
2012:133
1987:doi
1946:hdl
1938:doi
1926:683
1903:doi
1891:249
1852:doi
1769:doi
1729:doi
1681:doi
1655:doi
1604:doi
1592:366
1559:doi
1528:doi
1501:doi
1489:324
1458:doi
1454:125
1427:doi
1384:doi
1372:278
1334:doi
1322:320
1299:doi
1235:doi
1197:doi
1154:in
992:exp
509:by
273:).
19:In
2254::
2228:.
2205:.
2197:.
2187:17
2185:.
2157:.
2153:.
2130:.
2120:13
2118:.
2075:.
2067:.
2057:24
2055:.
2051:.
2031:29
2029:.
2010:.
1985:.
1975:13
1973:.
1969:.
1944:.
1936:.
1924:.
1901:.
1889:.
1866:.
1858:.
1848:13
1846:.
1834:^
1806:^
1767:.
1757:28
1755:.
1741:^
1727:.
1717:11
1715:.
1703:^
1687:.
1667:^
1653:.
1643:10
1641:.
1618:.
1610:.
1602:.
1590:.
1565:.
1534:.
1524:41
1522:.
1499:.
1487:.
1464:.
1452:.
1448:.
1425:.
1415:64
1413:.
1390:.
1382:.
1370:.
1366:.
1340:.
1332:.
1320:.
1295:57
1293:.
1279:^
1241:.
1217:^
1203:.
1195:.
1185:96
1183:.
733:.
700:1.
605:.
63:.
23:,
2240:.
2236::
2213:.
2201::
2193::
2169:.
2165::
2138:.
2134::
2126::
2083:.
2071::
2063::
1995:.
1989::
1981::
1954:.
1948::
1940::
1932::
1909:.
1905::
1897::
1874:.
1854::
1828:.
1800:.
1775:.
1771::
1763::
1735:.
1731::
1723::
1697:.
1683::
1661:.
1657::
1649::
1626:.
1606::
1598::
1575:.
1561::
1542:.
1530::
1507:.
1503::
1495::
1472:.
1460::
1433:.
1429::
1421::
1398:.
1386::
1378::
1348:.
1336::
1328::
1305:.
1301::
1273:.
1251:.
1237::
1211:.
1199::
1191::
1132:0
1129:=
1124:0
1120:G
1096:4
1092:/
1054:T
1045:k
1038:4
1031:2
1027:)
1020:+
1015:0
1011:G
1004:(
984:2
980:/
976:1
972:)
968:T
959:k
949:4
946:(
936:2
931:A
928:D
924:V
917:2
911:=
908:k
880:R
871:R
855:A
852:D
848:V
803:/
797:A
794:D
790:V
786:2
761:/
755:A
752:D
748:V
744:2
687:|
681:A
678:D
674:J
669:|
665:2
636:A
633:D
629:V
581:.
573:0
569:G
562:+
552:2
548:R
542:2
537:A
534:D
530:V
520:k
493:1
485:2
476:/
470:2
465:A
462:D
458:V
454:4
432:0
428:G
382:A
379:D
375:V
347:0
343:G
297:A
294:D
290:V
240:0
236:G
210:A
207:D
203:V
175:0
171:G
125:A
122:D
118:V
93:R
83:R
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.