33:
410:
394:
382:
372:
402:
352:
362:
154:
494:
1022:
836:
1122:
of stars, which is primarily a determination of surface temperature, is based on the relative strength of spectral lines, and the Balmer series in particular is very important. Other characteristics of a star that can be determined by close analysis of its spectrum include
423:
Although physicists were aware of atomic emissions before 1885, they lacked a tool to accurately predict where the spectral lines should appear. The Balmer equation predicts the four visible spectral lines of hydrogen with high accuracy. Balmer's equation inspired the
669:
533:
were slightly inaccurate, and his formula also predicted lines that had not yet been observed but were found later. His number also proved to be the limit of the series. The Balmer equation could be used to find the
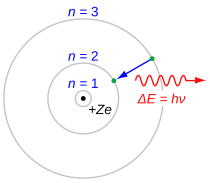
165:, the Balmer lines result from an electron jump between the second energy level closest to the nucleus, and those levels more distant. Shown here is a photon emission. The 3→2 transition depicted here produces
1017:{\displaystyle {\frac {1}{\lambda }}={\frac {4}{B}}\left({\frac {1}{2^{2}}}-{\frac {1}{n^{2}}}\right)=R_{\mathrm {H} }\left({\frac {1}{2^{2}}}-{\frac {1}{n^{2}}}\right)\quad \mathrm {for~} n=3,4,5,\dots }
134:
Balmer lines with wavelengths shorter than 400 nm. The series continues with an infinite number of lines whose wavelengths asymptotically approach the limit of 364.5 nm in the ultraviolet.
545:
1231:). H-epsilon is separated by 0.16 nm from Ca II H at 396.847 nm, and cannot be resolved in low-resolution spectra. The H-zeta line (transition 8→2) is similarly mixed in with a neutral
810:
756:
478:
Later, it was discovered that when the Balmer series lines of the hydrogen spectrum were examined at very high resolution, they were closely spaced doublets. This splitting is called
213: = 2 is called H-α, 4 to 2 is H-β, 5 to 2 is H-γ, and 6 to 2 is H-δ. As the first spectral lines associated with this series are located in the visible part of the
529:(see equation below) gave the wavelength of another line in the hydrogen spectrum. By this formula, he was able to show that some measurements of lines made in his time by
1391:
475:
found in star forming regions. In true-colour pictures, these nebula have a reddish-pink colour from the combination of visible Balmer lines that hydrogen emits.
819:
generalized the Balmer equation for all transitions of hydrogen. The equation commonly used to calculate the Balmer series is a specific example of the
1384:
1111:, and therefore are commonly seen and relatively strong compared to lines from other elements. The first two Balmer lines correspond to the
1219:
In stellar spectra, the H-epsilon line (transition 7→2, 397.007 nm) is often mixed in with another absorption line caused by ionized
1377:
538:
of the absorption/emission lines and was originally presented as follows (save for a notation change to give Balmer's constant as
1193:, the Balmer lines are usually seen in absorption, and they are "strongest" in stars with a surface temperature of about 10,000
1331:
Kramida, A., Ralchenko, Yu., Reader, J., and NIST ASD Team (2019). NIST Atomic
Spectra Database (ver. 5.7.1), . Available:
217:, these lines are historically referred to as "H-alpha", "H-beta", "H-gamma", and so on, where H is the element hydrogen.
521:. When any integer higher than 2 was squared and then divided by itself squared minus 4, then that number multiplied by
823:
and follows as a simple reciprocal mathematical rearrangement of the formula above (conventionally using a notation of
509:
Balmer noticed that a single wavelength had a relation to every line in the hydrogen spectrum that was in the visible
1436:
1426:
1421:
766:
712:
1431:
664:{\displaystyle \lambda \ =B\left({\frac {m^{2}}{m^{2}-n^{2}}}\right)=B\left({\frac {m^{2}}{m^{2}-2^{2}}}\right)}
1462:
1130:
Because the Balmer lines are commonly seen in the spectra of various objects, they are often used to determine
17:
1467:
1244:
1107:
because the Balmer lines appear in numerous stellar objects due to the abundance of hydrogen in the
1441:
1400:
1298:
1254:
214:
202:
138:
124:
73:
37:
1269:
1119:
451:
spectral line of the Balmer series of atomic hydrogen, which is the transition from the shell
1228:
198:
1290:
8:
1158:
around them), identifying groups of objects with similar motions and presumably origins (
112:
44:
is the red line at the right. Four lines (counting from the right) are formally in the
497:
Two of the Balmer lines (α and β) are clearly visible in this emission spectrum of a
1348:
1224:
1210:
1112:
1038:
816:
441:
425:
45:
1264:
1182:
1131:
1124:
820:
464:
437:
1138:
of the Balmer lines. This has important uses all over astronomy, from detecting
1167:
1155:
498:
479:
433:
377:
69:
32:
1456:
1202:
1198:
1186:
1135:
409:
393:
381:
371:
162:
115:, 434 nm, 486 nm, and 656 nm, that correspond to emissions of
89:
81:
77:
401:
1411:
1369:
1259:
1163:
1159:
1147:
530:
468:
429:
367:
357:
351:
1335:. National Institute of Standards and Technology, Gaithersburg, MD. DOI:
1206:
1139:
1059:
in Balmer's formula, and this value, for an infinitely heavy nucleus, is
472:
388:
205:
of the electron. The transitions are named sequentially by Greek letter:
131:
53:
1189:
lines in a spectrum, depending on the nature of the object observed. In
361:
1349:"CODATA Recommended Values of the Fundamental Physical Constants: 2006"
1249:
1151:
535:
428:
as a generalization of it, and this in turn led physicists to find the
174:
158:
141:
were discovered, corresponding to electrons transitioning to values of
108:
123:
in excited states transitioning to the quantum level described by the
1143:
1104:
440:, which predicted other spectral lines of hydrogen found outside the
85:
49:
1178:, and identifying unfamiliar objects by analysis of their spectrum.
490: = 2 shell, emitting shades of ultraviolet when doing so.
1171:
1108:
493:
460:
182:
120:
104:
96:
1336:
153:
1220:
448:
166:
41:
1332:
1232:
1213:
1194:
1175:
1170:, and debris from collisions), determining distances (actually
116:
84:. The Balmer series is calculated using the Balmer formula, an
510:
100:
482:. It was also found that excited electrons from shells with
1359:
1190:
1201:
A). In the spectra of most spiral and irregular galaxies,
1041:
for hydrogen. The
Rydberg constant is seen to be equal to
459: = 2, is one of the conspicuous colours of the
347:
173: = 1) this transition results in a photon of
1356:
Committee on Data for
Science and Technology (CODATA)
839:
769:
715:
548:
463:. It contributes a bright red line to the spectra of
169:, the first line of the Balmer series. For hydrogen (
1030:
is the wavelength of the absorbed/emitted light and
1016:
804:
750:
663:
1454:
1385:
1399:
1127:(related to physical size) and composition.
1103:The Balmer series is particularly useful in
805:{\displaystyle (n\in \mathbb {N} ,n\geq m).}
751:{\displaystyle (m\in \mathbb {N} ,m\geq 1).}
1392:
1378:
408:
400:
392:
380:
370:
360:
350:
181:The Balmer series is characterized by the
56:as they have wavelengths less than 400 nm.
48:. Lines five and six can be seen with the
831:as the single integral constant needed):
780:
726:
492:
152:
31:
1216:, the Balmer lines are emission lines.
14:
1455:
130:equals 2. There are several prominent
1373:
1341:
137:After Balmer's discovery, five other
1327:
1325:
1323:
1321:
1319:
1317:
1315:
1288:
1098:
504:
24:
980:
977:
974:
919:
25:
1479:
1312:
486:greater than 6 could jump to the
684:is a constant with the value of
38:hydrogen emission spectrum lines
1337:https://doi.org/10.18434/T4W30F
972:
467:or ionisation nebula, like the
1282:
1154:(by the motion of hydrogen in
796:
770:
742:
716:
13:
1:
1333:https://physics.nist.gov/asd
513:region. That wavelength was
455: = 3 to the shell
414:
406:
398:
386:
376:
366:
356:
346:
338:
335:
332:
329:
326:
323:
320:
317:
309:
306:
303:
300:
297:
294:
291:
288:
280:
277:
274:
271:
268:
265:
262:
259:
251:
248:
245:
242:
239:
236:
233:
230:
7:
1238:
1181:Balmer lines can appear as
148:
52:, but are considered to be
10:
1484:
1146:, compact objects such as
1407:
1245:Astronomical spectroscopy
1401:Hydrogen spectral series
1299:Georgia State University
1275:
1255:Hydrogen spectral series
1235:line seen in hot stars.
215:electromagnetic spectrum
203:principal quantum number
139:hydrogen spectral series
125:principal quantum number
27:Hydrogen spectral series
1120:spectral classification
315:Energy difference (eV)
88:equation discovered by
1270:Stellar classification
1203:active galactic nuclei
1018:
815:In 1888 the physicist
806:
752:
665:
501:
193: = 2, where
178:
57:
40:in the Balmer series.
1463:Emission spectroscopy
1229:Joseph von Fraunhofer
1019:
807:
753:
709:is the initial state
666:
496:
286:Wavelength (nm, air)
199:radial quantum number
159:Rutherford Bohr model
156:
72:, is one of a set of
35:
1289:Nave, C. R. (2006).
1225:original designation
837:
767:
713:
546:
1291:"Hydrogen Spectrum"
763:is the final state
185:transitioning from
1223:known as "H" (the
1014:
802:
748:
678:is the wavelength.
661:
502:
471:, which are often
209: = 3 to
189: ≥ 3 to
179:
177:656 nm (red).
157:In the simplified
58:
1450:
1449:
1174:) of galaxies or
1132:radial velocities
1099:Role in astronomy
985:
965:
945:
903:
883:
861:
848:
655:
602:
554:
419:
418:
145:other than two .
80:emissions of the
16:(Redirected from
1475:
1468:Hydrogen physics
1437:Humphreys series
1394:
1387:
1380:
1371:
1370:
1364:
1363:
1353:
1345:
1339:
1329:
1310:
1309:
1307:
1305:
1286:
1136:doppler shifting
1113:Fraunhofer lines
1094:
1092:
1089:
1083:
1081:
1080:
1079:
1077:
1074:
1068:
1065:
1058:
1056:
1055:
1050:
1047:
1039:Rydberg constant
1023:
1021:
1020:
1015:
986:
983:
971:
967:
966:
964:
963:
951:
946:
944:
943:
931:
924:
923:
922:
909:
905:
904:
902:
901:
889:
884:
882:
881:
869:
862:
854:
849:
841:
817:Johannes Rydberg
811:
809:
808:
803:
783:
757:
755:
754:
749:
729:
702:
700:
694:
692:
689:
670:
668:
667:
662:
660:
656:
654:
653:
652:
640:
639:
629:
628:
619:
607:
603:
601:
600:
599:
587:
586:
576:
575:
566:
552:
528:
526:
520:
518:
505:Balmer's formula
442:visible spectrum
426:Rydberg equation
412:
404:
396:
384:
374:
364:
354:
222:
221:
74:six named series
21:
1483:
1482:
1478:
1477:
1476:
1474:
1473:
1472:
1453:
1452:
1451:
1446:
1427:Brackett series
1403:
1398:
1368:
1367:
1351:
1347:
1346:
1342:
1330:
1313:
1303:
1301:
1287:
1283:
1278:
1265:Rydberg formula
1241:
1168:galaxy clusters
1156:accretion disks
1125:surface gravity
1101:
1090:
1087:
1085:
1075:
1072:
1070:
1069:
1066:
1063:
1062:
1060:
1051:
1048:
1045:
1044:
1042:
1036:
973:
959:
955:
950:
939:
935:
930:
929:
925:
918:
917:
913:
897:
893:
888:
877:
873:
868:
867:
863:
853:
840:
838:
835:
834:
821:Rydberg formula
779:
768:
765:
764:
725:
714:
711:
710:
698:
696:
690:
687:
685:
648:
644:
635:
631:
630:
624:
620:
618:
614:
595:
591:
582:
578:
577:
571:
567:
565:
561:
547:
544:
543:
524:
522:
516:
514:
507:
438:Brackett series
151:
76:describing the
28:
23:
22:
15:
12:
11:
5:
1481:
1471:
1470:
1465:
1448:
1447:
1445:
1444:
1439:
1434:
1429:
1424:
1422:Paschen series
1419:
1414:
1408:
1405:
1404:
1397:
1396:
1389:
1382:
1374:
1366:
1365:
1340:
1311:
1280:
1279:
1277:
1274:
1273:
1272:
1267:
1262:
1257:
1252:
1247:
1240:
1237:
1100:
1097:
1034:
1013:
1010:
1007:
1004:
1001:
998:
995:
992:
989:
982:
979:
976:
970:
962:
958:
954:
949:
942:
938:
934:
928:
921:
916:
912:
908:
900:
896:
892:
887:
880:
876:
872:
866:
860:
857:
852:
847:
844:
813:
812:
801:
798:
795:
792:
789:
786:
782:
778:
775:
772:
758:
747:
744:
741:
738:
735:
732:
728:
724:
721:
718:
704:
679:
659:
651:
647:
643:
638:
634:
627:
623:
617:
613:
610:
606:
598:
594:
590:
585:
581:
574:
570:
564:
560:
557:
551:
506:
503:
499:deuterium lamp
480:fine structure
421:
420:
417:
416:
415:(Ultraviolet)
413:
407:(Ultraviolet)
405:
399:(Ultraviolet)
397:
385:
375:
365:
355:
345:
341:
340:
337:
334:
331:
328:
325:
322:
319:
316:
312:
311:
308:
305:
302:
299:
296:
293:
290:
287:
283:
282:
279:
276:
273:
270:
267:
264:
261:
258:
254:
253:
250:
247:
244:
241:
238:
235:
232:
229:
225:Transition of
197:refers to the
150:
147:
107:displays four
70:atomic physics
36:The "visible"
26:
18:Balmer formula
9:
6:
4:
3:
2:
1480:
1469:
1466:
1464:
1461:
1460:
1458:
1443:
1440:
1438:
1435:
1433:
1430:
1428:
1425:
1423:
1420:
1418:
1417:Balmer series
1415:
1413:
1410:
1409:
1406:
1402:
1395:
1390:
1388:
1383:
1381:
1376:
1375:
1372:
1361:
1357:
1350:
1344:
1338:
1334:
1328:
1326:
1324:
1322:
1320:
1318:
1316:
1300:
1296:
1292:
1285:
1281:
1271:
1268:
1266:
1263:
1261:
1258:
1256:
1253:
1251:
1248:
1246:
1243:
1242:
1236:
1234:
1230:
1226:
1222:
1217:
1215:
1212:
1208:
1204:
1200:
1199:spectral type
1196:
1192:
1188:
1184:
1179:
1177:
1173:
1169:
1165:
1164:star clusters
1161:
1160:moving groups
1157:
1153:
1149:
1148:neutron stars
1145:
1141:
1137:
1133:
1128:
1126:
1121:
1116:
1114:
1110:
1106:
1096:
1054:
1040:
1033:
1029:
1024:
1011:
1008:
1005:
1002:
999:
996:
993:
990:
987:
968:
960:
956:
952:
947:
940:
936:
932:
926:
914:
910:
906:
898:
894:
890:
885:
878:
874:
870:
864:
858:
855:
850:
845:
842:
832:
830:
826:
822:
818:
799:
793:
790:
787:
784:
776:
773:
762:
759:
745:
739:
736:
733:
730:
722:
719:
708:
705:
683:
680:
677:
674:
673:
672:
657:
649:
645:
641:
636:
632:
625:
621:
615:
611:
608:
604:
596:
592:
588:
583:
579:
572:
568:
562:
558:
555:
549:
541:
537:
532:
512:
500:
495:
491:
489:
485:
481:
476:
474:
470:
466:
462:
458:
454:
450:
445:
443:
439:
435:
431:
427:
411:
403:
395:
390:
383:
379:
373:
369:
363:
359:
353:
349:
343:
342:
314:
313:
285:
284:
281:Balmer break
256:
255:
228:
224:
223:
220:
219:
218:
216:
212:
208:
204:
200:
196:
192:
188:
184:
176:
172:
168:
164:
163:hydrogen atom
160:
155:
146:
144:
140:
135:
133:
129:
126:
122:
118:
114:
110:
106:
102:
98:
93:
91:
90:Johann Balmer
87:
83:
82:hydrogen atom
79:
78:spectral line
75:
71:
67:
63:
62:Balmer series
55:
51:
47:
46:visible range
43:
39:
34:
30:
19:
1442:Other series
1432:Pfund series
1416:
1412:Lyman series
1355:
1343:
1302:. Retrieved
1295:HyperPhysics
1294:
1284:
1260:Lyman series
1218:
1207:H II regions
1180:
1140:binary stars
1129:
1117:
1102:
1052:
1031:
1027:
1025:
833:
828:
824:
814:
760:
706:
681:
675:
539:
531:spectroscopy
508:
487:
483:
477:
473:H II regions
469:Orion Nebula
456:
452:
446:
422:
226:
210:
206:
194:
190:
186:
180:
170:
142:
136:
127:
95:The visible
94:
66:Balmer lines
65:
61:
59:
29:
1152:black holes
389:Ultraviolet
278:H-η / Ba-η
275:H-ζ / Ba-ζ
272:H-ε / Ba-ε
269:H-δ / Ba-δ
266:H-γ / Ba-γ
263:H-β / Ba-β
260:H-α / Ba-α
132:ultraviolet
111:, 410
109:wavelengths
54:ultraviolet
1457:Categories
1250:Bohr model
1183:absorption
1144:exoplanets
1093:.57 m
536:wavelength
175:wavelength
1227:given by
1211:planetary
1172:redshifts
1115:C and F.
1105:astronomy
1078:10 m
1012:…
948:−
886:−
846:λ
791:≥
777:∈
737:≥
723:∈
693:10 m
642:−
589:−
550:λ
307:383.5397
304:388.9064
301:397.0075
298:410.1734
295:434.0472
121:electrons
92:in 1885.
86:empirical
50:naked eye
1304:March 1,
1239:See also
1187:emission
1109:universe
1084: =
701: nm
527: nm
519: nm
465:emission
461:universe
447:The red
292:486.135
289:656.279
183:electron
149:Overview
105:hydrogen
97:spectrum
1221:calcium
1214:nebulae
1195:kelvins
1176:quasars
1134:due to
1082:
1061:
1057:
1043:
1037:is the
697:364.506
523:364.506
515:364.506
449:H-alpha
434:Paschen
167:H-alpha
161:of the
117:photons
42:H-alpha
1233:helium
1026:where
984:
671:Where
553:
436:, and
378:Violet
344:Color
310:364.5
1352:(PDF)
1276:Notes
1191:stars
1071:3.645
686:3.645
511:light
430:Lyman
339:3.40
336:3.23
333:3.19
330:3.13
327:3.03
324:2.86
321:2.55
318:1.89
257:Name
103:from
101:light
64:, or
1360:NIST
1306:2008
1209:and
1150:and
1118:The
1073:0682
827:for
688:0682
368:Blue
358:Cyan
252:∞→2
249:9→2
246:8→2
243:7→2
240:6→2
237:5→2
234:4→2
231:3→2
60:The
1185:or
1091:731
1088:973
695:or
542:):
348:Red
201:or
119:by
99:of
68:in
1459::
1358:.
1354:.
1314:^
1297:.
1293:.
1205:,
1166:,
1162:,
1142:,
1095:.
1086:10
699:82
525:82
517:82
444:.
432:,
391:)
113:nm
1393:e
1386:t
1379:v
1362:.
1308:.
1197:(
1076:×
1067:/
1064:4
1053:B
1049:/
1046:4
1035:H
1032:R
1028:λ
1009:,
1006:5
1003:,
1000:4
997:,
994:3
991:=
988:n
981:r
978:o
975:f
969:)
961:2
957:n
953:1
941:2
937:2
933:1
927:(
920:H
915:R
911:=
907:)
899:2
895:n
891:1
879:2
875:2
871:1
865:(
859:B
856:4
851:=
843:1
829:n
825:m
800:.
797:)
794:m
788:n
785:,
781:N
774:n
771:(
761:n
746:.
743:)
740:1
734:m
731:,
727:N
720:m
717:(
707:m
703:.
691:×
682:B
676:λ
658:)
650:2
646:2
637:2
633:m
626:2
622:m
616:(
612:B
609:=
605:)
597:2
593:n
584:2
580:m
573:2
569:m
563:(
559:B
556:=
540:B
488:n
484:n
457:n
453:n
387:(
227:n
211:n
207:n
195:n
191:n
187:n
171:Z
143:n
128:n
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.