265:
293:
233:
197:
1331:
240:
1370:
316:
38:
160:
halides ion has moved sufficiently away from the carbocation. The negatively charged halide ion shields the carbocation from being attacked on the front side, and backside attack, which leads to inversion of configuration, is preferred. Thus the actual product no doubt consists of a mixture of enantiomers but the enantiomers with inverted configuration would predominate and complete racemization does not occurs.
1247:
However, under certain conditions, non-first-order reaction kinetics can be observed. In particular, when a large concentration of bromide is present while the concentration of water is limited, the reverse of the first step becomes important kinetically. As the SSA rate law indicates, under these
159:
1 mechanism, a carbocation is formed which is planar and hence attack of nucleophile (second step) may occur from either side to give a racemic product, but actually complete racemization does not take place. This is because the nucleophilic species attacks the carbocation even before the departing
307:
1 reaction is often regarded as being first order in alkyl halide and zero order in nucleophile, this is a simplification that holds true only under certain conditions. While it, too, is an approximation, the rate law derived from the steady state approximation (SSA) provides more insight into the
1243:
O to MeOH) does not affect the reaction rate, though the product is, of course, different. In this regime, the first step (ionization of the alkyl bromide) is slow, rate-determining, and irreversible, while the second step (nucleophilic addition) is fast and kinetically invisible.
939:
Under normal synthetic conditions, the entering nucleophile is more nucleophilic than the leaving group and is present in excess. Moreover, kinetic experiments are often conducted under initial rate conditions (5 to 10% conversion) and without the addition of bromide, so
1431:. This will be especially true if the reaction is heated. Finally, if the carbocation intermediate can rearrange to a more stable carbocation, it will give a product derived from the more stable carbocation rather than the simple substitution product.
1663:
Bateman LC, Church MG, Hughes ED, Ingold CK, Taher NA (1940). "188. Mechanism of substitution at a saturated carbon atom. Part XXIII. A kinetic demonstration of the unimolecular solvolysis of alkyl halides. (Section E) a general discussion".
1357:
hybridized carbon with trigonal planar molecular geometry. This allows two different ways for the nucleophilic attack, one on either side of the planar molecule. If neither approach is favored, then these two ways occur equally, yielding a
1234:
1449:
1 reaction involves formation of an unstable carbocation intermediate in the rate-determining step (RDS), anything that can facilitate this process will speed up the reaction. The normal solvents of choice are both
1238:
the simple first-order rate law described in introductory textbooks. Under these conditions, the concentration of the nucleophile does not affect the rate of the reaction, and changing the nucleophile (e.g. from
1381:
However, an excess of one stereoisomer can be observed, as the leaving group can remain in proximity to the carbocation intermediate for a short time and block nucleophilic attack. This stands in contrast to the
151:
2 mechanism. This type of mechanism involves two steps. The first step is the ionization of alkyl halide in the presence of aqueous acetone or ethyl alcohol. This step provides a carbocation as an intermediate.
935:
1557:
1414:
1 and E1 reactions are competitive reactions and it becomes difficult to favor one over the other. Even if the reaction is performed cold, some alkene may be formed. If an attempt is made to perform an
519:
655:
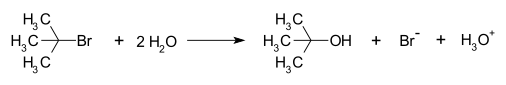
330:-butyl cation is a high-energy species that is present only at very low concentration and cannot be directly observed under normal conditions. Thus, the SSA can be applied to this species:
97:. This relationship holds for situations where the amount of nucleophile is much greater than that of the intermediate. Instead, the rate equation may be more accurately described using
756:
1057:
1466:
the leaving group in particular). Typical polar protic solvents include water and alcohols, which will also act as nucleophiles, and the process is known as solvolysis.
1252:
1 reactions are often observed to slow down when an exogenous source of the leaving group (in this case, bromide) is added to the reaction mixture. This is known as the
264:
1386:
2 mechanism, which is a stereospecific mechanism where stereochemistry is always inverted as the nucleophile comes in from the rear side of the leaving group.
973:
1248:
conditions there is a fractional (between zeroth and first order) dependence on , while there is a negative fractional order dependence on . Thus, S
1064:
1767:
1932:
1771:
292:
232:
1330:
258:) a third step is required to complete the reaction. When the solvent is water, the intermediate is an oxonium ion. This reaction step is fast.
1960:
1893:
Arnold H. Fainberg & S. Winstein (1956). "Correlation of
Solvolysis Rates. III.1 t-Butyl Chloride in a Wide Range of Solvent Mixtures".
2407:
196:
763:
1282:
2 reaction. Additionally, bulky substituents on the central carbon increase the rate of carbocation formation because of the relief of
1369:
1577:
a solvent parameter. For example, 100% ethanol gives Y = −2.3, 50% ethanol in water Y = +1.65 and 15% concentration Y = +3.2.
1502:
336:
2172:
2049:
2006:
2313:
1953:
1796:
1743:
1647:
1366:
1 reaction of S-3-chloro-3-methylhexane with an iodide ion, which yields a racemic mixture of 3-iodo-3-methylhexane:
526:
1596:
1820:
Wagner, Carl E.; Marshall, Pamela A. (2010). "Synthesis of 2,5-Dichloro-2,5-dimethylhexane by an SN1 Reaction".
2221:
2216:
2026:
1591:
2386:
2381:
1299:
1274:
1 mechanism tends to dominate when the central carbon atom is surrounded by bulky groups because such groups
1337:
As the alpha and beta substitutions increase with respect to leaving groups, the reaction is diverted from S
2412:
1946:
662:
1428:
1403:
2351:
2041:
1927:
2078:
1978:
978:
131:
78:
1738:. Dougherty, Dennis A., 1952-. Mill Valley, California: University Science Books. pp. 638–639.
2308:
147:
et al. in 1940. This reaction does not depend much on the strength of the nucleophile, unlike the S
98:
1402:. If the reaction is performed under warm or hot conditions (which favor an increase in entropy),
93:
is often shown as having first-order dependence on the substrate and zero-order dependence on the
2356:
2157:
1791:. Richardson, Kathleen Schueller. (3rd ed.). New York: Harper & Row. pp. 330–331.
1696:
Peters, K. S. (2007). "Nature of
Dynamic Processes Associated with the SN1 Reaction Mechanism".
1362:
of enantiomers if the reaction takes place at a stereocenter. This is illustrated below in the S
2111:
1452:
1399:
2341:
2273:
2131:
2121:
82:
62:
2336:
2064:
1830:
1395:
70:
8:
2346:
2278:
2263:
2206:
1567:
280:
122:
110:
17:
1834:
1353:
The carbocation intermediate formed in the reaction's rate determining step (RDS) is an
2371:
2141:
1970:
1761:
181:
173:
144:
140:
943:
2366:
2361:
2323:
2268:
2187:
2167:
2103:
1867:
1802:
1792:
1749:
1739:
1714:
1643:
1322:
1254:
239:
66:
45:
1 reaction. The leaving group is denoted "X", and the nucleophile is denoted "Nu–H".
2298:
2247:
2201:
1902:
1875:
1838:
1706:
1669:
1291:
1287:
1275:
1855:
Sorrell, Thomas N. "Organic
Chemistry, 2nd Edition" University Science Books, 2006
312:
1 reaction. Consider the following reaction scheme for the mechanism shown above:
2376:
2288:
2237:
1865:
Ernest
Grunwald & S. Winstein (1948). "The Correlation of Solvolysis Rates".
1440:
1359:
315:
1262:
1 mechanism (although the absence of a common ion effect does not rule it out).
2083:
2072:
1822:
1458:
1307:
1302:
suggests that this, too, will increase the rate of carbocation formation. The S
1229:{\displaystyle {\text{rate}}={\frac {d}{dt}}={\frac {k_{1}k_{2}}{k_{2}}}=k_{1}}
2401:
2331:
2303:
2211:
2162:
2136:
1586:
1283:
284:
272:
219:
90:
31:
1938:
1806:
1753:
2283:
2089:
1996:
1718:
1628:, 6th ed., Pearson/Prentice Hall, Upper Saddle River, New Jersey, USA, 2005
1601:
185:
114:
109:
under strongly basic conditions or, under strongly acidic conditions, with
106:
86:
2242:
2177:
1786:
1733:
1673:
323:
276:
251:
250:
Nucleophilic attack: the carbocation reacts with the nucleophile. If the
102:
94:
1906:
1879:
105:
intermediate and is commonly seen in reactions of secondary or tertiary
1698:
1474:
523:(2) Concentration of t-butyl cation, based on steady state assumption:
177:
136:
37:
1842:
1710:
2293:
1923:
1463:
1424:
1420:
1286:
that occurs. The resultant carbocation is also stabilized by both
1059:
often holds. Under these conditions, the SSA rate law reduces to:
930:{\displaystyle {\frac {d}{dt}}={\frac {k_{1}k_{2}}{k_{-1}+k_{2}}}}
1482:
255:
223:
1407:
2196:
1892:
1486:
1295:
212:
113:. With primary and secondary alkyl halides, the alternative
1552:{\displaystyle \log {\left({\frac {k}{k_{0}}}\right)}=mY\,}
30:"SN1" redirects here. For the Canadian sports channel, see
1864:
2016:
1662:
1427:
ion, the alkene will again be formed, this time via an
1258:
and the observation of this effect is evidence for an S
1731:
1419:
1 reaction using a strongly basic nucleophile such as
659:(3) Overall reaction rate, assuming rapid final step:
1505:
1406:
is likely to predominate, leading to formation of an
1067:
981:
946:
766:
760:(4) Steady state rate law, by plugging (2) into (3):
665:
529:
339:
135:. This dissociation pathway is well-described by the
27:
Substitution reaction with a carbocation intermediate
514:{\displaystyle {\frac {d}{dt}}=0=k_{1}-k_{-1}-k_{2}}
1551:
1456:(to stabilize ionic intermediates in general) and
1228:
1051:
967:
929:
750:
649:
513:
279:nucleophile by water acting as a base forming the
1686:Dr Hussain | Class Lecture | Archeived 17/08/2024
2399:
1784:
1306:1 mechanism therefore dominates in reactions at
226:anion) from the carbon atom: this step is slow.
168:An example of a reaction taking place with an S
1819:
1321:from the corresponding diol with concentrated
650:{\displaystyle ={\frac {k_{1}}{k_{-1}+k_{2}}}}
1968:
1954:
1695:
243:Recombination of carbocation with nucleophile
1766:: CS1 maint: multiple names: authors list (
314:
73:of the mechanism expresses two properties—"S
1481:) with that of a standard solvent (80% v/v
1377:1 reaction, showing how racemisation occurs
1961:
1947:
1770:) CS1 maint: numeric names: authors list (
1313:An example of a reaction proceeding in a S
1788:Mechanism and theory in organic chemistry
1666:Journal of the Chemical Society (Resumed)
1548:
238:
36:
207:1 reaction takes place in three steps:
14:
2400:
1987:Unimolecular nucleophilic substitution
51:unimolecular nucleophilic substitution
1997:Bimolecular nucleophilic substitution
1942:
1637:
751:{\displaystyle {\frac {d}{dt}}=k_{2}}
322:Though a relatively stable tertiary
2408:Nucleophilic substitution reactions
2050:Electrophilic aromatic substitution
24:
2017:Nucleophilic internal substitution
2007:Nucleophilic aromatic substitution
1434:
1368:
1348:
975:is negligible. For these reasons,
25:
2424:
1917:
1735:Modern physical organic chemistry
1642:(4th ed.). New York: Wiley.
1389:
129:1 reaction is often known as the
41:General reaction scheme for the S
1597:Neighbouring group participation
1329:
291:
263:
231:
195:
2173:Lindemann–Hinshelwood mechanism
1886:
1858:
1732:Anslyn, Eric V., 1960- (2006).
1566:a reactant constant (m = 1 for
1477:reaction rates of any solvent (
1319:2,5-dichloro-2,5-dimethylhexane
1052:{\displaystyle k_{-1}\ll k_{2}}
218:carbocation by separation of a
2222:Outer sphere electron transfer
2217:Inner sphere electron transfer
2027:Nucleophilic acyl substitution
1849:
1813:
1778:
1725:
1689:
1680:
1656:
1631:
1618:
1592:Nucleophilic acyl substitution
1394:Two common side reactions are
1317:1 fashion is the synthesis of
1223:
1215:
1196:
1176:
1161:
1141:
1138:
1130:
1090:
1082:
1046:
1026:
1010:
995:
962:
947:
921:
901:
885:
870:
852:
832:
829:
821:
781:
773:
745:
725:
722:
707:
680:
672:
641:
621:
605:
590:
572:
564:
545:
530:
508:
488:
485:
470:
454:
439:
436:
421:
402:
394:
361:
346:
333:(1) Steady state assumption:
303:Although the rate law of the S
254:is a neutral molecule (i.e. a
111:secondary or tertiary alcohols
13:
1:
2387:Diffusion-controlled reaction
1611:
287:. This reaction step is fast.
275:: Removal of a proton on the
81:", and the "1" says that the
163:
7:
2042:Electrophilic substitutions
1580:
298:
101:. The reaction involves a
10:
2429:
2352:Energy profile (chemistry)
2314:More O'Ferrall–Jencks plot
1979:Nucleophilic substitutions
1928:Frostburg State University
1640:Advanced Organic Chemistry
1438:
1410:. At lower temperatures, S
29:
2382:Michaelis–Menten kinetics
2322:
2256:
2230:
2186:
2150:
2102:
2063:
2040:
1977:
1935:: the University of Maine
1785:Lowry, Thomas H. (1987).
1400:carbocation rearrangement
1300:Hammond–Leffler postulate
308:kinetic behavior of the S
132:dissociative substitution
79:nucleophilic substitution
2309:Potential energy surface
2188:Electron/Proton transfer
2073:Unimolecular elimination
1265:
143:was first introduced by
2357:Transition state theory
2158:Intramolecular reaction
2084:Bimolecular elimination
2151:Unimolecular reactions
2112:Electrophilic addition
1553:
1378:
1230:
1053:
969:
931:
752:
651:
515:
319:
244:
155:In the first step of S
46:
2342:Rate-determining step
2274:Reactive intermediate
2132:Free-radical addition
2122:Nucleophilic addition
2065:Elimination reactions
1554:
1396:elimination reactions
1372:
1231:
1054:
970:
932:
753:
652:
516:
318:
242:
99:steady-state kinetics
83:rate-determining step
63:substitution reaction
40:
2337:Equilibrium constant
1674:10.1039/JR9400000979
1503:
1065:
979:
944:
764:
663:
527:
337:
71:Hughes-Ingold symbol
2413:Reaction mechanisms
2347:Reaction coordinate
2279:Radical (chemistry)
2264:Elementary reaction
2207:Grotthuss mechanism
1971:reaction mechanisms
1907:10.1021/ja01593a033
1880:10.1021/ja01182a117
1835:2010JChEd..87...81W
123:inorganic chemistry
2372:Arrhenius equation
2142:Oxidative addition
2104:Addition reactions
1638:March, J. (1992).
1549:
1379:
1290:stabilization and
1226:
1049:
965:
927:
748:
647:
511:
320:
245:
182:tert-butyl bromide
174:reaction mechanism
145:Christopher Ingold
141:reaction mechanism
47:
2395:
2394:
2367:Activated complex
2362:Activation energy
2324:Chemical kinetics
2269:Reaction dynamics
2168:Photodissociation
1868:J. Am. Chem. Soc.
1843:10.1021/ed8000057
1711:10.1021/cr068021k
1626:Organic Chemistry
1624:L. G. Wade, Jr.,
1532:
1323:hydrochloric acid
1276:sterically hinder
1255:common ion effect
1221:
1200:
1194:
1183:
1159:
1148:
1136:
1102:
1088:
1071:
1044:
1033:
1002:
954:
925:
919:
908:
877:
850:
839:
827:
793:
779:
743:
732:
714:
692:
678:
645:
639:
628:
597:
570:
537:
506:
495:
477:
446:
428:
400:
373:
353:
67:organic chemistry
16:(Redirected from
2420:
2299:Collision theory
2248:Matrix isolation
2202:Harpoon reaction
2079:E1cB-elimination
1963:
1956:
1949:
1940:
1939:
1911:
1910:
1895:J. Am. Chem. Soc
1890:
1884:
1883:
1862:
1856:
1853:
1847:
1846:
1817:
1811:
1810:
1782:
1776:
1775:
1765:
1757:
1729:
1723:
1722:
1693:
1687:
1684:
1678:
1677:
1660:
1654:
1653:
1635:
1629:
1622:
1558:
1556:
1555:
1550:
1538:
1537:
1533:
1531:
1530:
1518:
1333:
1292:hyperconjugation
1235:
1233:
1232:
1227:
1222:
1219:
1214:
1213:
1201:
1199:
1195:
1192:
1190:
1189:
1184:
1181:
1175:
1174:
1164:
1160:
1157:
1155:
1154:
1149:
1146:
1137:
1134:
1129:
1128:
1119:
1118:
1108:
1103:
1101:
1093:
1089:
1086:
1077:
1072:
1069:
1058:
1056:
1055:
1050:
1045:
1042:
1040:
1039:
1034:
1031:
1025:
1024:
1009:
1008:
1003:
1000:
994:
993:
974:
972:
971:
968:{\displaystyle }
966:
961:
960:
955:
952:
936:
934:
933:
928:
926:
924:
920:
917:
915:
914:
909:
906:
900:
899:
884:
883:
878:
875:
869:
868:
855:
851:
848:
846:
845:
840:
837:
828:
825:
820:
819:
810:
809:
799:
794:
792:
784:
780:
777:
768:
757:
755:
754:
749:
744:
741:
739:
738:
733:
730:
721:
720:
715:
712:
706:
705:
693:
691:
683:
679:
676:
667:
656:
654:
653:
648:
646:
644:
640:
637:
635:
634:
629:
626:
620:
619:
604:
603:
598:
595:
589:
588:
575:
571:
568:
563:
562:
552:
544:
543:
538:
535:
520:
518:
517:
512:
507:
504:
502:
501:
496:
493:
484:
483:
478:
475:
469:
468:
453:
452:
447:
444:
435:
434:
429:
426:
420:
419:
401:
398:
393:
392:
374:
372:
364:
360:
359:
354:
351:
341:
295:
267:
235:
199:
61:) reaction is a
21:
2428:
2427:
2423:
2422:
2421:
2419:
2418:
2417:
2398:
2397:
2396:
2391:
2377:Eyring equation
2318:
2289:Stereochemistry
2252:
2238:Solvent effects
2226:
2182:
2146:
2127:
2117:
2098:
2093:
2059:
2055:
2036:
2032:
2022:
2012:
2002:
1992:
1973:
1967:
1920:
1915:
1914:
1891:
1887:
1863:
1859:
1854:
1850:
1818:
1814:
1799:
1783:
1779:
1759:
1758:
1746:
1730:
1726:
1694:
1690:
1685:
1681:
1661:
1657:
1650:
1636:
1632:
1623:
1619:
1614:
1605:
1583:
1571:-butyl chloride
1526:
1522:
1517:
1513:
1512:
1504:
1501:
1500:
1494:
1459:protic solvents
1448:
1443:
1441:Solvent effects
1437:
1435:Solvent effects
1418:
1413:
1392:
1385:
1376:
1365:
1360:racemic mixture
1351:
1349:Stereochemistry
1344:
1340:
1316:
1305:
1281:
1273:
1268:
1261:
1251:
1242:
1218:
1209:
1205:
1191:
1185:
1180:
1179:
1170:
1166:
1165:
1156:
1150:
1145:
1144:
1133:
1124:
1120:
1114:
1110:
1109:
1107:
1094:
1085:
1078:
1076:
1068:
1066:
1063:
1062:
1041:
1035:
1030:
1029:
1020:
1016:
1004:
999:
998:
986:
982:
980:
977:
976:
956:
951:
950:
945:
942:
941:
916:
910:
905:
904:
895:
891:
879:
874:
873:
861:
857:
856:
847:
841:
836:
835:
824:
815:
811:
805:
801:
800:
798:
785:
776:
769:
767:
765:
762:
761:
740:
734:
729:
728:
716:
711:
710:
701:
697:
684:
675:
668:
666:
664:
661:
660:
636:
630:
625:
624:
615:
611:
599:
594:
593:
581:
577:
576:
567:
558:
554:
553:
551:
539:
534:
533:
528:
525:
524:
503:
497:
492:
491:
479:
474:
473:
464:
460:
448:
443:
442:
430:
425:
424:
412:
408:
397:
388:
384:
365:
355:
350:
349:
342:
340:
338:
335:
334:
311:
306:
301:
211:Formation of a
206:
171:
166:
158:
150:
128:
118:
76:
58:
44:
35:
28:
23:
22:
15:
12:
11:
5:
2426:
2416:
2415:
2410:
2393:
2392:
2390:
2389:
2384:
2379:
2374:
2369:
2364:
2359:
2354:
2349:
2344:
2339:
2334:
2328:
2326:
2320:
2319:
2317:
2316:
2311:
2306:
2301:
2296:
2291:
2286:
2281:
2276:
2271:
2266:
2260:
2258:
2257:Related topics
2254:
2253:
2251:
2250:
2245:
2240:
2234:
2232:
2231:Medium effects
2228:
2227:
2225:
2224:
2219:
2214:
2209:
2204:
2199:
2193:
2191:
2184:
2183:
2181:
2180:
2175:
2170:
2165:
2160:
2154:
2152:
2148:
2147:
2145:
2144:
2139:
2134:
2129:
2125:
2119:
2115:
2108:
2106:
2100:
2099:
2097:
2096:
2091:
2087:
2081:
2076:
2069:
2067:
2061:
2060:
2058:
2057:
2053:
2046:
2044:
2038:
2037:
2035:
2034:
2030:
2024:
2020:
2014:
2010:
2004:
2000:
1994:
1990:
1983:
1981:
1975:
1974:
1966:
1965:
1958:
1951:
1943:
1937:
1936:
1930:
1919:
1918:External links
1916:
1913:
1912:
1885:
1857:
1848:
1823:J. Chem. Educ.
1812:
1797:
1777:
1744:
1724:
1705:(3): 859–873.
1688:
1679:
1655:
1648:
1630:
1616:
1615:
1613:
1610:
1609:
1608:
1603:
1599:
1594:
1589:
1582:
1579:
1560:
1559:
1547:
1544:
1541:
1536:
1529:
1525:
1521:
1516:
1511:
1508:
1492:
1446:
1436:
1433:
1429:E2 elimination
1416:
1411:
1404:E1 elimination
1391:
1390:Side reactions
1388:
1383:
1374:
1363:
1350:
1347:
1342:
1338:
1335:
1334:
1314:
1308:tertiary alkyl
1303:
1294:from attached
1279:
1271:
1267:
1264:
1259:
1249:
1240:
1225:
1217:
1212:
1208:
1204:
1198:
1188:
1178:
1173:
1169:
1163:
1153:
1143:
1140:
1132:
1127:
1123:
1117:
1113:
1106:
1100:
1097:
1092:
1084:
1081:
1075:
1048:
1038:
1028:
1023:
1019:
1015:
1012:
1007:
997:
992:
989:
985:
964:
959:
949:
923:
913:
903:
898:
894:
890:
887:
882:
872:
867:
864:
860:
854:
844:
834:
831:
823:
818:
814:
808:
804:
797:
791:
788:
783:
775:
772:
747:
737:
727:
724:
719:
709:
704:
700:
696:
690:
687:
682:
674:
671:
643:
633:
623:
618:
614:
610:
607:
602:
592:
587:
584:
580:
574:
566:
561:
557:
550:
547:
542:
532:
510:
500:
490:
487:
482:
472:
467:
463:
459:
456:
451:
441:
438:
433:
423:
418:
415:
411:
407:
404:
396:
391:
387:
383:
380:
377:
371:
368:
363:
358:
348:
345:
309:
304:
300:
297:
289:
288:
269:
268:
260:
259:
247:
246:
236:
228:
227:
204:
201:
200:
169:
165:
162:
156:
148:
126:
116:
77:" stands for "
74:
56:
42:
26:
9:
6:
4:
3:
2:
2425:
2414:
2411:
2409:
2406:
2405:
2403:
2388:
2385:
2383:
2380:
2378:
2375:
2373:
2370:
2368:
2365:
2363:
2360:
2358:
2355:
2353:
2350:
2348:
2345:
2343:
2340:
2338:
2335:
2333:
2332:Rate equation
2330:
2329:
2327:
2325:
2321:
2315:
2312:
2310:
2307:
2305:
2304:Arrow pushing
2302:
2300:
2297:
2295:
2292:
2290:
2287:
2285:
2282:
2280:
2277:
2275:
2272:
2270:
2267:
2265:
2262:
2261:
2259:
2255:
2249:
2246:
2244:
2241:
2239:
2236:
2235:
2233:
2229:
2223:
2220:
2218:
2215:
2213:
2212:Marcus theory
2210:
2208:
2205:
2203:
2200:
2198:
2195:
2194:
2192:
2189:
2185:
2179:
2176:
2174:
2171:
2169:
2166:
2164:
2163:Isomerization
2161:
2159:
2156:
2155:
2153:
2149:
2143:
2140:
2138:
2137:Cycloaddition
2135:
2133:
2130:
2123:
2120:
2113:
2110:
2109:
2107:
2105:
2101:
2095:
2088:
2085:
2082:
2080:
2077:
2074:
2071:
2070:
2068:
2066:
2062:
2051:
2048:
2047:
2045:
2043:
2039:
2028:
2025:
2018:
2015:
2008:
2005:
1998:
1995:
1988:
1985:
1984:
1982:
1980:
1976:
1972:
1964:
1959:
1957:
1952:
1950:
1945:
1944:
1941:
1934:
1931:
1929:
1925:
1922:
1921:
1908:
1904:
1900:
1896:
1889:
1881:
1877:
1873:
1870:
1869:
1861:
1852:
1844:
1840:
1836:
1832:
1828:
1825:
1824:
1816:
1808:
1804:
1800:
1798:0-06-044084-8
1794:
1790:
1789:
1781:
1773:
1769:
1763:
1755:
1751:
1747:
1745:1-891389-31-9
1741:
1737:
1736:
1728:
1720:
1716:
1712:
1708:
1704:
1701:
1700:
1692:
1683:
1675:
1671:
1667:
1659:
1651:
1649:0-471-60180-2
1645:
1641:
1634:
1627:
1621:
1617:
1607:
1600:
1598:
1595:
1593:
1590:
1588:
1587:Arrow pushing
1585:
1584:
1578:
1576:
1572:
1570:
1565:
1545:
1542:
1539:
1534:
1527:
1523:
1519:
1514:
1509:
1506:
1499:
1498:
1497:
1495:
1488:
1484:
1480:
1476:
1472:
1467:
1465:
1461:
1460:
1455:
1454:
1442:
1432:
1430:
1426:
1422:
1409:
1405:
1401:
1397:
1387:
1371:
1367:
1361:
1356:
1346:
1332:
1328:
1327:
1326:
1324:
1320:
1311:
1309:
1301:
1297:
1293:
1289:
1285:
1284:steric strain
1277:
1263:
1257:
1256:
1245:
1236:
1210:
1206:
1202:
1186:
1171:
1167:
1151:
1125:
1121:
1115:
1111:
1104:
1098:
1095:
1079:
1073:
1060:
1036:
1021:
1017:
1013:
1005:
990:
987:
983:
957:
937:
911:
896:
892:
888:
880:
865:
862:
858:
842:
816:
812:
806:
802:
795:
789:
786:
770:
758:
735:
717:
702:
698:
694:
688:
685:
669:
657:
631:
616:
612:
608:
600:
585:
582:
578:
559:
555:
548:
540:
521:
498:
480:
465:
461:
457:
449:
431:
416:
413:
409:
405:
389:
385:
381:
378:
375:
369:
366:
356:
343:
331:
329:
325:
317:
313:
296:
294:
286:
285:hydronium ion
282:
278:
274:
273:Deprotonation
271:
270:
266:
262:
261:
257:
253:
249:
248:
241:
237:
234:
230:
229:
225:
221:
220:leaving group
217:
215:
210:
209:
208:
198:
194:
193:
192:
190:
188:
183:
179:
175:
161:
153:
146:
142:
138:
134:
133:
124:
120:
112:
108:
107:alkyl halides
104:
100:
96:
92:
91:rate equation
88:
84:
80:
72:
68:
64:
60:
52:
39:
33:
32:Sportsnet One
19:
2284:Molecularity
1986:
1901:(12): 2770.
1898:
1894:
1888:
1871:
1866:
1860:
1851:
1829:(1): 81–83.
1826:
1821:
1815:
1787:
1780:
1734:
1727:
1702:
1697:
1691:
1682:
1665:
1658:
1639:
1633:
1625:
1620:
1574:
1568:
1563:
1561:
1490:
1478:
1470:
1468:
1457:
1451:
1444:
1393:
1380:
1354:
1352:
1336:
1318:
1312:
1298:groups. The
1269:
1253:
1246:
1237:
1061:
938:
759:
658:
522:
332:
327:
321:
302:
290:
213:
202:
186:
167:
154:
130:
89:. Thus, the
87:unimolecular
54:
50:
48:
2243:Cage effect
2178:RRKM theory
2094:elimination
1473:correlates
1445:Since the S
1373:A typical S
324:carbocation
252:nucleophile
121:occurs. In
103:carbocation
95:nucleophile
2402:Categories
1874:(2): 846.
1699:Chem. Rev.
1612:References
1606:2 reaction
1496:) through
1475:solvolysis
1439:See also:
277:protonated
178:hydrolysis
137:cis effect
119:2 reaction
2294:Catalysis
2190:reactions
1762:cite book
1510:
1425:methoxide
1421:hydroxide
1310:centers.
1288:inductive
1014:≪
1006:−
988:−
958:−
881:−
863:−
601:−
583:−
458:−
450:−
414:−
406:−
164:Mechanism
1933:Exercise
1924:Diagrams
1807:14214254
1754:55600610
1719:17319730
1581:See also
299:Rate law
189:-butanol
184:forming
1831:Bibcode
1668:: 979.
1483:ethanol
1471:Y scale
1464:solvate
281:alcohol
256:solvent
224:bromide
176:is the
125:, the S
1969:Basic
1805:
1795:
1752:
1742:
1717:
1646:
1573:) and
1408:alkene
1341:2 to S
283:and a
216:-butyl
203:This S
69:. The
2197:Redox
2033:Acyl)
1562:with
1487:water
1453:polar
1296:alkyl
1278:the S
1270:The S
1266:Scope
1220:tBuBr
1135:tBuBr
1087:tBuOH
826:tBuBr
778:tBuOH
677:tBuOH
569:tBuBr
399:tBuBr
2086:(E2)
2075:(E1)
1803:OCLC
1793:ISBN
1772:link
1768:link
1750:OCLC
1740:ISBN
1715:PMID
1644:ISBN
1569:tert
1469:The
1462:(to
1398:and
1070:rate
328:tert
214:tert
187:tert
139:. A
49:The
2056:Ar)
2013:Ar)
1903:doi
1876:doi
1839:doi
1707:doi
1703:107
1670:doi
1507:log
1489:) (
1423:or
1345:1.
713:tBu
536:tBu
476:tBu
427:tBu
352:tBu
222:(a
180:of
85:is
65:in
18:SN1
2404::
2124:(A
2114:(A
2052:(S
2029:(S
2023:i)
2019:(S
2009:(S
2003:2)
1999:(S
1993:1)
1989:(S
1926::
1899:78
1897:.
1872:70
1837:.
1827:87
1801:.
1764:}}
1760:{{
1748:.
1713:.
1355:sp
1325::
1001:Br
953:Br
876:Br
596:Br
445:Br
326:,
191::
172:1
2128:)
2126:N
2118:)
2116:E
2092:i
2090:E
2054:E
2031:N
2021:N
2011:N
2001:N
1991:N
1962:e
1955:t
1948:v
1909:.
1905::
1882:.
1878::
1845:.
1841::
1833::
1809:.
1774:)
1756:.
1721:.
1709::
1676:.
1672::
1652:.
1604:N
1602:S
1575:Y
1564:m
1546:Y
1543:m
1540:=
1535:)
1528:0
1524:k
1520:k
1515:(
1493:0
1491:k
1485:/
1479:k
1447:N
1417:N
1415:S
1412:N
1384:N
1382:S
1375:N
1364:N
1343:N
1339:N
1315:N
1304:N
1280:N
1272:N
1260:N
1250:N
1241:2
1239:H
1224:]
1216:[
1211:1
1207:k
1203:=
1197:]
1193:O
1187:2
1182:H
1177:[
1172:2
1168:k
1162:]
1158:O
1152:2
1147:H
1142:[
1139:]
1131:[
1126:2
1122:k
1116:1
1112:k
1105:=
1099:t
1096:d
1091:]
1083:[
1080:d
1074:=
1047:]
1043:O
1037:2
1032:H
1027:[
1022:2
1018:k
1011:]
996:[
991:1
984:k
963:]
948:[
922:]
918:O
912:2
907:H
902:[
897:2
893:k
889:+
886:]
871:[
866:1
859:k
853:]
849:O
843:2
838:H
833:[
830:]
822:[
817:2
813:k
807:1
803:k
796:=
790:t
787:d
782:]
774:[
771:d
746:]
742:O
736:2
731:H
726:[
723:]
718:+
708:[
703:2
699:k
695:=
689:t
686:d
681:]
673:[
670:d
642:]
638:O
632:2
627:H
622:[
617:2
613:k
609:+
606:]
591:[
586:1
579:k
573:]
565:[
560:1
556:k
549:=
546:]
541:+
531:[
509:]
505:O
499:2
494:H
489:[
486:]
481:+
471:[
466:2
462:k
455:]
440:[
437:]
432:+
422:[
417:1
410:k
403:]
395:[
390:1
386:k
382:=
379:0
376:=
370:t
367:d
362:]
357:+
347:[
344:d
310:N
305:N
205:N
170:N
157:N
149:N
127:N
117:N
115:S
75:N
59:1
57:N
55:S
53:(
43:N
34:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.