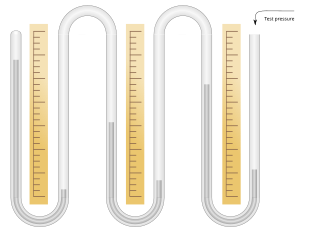
130:. The centre column is initially completely filled with mercury, as is the connecting capillary. The other two columns are partially filled. The top of both the main column on the right and the reservoir column on the left are connected together and to an inlet for the pressure to be measured. When the test pressure is applied, the mercury rises in both the left and right columns and falls in the centre column. The mercury at the top of the capillary breaks and a vacuum forms there. The pressure is then measured in the usual way by the difference between the heights of the right and centre columns.
143:
106:
20:
159:. This consisted of a series of mercury filled U-tubes connected together with inverted U-tubes. The inverted U-tubes contain compressed air at a pressure designed to bring the instrument into the pressure range of interest. The pressure is found from this instrument by summing together the difference in column heights in each of the U-tubes.
223:
are used to measure the mercury column height. However, in 2019 the backup gauge was decommissioned after being out of service for years. It was so large that it could not be removed by normal means; a hole was cut in the ceiling to extract it. The decommissioning was part of an international move
194:
The instrument was formerly widely used in education, laboratories, and medical measurements as well as its industrial applications. However, the toxicity of mercury and the risk of spills, through broken glassware, has led to its decline. It is also easier to interface other types of sensor to
125:
In 1938, Adolph
Zimmerli (1886–1967) invented a gauge that overcame the filling problems, at least for pressures below ambient pressure. Zimmerli's gauge consists of three relatively wide columns. Referring to the diagram, the columns in the centre and on the right function as a standard U-tube
150:
For extremely high pressures, the column can still be very high, even when using mercury. Gauges for measuring pressure in the range 20–30 standard atmospheres (15,000–23,000 mmHg) have been built. A 23-metre-tall mercury column is difficult to read and suffers from inaccuracies caused by
48:
The instrument consists of a glass U-tube half-filled with mercury. One end is connected to the vessel whose pressure is being measured. The other may be either left open or sealed. If it is left open, the pressure measured is relative to air pressure, which is variable. If it is sealed, the
77:
might be employed for accuracy. When relative pressure is being measured the difference may be negative, meaning the test pressure is below the reference pressure. The ubiquity of this instrument led to mmHg becoming a common unit of measure of pressure. It is also related to another unit of
191:. A gauge for use on steam engines very similar to the later Kamerlingh-Onnes gauge was patented in 1858 by Thomas Purssglove. Like the Kamerlingh-Onnes device, it had multiple U-tubes connected in series. The connecting tubes were filled with an incompressible fluid.
72:
The pressure is determined by measuring the difference in height between the reference column and the column connected to the item under test. Calibration marks are usually provided to aid in this measurement and in laboratories a
133:
Since a new vacuum is formed each time a measurement is made, there is no problem with the vacuum becoming contaminated. Any bubbles that do form in the capillary are easily removed by inverting the gauge and shaking or tapping.
39:
as the working fluid. The most basic form of this instrument is a U-shaped glass tube filled with mercury. More complex versions deal with very high pressure or have better means of filling with mercury.
117:
of the gauge to a vessel of mercury, pumping out the air and boiling the mercury. After filling, the gauge is then cut away again. Further, the vacuum in the gauge eventually deteriorates due to slow
578:
224:
to stop using mercury in standards laboratories for environmental reasons. NIST will eventually also take the main mercury gauge out of service after a portable
175:
in 1643. An early engineering application of the mercury pressure gauge was to measure pressure in steam boilers during the age of steam. The first use on
657:
Suski, J.; Puers, R.; Ehrlich, C.D.; Schmidt, J.W.; Abramson, E.H.; Sutton, C.M., "Pressure", ch. 3 in, Goodwin, A.R.H.; Marsh, K.N.; Wakeham, W.A. (eds),
53:. The tube is sealed during manufacture with the sealed end containing a vacuum. Mercury is a useful material to use in a manometer because of its high
574:
212:
151:
different parts of the column being at different temperatures. A more compact mercury pressure gauge suitable for high pressure was built by
57:. This means that a much shorter column is needed compared to water. For instance, the pressure represented by a column of
126:
gauge. Additionally, the top of the centre column is connected to the bottom of the third column on the left with a
208:
666:
651:
636:
593:
556:
541:
526:
508:
493:
563:
188:
693:
659:
Experimental
Thermodynamics (vol. 6): Measurement of the Thermodynamic Properties of Single Phases
172:
152:
617:
113:
The initial filling of a sealed gauge with mercury can be problematic. One method involves
142:
8:
570:, vol. 49–51, pp. 20–45, Associated Friends of the Library of Rutgers University, 1987.
662:
647:
632:
606:
589:
552:
537:
522:
504:
489:
184:
156:
50:
36:
673:
514:
204:
127:
240:, a type of mercury pressure gauge used for calibrating electronic pressure gauges
105:
600:
483:
195:
electronic systems. By 1991 it had mostly been replaced by other technologies.
499:
Hála, Eduard; Pick, Jiří; Fried, Vojtěch; Vilím, O.; Standart, George (trans),
687:
584:
Lindh, Wilburta Q.; Pooler, Marilyn S.; Tamparo, Carol D.; Dahl, Barbara M.,
93:
86:
but is still sometimes found in use, particularly in medicine. In SI units,
237:
176:
114:
74:
220:
19:
180:
215:(NIST) in the US uses a gauge that is three metres tall and contains
187:
between 1763 and 1775. This engine was a development of the popular
168:
119:
32:
610:
575:"No longer under pressure: NIST dismantles giant mercury manometer"
479:
225:
83:
54:
605:, United States Government Priniting Office, 1990 (1973 edition
622:
English
Patents of Inventions, Specifications: 1858, 2675-2752
624:, patent no. 2739, filed 1 December 1858, issued 31 May 1859.
122:
of gases through the mercury, making the device inaccurate.
79:
66:
678:
Industrial & Engineering
Chemistry: Analytical Edition
167:
The parent of all mercury pressure gauges is the mercury
485:
Mercury: A Global
Pollutant Requiring Global Initiatives
644:
Building
Technology: Mechanical and Electrical Systems
564:"The Waksman Institute of Microbiology, 1954 to 1984"
547:
Kotz, John C.; Treichel, Paul M.; Townsend, John R.,
577:, NIST, 28 June 2019/15 January 2020, retrieved and
685:
532:Kopp, Brian, "Industrial telemetry", ch. 18 in,
503:, Pergamon Press, 1967 (Elsevier reprint, 2013
521:, Springer Science & Business Media, 2007
213:National Institute of Standards and Technology
23:An open (differential) mercury pressure gauge
137:
680:, vol. 10, iss. 5, pp. 283–284, 1 May 1938.
203:Mercury gauges are commonly used as the
141:
104:
18:
386:
146:Kamerlingh Onnes mercury pressure gauge
100:
686:
568:The Journal of the Rutgers University
488:, Nordic Council of Ministers, 2002
198:
586:Delmar's Clinical Medical Assisting
482:Consulting Engineers and Planners,
228:device is installed to replace it.
13:
209:measurement standards laboratories
14:
705:
551:, vol. 1, Cengage Learning, 2008
549:Chemistry and Chemical Reactivity
109:Zimmerli's mercury pressure gauge
473:
461:
448:
439:
430:
421:
412:
399:
373:
360:
351:
646:, John Wiley & Sons, 1996
629:Experiments in Fluid Mechanics
342:
329:
316:
303:
294:
281:
272:
263:
250:
43:
1:
616:Purssglove, Thomas Paramore,
534:Telemetry Systems Engineering
348:<Rutgers author>, p. 34
244:
674:"An improved mercury U-gage"
219:of mercury. For precision,
7:
231:
189:Newcomen atmospheric engine
10:
710:
162:
588:, Cengage Learning, 2009
519:Physics of the Human Body
501:Vapour–Liquid Equilibrium
207:for pressure by national
138:High pressure measurement
49:pressure measured is the
562:Lechevalier, Hubert A.,
602:Instrumentman 1 & C
61:of water is just under
573:Lee, Jennifer Lauren,
173:Evangelista Torricelli
153:Heike Kamerlingh Onnes
147:
110:
82:. The mmHg is not an
29:mercury pressure gauge
24:
631:, PHI Learning, 2012
536:, Artech House, 2002
211:. For instance, the
183:while developing the
145:
108:
22:
155:, the discoverer of
101:Filling with mercury
672:Zimmerli, Adolph,
148:
111:
25:
661:, Elsevier, 2003
642:Stein, Benjamin,
599:Mack, Donald M.,
445:COWI, pp. 138–139
199:Use as a standard
185:Watt steam engine
157:superconductivity
90:is approximately
51:absolute pressure
16:Type of manometer
701:
627:Singh, Sarbjit,
618:"Pressure gauge"
468:
465:
459:
452:
446:
443:
437:
436:Purssglove, p. 1
434:
428:
425:
419:
416:
410:
403:
397:
390:
384:
377:
371:
364:
358:
357:Zimmerli, p. 283
355:
349:
346:
340:
333:
327:
320:
314:
307:
301:
298:
292:
285:
279:
276:
270:
267:
261:
254:
218:
205:primary standard
115:fusing the glass
96:
89:
64:
60:
709:
708:
704:
703:
702:
700:
699:
698:
694:Pressure gauges
684:
683:
581:29 August 2020.
476:
471:
466:
462:
453:
449:
444:
440:
435:
431:
426:
422:
417:
413:
404:
400:
391:
387:
378:
374:
365:
361:
356:
352:
347:
343:
334:
330:
321:
317:
308:
304:
299:
295:
286:
282:
277:
273:
268:
264:
255:
251:
247:
234:
216:
201:
165:
140:
103:
91:
87:
62:
58:
46:
17:
12:
11:
5:
707:
697:
696:
682:
681:
670:
655:
640:
625:
614:
597:
582:
571:
560:
545:
530:
515:Herman, Irving
512:
497:
475:
472:
470:
469:
460:
447:
438:
429:
420:
411:
398:
385:
372:
359:
350:
341:
328:
315:
302:
293:
280:
271:
262:
248:
246:
243:
242:
241:
233:
230:
200:
197:
164:
161:
139:
136:
128:capillary tube
102:
99:
78:pressure, the
45:
42:
15:
9:
6:
4:
3:
2:
706:
695:
692:
691:
689:
679:
675:
671:
668:
664:
660:
656:
653:
649:
645:
641:
638:
634:
630:
626:
623:
619:
615:
612:
608:
604:
603:
598:
595:
591:
587:
583:
580:
576:
572:
569:
565:
561:
558:
554:
550:
546:
543:
539:
535:
531:
528:
524:
520:
516:
513:
510:
506:
502:
498:
495:
491:
487:
486:
481:
478:
477:
464:
457:
451:
442:
433:
424:
418:Mack, p. 3-12
415:
408:
402:
395:
389:
382:
376:
370:, pp. 211–212
369:
363:
354:
345:
339:, pp. 209–211
338:
332:
325:
319:
312:
306:
297:
290:
284:
278:Herman, p. 82
275:
269:Stein, p. 409
266:
260:, pp. 207–209
259:
253:
249:
239:
236:
235:
229:
227:
222:
214:
210:
206:
196:
192:
190:
186:
182:
178:
177:steam engines
174:
170:
160:
158:
154:
144:
135:
131:
129:
123:
121:
116:
107:
98:
95:
85:
81:
76:
70:
68:
56:
52:
41:
38:
34:
31:is a type of
30:
21:
677:
658:
643:
628:
621:
601:
585:
567:
548:
533:
518:
500:
484:
474:Bibliography
463:
455:
450:
441:
432:
427:Kopp, p. 494
423:
414:
406:
401:
393:
388:
380:
375:
367:
362:
353:
344:
336:
331:
323:
318:
310:
305:
296:
288:
283:
274:
265:
257:
252:
238:McLeod gauge
202:
193:
171:invented by
166:
149:
132:
124:
112:
75:cathetometer
71:
65:of mercury (
47:
28:
26:
300:Singh, p. 8
221:ultrasonics
44:Description
667:008053144X
652:0471593192
637:8120345118
594:1435419251
557:0495387118
542:1580532578
527:3540296042
509:1483160866
494:9289307544
245:References
181:James Watt
611:73-603249
169:barometer
120:diffusion
33:manometer
688:Category
579:archived
409:, p. 220
396:, p. 220
383:, p. 212
326:, p. 516
313:, p. 271
291:, p. 208
232:See also
226:photonic
458:, p. 75
179:was by
163:History
84:SI unit
55:density
37:mercury
665:
650:
635:
609:
592:
555:
540:
525:
507:
492:
456:et al.
454:Suski
407:et al.
394:et al.
381:et al.
368:et al.
337:et al.
324:et al.
311:et al.
309:Lindh
289:et al.
258:et al.
217:225 kg
88:1 mmHg
63:7.4 mm
59:100 mm
35:using
405:Hála
392:Hála
379:Hála
366:Hála
335:Hála
322:Kotz
287:Hála
256:Hála
663:ISBN
648:ISBN
633:ISBN
607:LCCN
590:ISBN
553:ISBN
538:ISBN
523:ISBN
505:ISBN
490:ISBN
480:COWI
92:133
80:torr
67:mmHg
467:Lee
94:Pa
69:).
690::
676:,
620:,
613:).
566:,
517:,
511:).
97:.
27:A
669:.
654:.
639:.
596:.
559:.
544:.
529:.
496:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.