620:
190:
20:
176:
is much shorter at 1.414(2) Å with a more acute C−O−C angle of 111°. It can be appreciated that the siloxanes would have low barriers for rotation about the Si−O bonds as a consequence of low steric hindrance. This geometric consideration is the basis of the useful properties of some
851:
Findings about bioaccumulation have been largely based on laboratory studies. Field studies of bioaccumulation have not reached consensus. "Even if the concentrations of siloxanes we have found in fish are high compared to concentrations of classical contaminants like
544:
is used to make dental impressions and industrial impressions. The use of a poly-siloxane precursor in polymer derived ceramics allows the formation of ceramic bodies with complex shapes, although the significant shrinkage in pyrolysis needs to be taken into account.
871:
Cyclomethicones are ubiquitous because they are widely used in biomedical and cosmetic applications. They can be found at high levels in
American cities. They can be toxic to aquatic animals in concentrations often found in the environment. The cyclomethicones
1413:
1346:
Wang, De-Gao; Norwood, Warren; Alaee, Mehran; Byer, Jonatan D.; Brimble, Samantha (October 2013). "Review of recent advances in research on the toxicity, detection, occurrence and fate of cyclic volatile methyl siloxanes in the environment".
843:
Because silicones are heavily used in biomedical and cosmetic applications, their toxicology has been intensively examined. "The inertness of silicones toward warmblooded animals has been demonstrated in a number of tests." With an
868:
in Canada have shown concentrations of siloxanes decrease at higher range in the food chain. This finding raises questions about which factors influence the bioaccumulation potential of siloxanes."
906:
1173:
S. Varaprath, K. L. Salyers, K. P. Plotzke and S. Nanavati "Identification of
Metabolites of Octamethylcyclotetrasiloxane (D4) in Rat Urine" Drug Metab Dispos 1999, 27, 1267-1273.
848:
in rats of >50 g/kg, they are virtually nontoxic. Questions remain however about chronic toxicity or the consequences of bioaccumulation since siloxanes can be long-lived.
1033:
K. Vojinović, U. Losehand, N. W. Mitzel. "Dichlorosilane–dimethyl ether aggregation: a new motif in halosilane adduct formation". Dalton Trans., 2004, 2578–2581.
607:. These silanols are produced at such low levels that they do not interfere with hydrolytic enzymes. Even though some cyclomethicones structurally resemble
560:
Cyclomethicones are a group of methyl siloxanes, a class of liquid silicones (cyclic polydimethylsiloxane polymers) that possess the characteristics of low
999:
Röshe, L.; John, P.; Reitmeier, R. "Organic
Silicon Compounds" Ullmann’s Encyclopedia of Industrial Chemistry. John Wiley and Sons: San Francisco, 2003.
653:. In some cases, siloxane materials are composed of several different types of siloxane groups; these are labeled according to the number of Si−O bonds:
1247:
914:
933:
under a pollution prevention plan. A scientific review in Canada in 2011 concluded that "Siloxane D5 does not pose a danger to the environment."
596:. They are used in many cosmetic products including deodorants and antiperspirants which need to coat the skin but not remain tacky afterward.
540:(SiC). By exploiting this reaction, polysiloxanes have been used as preceramic polymers in various processes including additive manufacturing.
101:, the defining feature of which is that each pair of silicon centres is separated by one oxygen atom. The siloxane functional group forms the
1321:
1279:
1016:
H. Steinfink, B. Post and I. Fankuchen. "The crystal structure of octamethyl cyclotetrasiloxane". Acta
Crystallogr. 1955, vol. 8, 420–424.
525:
This reaction proceeds by production of silanols. Similar reactions are used industrially to convert cyclic siloxanes to linear polymers.
1210:
F. Dankert, C. von Hänisch (2021). "Siloxane
Coordination Revisited: Si−O Bond Character, Reactivity and Magnificent Molecular Shapes".
926:
1158:
1131:
1104:
323:
Hydrolysis of a silyldichloride can afford linear or cyclic products. Linear products are terminated with silanol groups:
1280:"Siloxanes: Soft, shiny -- and dangerous?" by Christine Solbakken, Science Nordic, August 28, 2015. Retrieved May 31, 2016
983:
1050:
Silicon: Organosilicon
Chemistry. Encyclopedia of Inorganic Chemistry Online, 2nd ed.; Wiley: New Jersey, 2005.
1264:
910:
169:
98:
1150:
Delivery System
Handbook for Personal Care and Cosmetic Products: Technology, Applications and Formulations
1072:
918:
880:
873:
787:
758:
623:
178:
1433:
733:
447:
402:
141:. Siloxanes are manmade and have many commercial and industrial applications because of the compounds’
280:
The siloxane bond can then form via a silanol + silanol pathway or a silanol + chlorosilane pathway:
536:, upon combustion in an inert atmosphere, generally undergo pyrolysis to form silicon oxycarbide or
434:, with silicon centers at the corners of a cube oxygen centres spanning each of the twelve edges.
28:
565:
193:
1148:
1121:
1094:
1295:
1356:
597:
398:
146:
126:
8:
1428:
431:
408:
Starting from trisilanols, cages are possible, such as the species with the formula (RSi)
162:
1360:
1448:
1227:
541:
1368:
1372:
1260:
1231:
1154:
1127:
1100:
1364:
1252:
1219:
1192:
1076:
1051:
1034:
1017:
1000:
954:
948:
490:
401:(PDMS), are of great commercial value. Their production requires the production of
225:
102:
56:
40:
36:
922:
887:
537:
443:
1245:
Moretto, Hans-Heinrich; Schulze, Manfred; Wagner, Gebhard (2005). "Silicones".
619:
589:
549:
1434:
Journal Of
Protective Coatings And Linings: Field Performance of Polysiloxanes
1390:
1196:
1021:
1442:
1256:
1055:
1004:
1414:
1376:
1223:
1067:
S. D. Kinrade, J. C. H. Donovan, A. S. Schach and C. T. G. Knight (2002),
929:(very persistent and very bioaccumulative) properties. Canada regulates D
608:
581:
573:
189:
165:
1183:
R. Pietschnig, S. Spirk (2016). "The
Chemistry of Organo Silanetriols".
978:
861:
857:
221:
157:
Siloxanes generally adopt structures expected for linked tetrahedral ("
142:
958:
603:
Cyclomethicones, like all siloxanes, degrade by hydrolysis, producing
172:
is rather open at 142.5°. By contrast, the C−O distance in a typical
1391:"Candidate List of substances of very high concern for Authorisation"
1080:
1038:
865:
569:
561:
1092:
533:
487:
106:
60:
1069:
Two substituted cubic octameric silicate cages in aqueous solution
604:
442:
Oxidation of organosilicon compounds, including siloxanes, gives
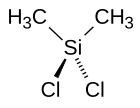
253:
67:
64:
44:
1322:"Chemicals from Personal Care Products Pervasive in Chicago Air"
19:
647:
168:
is 1.64 Å (vs Si–C distance of 1.92 Å) and the
48:
572:
and in certain circumstances useful cleaning solvents. Unlike
173:
1394:
1296:"Exec: Silicone industry must focus on safety, environment"
1096:
Handbook for Critical Cleaning: Cleaning Agents and Systems
853:
1209:
838:
1123:
Sustainability: How the Cosmetics Industry is Greening Up
1182:
1086:
446:. This conversion is illustrated by the combustion of
486:
Strong base degrades siloxane group, often affording
1345:
1244:
890:
in some aquatic organisms, according to one report.
252:The reaction proceeds via the initial formation of
220:The main route to siloxane functional group is by
430: = 12) structures. The cubic cages are
177:siloxane-containing materials, such as their low
1440:
137:(where the three Rs may be different) is called
1248:Ullmann's Encyclopedia of Industrial Chemistry
1119:
1093:Barbara Kanegsberg; Edward Kanegsberg (2011).
23:Polydimethylsiloxane is a prevalent siloxane.
1146:
944:Environmental Chemistry of Organosiloxanes.
1319:
1113:
426: = 8) and hexagonal prismatic (
360:Cyclic products have no silanol termini:
184:
1140:
995:
993:
618:
600:is a major producer of cyclomethicones.
188:
18:
1429:EPA report: Siloxane D5 in Dry-cleaning
909:regulation. They were characterized as
839:Safety and environmental considerations
542:Polyvinyl siloxane (vinyl polysiloxane)
1441:
1293:
1044:
905:have been deemed hazardous as per the
1289:
1287:
990:
611:, they bind metal ions only weakly.
1320:Bienkowski, Brian (30 April 2013).
1120:Amarjit Sahota (25 November 2013).
698:Cyclic siloxanes (cyclomethicones)
13:
1339:
1284:
942:Christoph Rücker, Klaus Kümmerer:
555:
125:, the premier example of which is
16:Organic functional group (Si–O–Si)
14:
1460:
1422:
1369:10.1016/j.chemosphere.2012.10.041
1294:Karpus, Jennifer (20 June 2014).
1147:Meyer Rosen (23 September 2005).
208:) is a key precursor to cyclic (D
815:: dodecamethylcyclohexasiloxane
1407:
1383:
1313:
1273:
1238:
1203:
1176:
1167:
1153:. William Andrew. p. 693.
953:115(1), 2015, p. 466–524,
911:substances of very high concern
856:, several other studies in the
829:M: tetradecamethylhexasiloxane
614:
1061:
1027:
1010:
972:
129:(PDMS). The functional group
1:
965:
936:
803:M: dodecamethylpentasiloxane
745:, MDM: octamethyltrisiloxane
216:, etc.) and linear siloxanes.
179:glass transition temperatures
1073:J. Chem. Soc., Dalton Trans.
788:decamethylcyclopentasiloxane
759:octamethylcyclotetrasiloxane
624:Decamethylcyclopentasiloxane
548:Trisiloxanes may be used as
437:
152:
7:
774:M: decamethyltetrasiloxane
720:, MM: hexamethyldisiloxane
588:: both groups consist of a
10:
1465:
734:hexamethylcyclotrisiloxane
638:is derived from the words
448:hexamethylcyclotrisiloxane
403:dimethylsilicon dichloride
1300:Rubber & Plastic News
1197:10.1016/j.ccr.2016.03.010
1022:10.1107/S0365110X55001333
534:Polysiloxanes (silicones)
509:O + 2 NaOH → 2 (CH
97:. Siloxanes also include
1257:10.1002/14356007.a24_057
1056:10.1002/0470862106.ia220
1005:10.1002/14356007.a24_021
893:In the European Union, D
149:, and high flexibility.
1251:. Weinheim: Wiley-VCH.
528:
29:organosilicon chemistry
1224:10.1002/ejic.202100275
1126:. Wiley. p. 208.
631:
584:, cyclomethicones are
580:siloxanes that do not
568:as well as being skin
217:
194:Dimethyldichlorosilane
185:Synthesis of siloxanes
161:-like") centers. The
59:siloxanes include the
24:
622:
397:The linear products,
192:
22:
913:(SVHC) due to their
432:cubane-type clusters
399:polydimethylsiloxane
147:thermal conductivity
127:polydimethylsiloxane
1361:2013Chmsp..93..711W
1326:Scientific American
1212:Eur. J. Inorg. Chem
1099:. CRC. p. 19.
630:, a cyclic siloxane
632:
218:
99:branched compounds
70:with the formulae
47:atoms bound to an
25:
1218:(29): 2907–2927.
1160:978-0-8155-1682-8
1133:978-1-118-67650-9
1106:978-1-4398-2827-4
959:10.1021/cr500319v
836:
835:
704:Linear siloxanes
226:silicon chlorides
1456:
1416:
1411:
1405:
1404:
1402:
1401:
1387:
1381:
1380:
1343:
1337:
1336:
1334:
1332:
1317:
1311:
1310:
1308:
1306:
1291:
1282:
1277:
1271:
1270:
1242:
1236:
1235:
1207:
1201:
1200:
1185:Coord. Chem. Rev
1180:
1174:
1171:
1165:
1164:
1144:
1138:
1137:
1117:
1111:
1110:
1090:
1084:
1081:10.1039/b107758a
1065:
1059:
1048:
1042:
1039:10.1039/B405684A
1031:
1025:
1014:
1008:
997:
988:
976:
949:Chemical Reviews
695:
694:
136:
124:
96:
83:
54:
41:functional group
37:organic compound
1464:
1463:
1459:
1458:
1457:
1455:
1454:
1453:
1439:
1438:
1425:
1420:
1419:
1412:
1408:
1399:
1397:
1389:
1388:
1384:
1344:
1340:
1330:
1328:
1318:
1314:
1304:
1302:
1292:
1285:
1278:
1274:
1267:
1243:
1239:
1208:
1204:
1181:
1177:
1172:
1168:
1161:
1145:
1141:
1134:
1118:
1114:
1107:
1091:
1087:
1066:
1062:
1049:
1045:
1032:
1028:
1015:
1011:
998:
991:
984:IUPAC Gold Book
977:
973:
968:
939:
932:
925:and toxic) and
923:bioaccumulative
904:
900:
896:
888:bioaccumulative
884:
877:
864:in the US, and
847:
841:
828:
824:
814:
802:
798:
785:
773:
769:
756:
744:
731:
719:
690:
686:
679:
675:
668:
664:
660:
629:
617:
595:
558:
556:Cyclomethicones
538:silicon carbide
531:
520:
516:
512:
508:
504:
500:
481:
477:
473:
469:
465:
461:
457:
444:silicon dioxide
440:
421:
413:
392:
384:
378:
374:
370:
355:
347:
341:
337:
333:
318:
314:
310:
306:
299:
295:
291:
287:
275:
271:
267:
259:
247:
243:
239:
235:
215:
211:
207:
203:
199:
187:
155:
134:
130:
123:
117:
113:
109:
95:
89:
85:
81:
75:
71:
52:
17:
12:
11:
5:
1462:
1452:
1451:
1437:
1436:
1431:
1424:
1423:External links
1421:
1418:
1417:
1406:
1382:
1355:(5): 711–725.
1338:
1312:
1283:
1272:
1265:
1237:
1202:
1175:
1166:
1159:
1139:
1132:
1112:
1105:
1085:
1060:
1043:
1026:
1009:
989:
970:
969:
967:
964:
963:
962:
938:
935:
930:
902:
898:
894:
882:
875:
845:
840:
837:
834:
833:
830:
826:
822:
819:
816:
812:
808:
807:
804:
800:
796:
793:
790:
783:
779:
778:
775:
771:
767:
764:
761:
754:
750:
749:
746:
742:
739:
736:
729:
725:
724:
721:
717:
714:
712:
709:
708:
705:
702:
699:
693:
692:
688:
684:
681:
677:
673:
670:
666:
662:
658:
627:
616:
613:
593:
557:
554:
550:diffusion pump
530:
527:
523:
522:
518:
514:
510:
506:
502:
498:
484:
483:
479:
475:
471:
467:
463:
459:
455:
439:
436:
415:
409:
395:
394:
390:
380:
376:
372:
368:
358:
357:
353:
343:
339:
335:
331:
321:
320:
316:
312:
308:
304:
301:
297:
293:
289:
285:
278:
277:
273:
269:
265:
257:
250:
249:
245:
241:
237:
233:
213:
209:
205:
201:
197:
186:
183:
154:
151:
143:hydrophobicity
132:
119:
115:
111:
91:
87:
77:
73:
15:
9:
6:
4:
3:
2:
1461:
1450:
1447:
1446:
1444:
1435:
1432:
1430:
1427:
1426:
1415:
1410:
1396:
1392:
1386:
1378:
1374:
1370:
1366:
1362:
1358:
1354:
1350:
1342:
1327:
1323:
1316:
1301:
1297:
1290:
1288:
1281:
1276:
1268:
1262:
1258:
1254:
1250:
1249:
1241:
1233:
1229:
1225:
1221:
1217:
1213:
1206:
1198:
1194:
1190:
1186:
1179:
1170:
1162:
1156:
1152:
1151:
1143:
1135:
1129:
1125:
1124:
1116:
1108:
1102:
1098:
1097:
1089:
1082:
1078:
1075:, 1250–1252.
1074:
1070:
1064:
1057:
1053:
1047:
1040:
1036:
1030:
1023:
1019:
1013:
1006:
1002:
996:
994:
986:
985:
980:
975:
971:
960:
956:
952:
950:
945:
941:
940:
934:
928:
924:
920:
916:
912:
908:
891:
889:
885:
878:
869:
867:
863:
859:
855:
849:
831:
820:
817:
810:
809:
805:
794:
791:
789:
781:
780:
776:
765:
762:
760:
752:
751:
747:
740:
737:
735:
727:
726:
722:
715:
713:
711:
710:
706:
703:
700:
697:
696:
682:
671:
656:
655:
654:
652:
651:
645:
641:
637:
625:
621:
612:
610:
606:
601:
599:
591:
587:
583:
579:
575:
571:
567:
563:
553:
551:
546:
543:
539:
535:
526:
496:
495:
494:
492:
489:
453:
452:
451:
449:
445:
435:
433:
429:
425:
419:
412:
406:
404:
400:
388:
383:
366:
363:
362:
361:
351:
346:
329:
326:
325:
324:
302:
283:
282:
281:
263:
262:
261:
255:
231:
230:
229:
227:
223:
195:
191:
182:
180:
175:
174:dialkyl ether
171:
170:Si-O-Si angle
167:
164:
160:
150:
148:
144:
140:
128:
122:
108:
104:
100:
94:
80:
69:
66:
62:
58:
50:
46:
42:
39:containing a
38:
34:
30:
21:
1409:
1398:. Retrieved
1385:
1352:
1348:
1341:
1329:. Retrieved
1325:
1315:
1303:. Retrieved
1299:
1275:
1246:
1240:
1215:
1211:
1205:
1188:
1184:
1178:
1169:
1149:
1142:
1122:
1115:
1095:
1088:
1068:
1063:
1046:
1029:
1012:
982:
974:
947:
943:
892:
870:
850:
842:
683:T-units: (CH
672:D-units: (CH
657:M-units: (CH
649:
643:
639:
635:
633:
615:Nomenclature
609:crown ethers
602:
585:
577:
576:, which are
574:dimethicones
559:
547:
532:
524:
517:SiONa + H
485:
441:
427:
423:
422:with cubic (
417:
410:
407:
396:
386:
381:
364:
359:
352: − 1) H
349:
344:
327:
322:
279:
251:
219:
158:
156:
138:
120:
92:
78:
32:
26:
1349:Chemosphere
860:in Norway,
276:Si−OH + HCl
166:bond length
1400:2019-12-18
1266:3527306730
1191:: 87–106.
966:References
937:Literature
919:persistent
862:Lake Pepin
858:Oslo Fjord
646:ygen, and
570:emollients
566:volatility
470:→ 3 SiO
222:hydrolysis
72:H[OSiH
61:oligomeric
1449:Siloxanes
1232:239645449
979:Siloxanes
866:Lake Erie
832:107-52-8
818:540-97-6
806:141-63-9
792:541-02-6
777:141-62-8
763:556-67-2
748:107-51-7
738:541-05-9
723:107-46-0
634:The word
582:evaporate
564:and high
562:viscosity
438:Reactions
311:Si−Cl → R
307:Si−OH + R
288:Si−OH → R
268:Si−Cl + H
236:Si−Cl + H
153:Structure
107:silicones
86:[OSiH
65:polymeric
1443:Category
1377:23211328
636:siloxane
605:silanols
590:backbone
488:siloxide
315:Si−O−SiR
292:Si−O−SiR
260:Si−OH):
254:silanols
244:Si−O−SiR
114:Si−O−SiR
103:backbone
68:hydrides
33:siloxane
1357:Bibcode
1331:8 April
1305:8 April
552:fluid.
474:+ 6 CO
466:+ 12 O
248:+ 2 HCl
110:[−R
55:. The
53:Si−O−Si
51:atom:
45:silicon
43:of two
35:is an
1375:
1263:
1230:
1157:
1130:
1103:
642:icon,
626:, or D
586:cyclic
578:linear
478:+ 9 H
371:Si(OH)
348:OH + (
334:Si(OH)
196:(Si(CH
145:, low
139:siloxy
57:parent
49:oxygen
1228:S2CID
907:REACH
901:and D
491:salts
338:→ H(R
319:+ HCl
272:O → R
240:O → R
1395:ECHA
1373:PMID
1333:2015
1307:2015
1261:ISBN
1216:2021
1155:ISBN
1128:ISBN
1101:ISBN
946:In:
927:vPvB
886:are
879:and
854:PCBs
825:, MD
799:, MD
770:, MD
707:CAS
701:CAS
687:)SiO
680:SiO,
529:Uses
497:((CH
462:SiO)
454:((CH
379:SiO)
375:→ (R
342:SiO)
163:Si−O
135:SiO−
84:and
63:and
31:, a
1365:doi
1253:doi
1220:doi
1193:doi
1189:323
1077:doi
1052:doi
1035:doi
1018:doi
1001:doi
955:doi
915:PBT
897:, D
689:1.5
667:0.5
665:SiO
650:ane
648:alk
640:sil
598:Dow
592:of
505:Si)
296:+ H
284:2 R
232:2 R
224:of
212:, D
105:of
27:In
1445::
1393:.
1371:.
1363:.
1353:93
1351:.
1324:.
1298:.
1286:^
1259:.
1226:.
1214:.
1187:.
1071:.
992:^
981:,
921:,
846:50
844:LD
786::
757::
732::
644:ox
493::
450::
420:/2
405:.
385:+
256:(R
228::
204:Cl
181:.
159:sp
118:−]
82:OH
1403:.
1379:.
1367::
1359::
1335:.
1309:.
1269:.
1255::
1234:.
1222::
1199:.
1195::
1163:.
1136:.
1109:.
1083:.
1079::
1058:.
1054::
1041:.
1037::
1024:.
1020::
1007:.
1003::
987:.
961:.
957::
951:.
931:4
917:(
903:6
899:5
895:4
883:5
881:D
876:4
874:D
827:4
823:6
821:L
813:6
811:D
801:3
797:5
795:L
784:5
782:D
772:2
768:4
766:L
755:4
753:D
743:3
741:L
730:3
728:D
718:2
716:L
691:.
685:3
678:2
676:)
674:3
669:,
663:3
661:)
659:3
628:5
594:n
521:O
519:2
515:3
513:)
511:3
507:2
503:3
501:)
499:3
482:O
480:2
476:2
472:2
468:2
464:3
460:2
458:)
456:3
428:n
424:n
418:n
416:3
414:O
411:n
393:O
391:2
389:H
387:n
382:n
377:2
373:2
369:2
367:R
365:n
356:O
354:2
350:n
345:n
340:2
336:2
332:2
330:R
328:n
317:3
313:3
309:3
305:3
303:R
300:O
298:2
294:3
290:3
286:3
274:3
270:2
266:3
264:R
258:3
246:3
242:3
238:2
234:3
214:4
210:3
206:2
202:2
200:)
198:3
133:3
131:R
121:n
116:2
112:2
93:n
90:]
88:2
79:n
76:]
74:2
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.