20:
119:
529:
429:
136:
304:
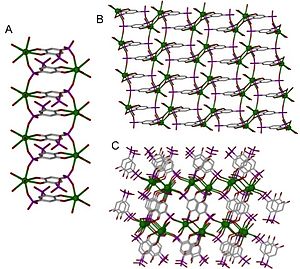
67:. A structure can be determined to be one-, two- or three-dimensional, depending on the number of directions in space the array extends in. A one-dimensional structure extends in a straight line (along the x axis); a two-dimensional structure extends in a plane (two directions, x and y axes); and a three-dimensional structure extends in all three directions (x, y, and z axes). This is depicted in Figure 1.
53:. Coordination polymers contain the subclass coordination networks that are coordination compounds extending, through repeating coordination entities, in 1 dimension, but with cross-links between two or more individual chains, loops, or spiro-links, or a coordination compound extending through repeating coordination entities in 2 or 3 dimensions. A subclass of these are the
595:, and blue upon the addition of diethyl ether. The polymer can thus act as a solvent sensor that physically changes color in the presence of certain solvents. The color changes are attributed to the incoming solvent displacing the water ligands on the cobalt atoms, resulting in a change of their geometry from octahedral to tetrahedral.
391:
400:
bonds with the surrounding lattice, but sometimes interact via intermolecular forces, such as hydrogen bonding or pi stacking. Most often, the guest molecule will be the solvent that the coordination polymer was crystallized in, but can really be anything (other salts present, atmospheric gases such as
494:
Flexible porous coordination polymers are potentially attractive for molecular storage, since their pore sizes can be altered by physical changes. An example of this might be seen in a polymer that contains gas molecules in its normal state, but upon compression the polymer collapses and releases the
399:
The structure of coordination polymers often incorporates empty space in the form of pores or channels. This empty space is thermodynamically unfavorable. In order to stabilize the structure and prevent collapse, the pores or channels are often occupied by guest molecules. Guest molecules do not form
1022:
Carlucci, L.; Ciani, G.; Proserpio, D. M.; Rizzato, S. (2002). "New polymeric networks from the self-assembly of silver(i) salts and the flexible ligand 1,3-bis(4-pyridyl)propane (bpp). A systematic investigation of the effects of the counterions and a survey of the coordination polymers based on
126:
The structure and dimensionality of the coordination polymer are determined by the linkers and the coordination geometry of the metal center. Coordination numbers are most often between 2 and 10. Examples of various coordination numbers are shown in planar geometry in Figure 2. In Figure 1 the 1D
577:
are cooperative phenomena of the magnetic spins within a solid arising from coupling between the spins of the paramagnetic centers. In order to allow efficient magnetic, metal ions should be bridged by small ligands allowing for short metal-metal contacts (such as oxo, cyano, and azido bridges).
590:
molecules incorporated into the structure. An example of this would be the two Co coordination polymers of the cluster that contains water ligands that coordinate to the cobalt atoms. This originally orange solution turns either purple or green with the replacement of water with
394:
The addition and removal of guest molecules can have a large effect on the resulting structure of a coordination polymer. A few examples are (top) change of a linear 1D chain to a zigzag pattern, (middle) staggered 2D sheets to stacked, and (bottom) 3D cubes become more widely
511:
Luminescent coordination polymers typically feature organic chromophoric ligands, which absorb light and then pass the excitation energy to the metal ion. For ligands that fluoresce without the presence of the metal linker (not due to LMCT), the intense
259:
from the salt used in synthesis, which is difficult to avoid. The coordination polymers shown in Figure 3 are all group two metals. In this case, the dimensionality of these structures increases as the radius of the metal increases down the group (from
319:
create more variety in the structure. There are examples of coordination polymers that include two configurations of the same ligand within one structure, as well as two separate structures where the only difference between them is ligand orientation.
552:
behavior. Three-dimensional structures consisting of sheets of silver-containing polymers demonstrate semi-conductivity when the metal centers are aligned, and conduction decreases as the silver atoms go from parallel to perpendicular.
238:
are large atoms with coordination numbers varying from 7 to 14. Their coordination environment can be difficult to predict, making them challenging to use as nodes. They offer the possibility of incorporating luminescent components.
139:
Figure 3. Three coordination polymers of different dimensionality. All three were made using the same ligand (4,5-dihydroxybenzene-1,3-disulfonate (L)), but different metal cations. All of the metals come from Group 2 on the
495:
stored molecules. Depending on the structure of the polymer, it is possible that the structure be flexible enough that collapsing the pores is reversible and the polymer can be reused to uptake the gas molecules again. The
314:
Ligands can be flexible or rigid. A rigid ligand is one that has no freedom to rotate around bonds or reorient within a structure. Flexible ligands can bend, rotate around bonds, and reorient themselves. These different
1107:
Tong, M. L.; Hu, S.; Wang, J.; Kitagawa, S.; Ng, S. W. (2005). "Supramolecular
Isomerism in Cadmium Hydroxide Phases. Temperature-Dependent Synthesis and Structure of Photoluminescent Coordination Polymers of α- and
893:
Hung-Low, F.; Klausmeyer, K. K.; Gary, J. B. (2009). "Effect of anion and ligand ratio in self-assembled silver(I) complexes of 4-(diphenylphosphinomethyl)pyridine and their derivatives with bipyridine ligands".
254:
exist as stable cations. Alkali metals readily form cations with stable valence shells, giving them different coordination behavior than lanthanides and transition metals. They are strongly affected by the
382:, exposure to light, or changes in temperature can all change the resulting structure. Influences on the structure based on changes in crystallization environment are determined on a case by case basis.
341:
Besides metal and ligand choice, there are many other factors that affect the structure of the coordination polymer. For example, most metal centers are positively charged ions which exist as salts. The
866:
Wen, M.; Munakata, M.; Suenaga, Y.; Kuroda-Sowa, T.; Maekawa, M.; Yan, S. G. (2001). "Silver(I) coordination polymers of cyclic sulfur ligand, 2,2′,3,3′-tetrahydro-4,4′-dithia-1,1′-binaphthylidene".
79:
and his contemporaries laid the groundwork for the study of coordination polymers. Many time-honored materials are now recognized as coordination polymers. These include the cyanide complexes
544:. Some one-dimensional coordination polymers built as shown in the figure exhibit conductivities in a range of 1x10 to 2x10 S/cm. The conductivity is due to the interaction between the metal
921:
Ricci, G.; Sommazzi, A.; Masi, F.; Ricci, M.; Boglia, A.; Leone, G. (2010). "Well-defined transition metal complexes with phosphorus and nitrogen ligands for 1,3-dienes polymerization".
370:
are all crystallized with the same ligand, the structures vary in terms of the coordination environment of the metal, as well as the dimensionality of the entire coordination polymer.
446:. These materials crystallize with small aromatic guests (benzene, certain xylenes), and this selectivity has been exploited commercially for the separation of these hydrocarbons.
614:
Batten, Stuart R.; Champness, Neil R.; Chen, Xiao-Ming; Garcia-Martinez, Javier; Kitagawa, Susumu; Öhrström, Lars; O'Keeffe, Michael; Suh, Myunghyun P.; Reedijk, Jan (2013).
806:
Cote, A; Shimizu, G. (2003). "Coordination Solids via
Assembly of Adaptable Components : Systematic Structural Variation in Alkaline Earth Organosulfonate Networks".
328:
A length of the ligand can be an important factor in determining possibility for formation of a polymeric structure versus non-polymeric (mono- or oligomeric) structures.
307:
532:
Structure of coordination polymers that exhibit conductivity, where M = Fe, Ru, OS; L = octaethylporphyrinato or pthalocyaninato; N belongs to pyrazine or bipyridine.
284:
are known. They range from polyfunctional heterocycles, such as pyrazine, to simple halides. Almost any type of atom with a lone pair of electrons can serve as a
1052:"PH- and mol-ratio dependent formation of zinc(II) coordination polymers with iminodiacetic acid: Synthesis, spectroscopic, crystal structure and thermal studies"
516:
emission of these materials tend to be magnitudes of order higher than that of the free ligand alone. These materials are candidates for light emitting diode (
188:, use the Schrödinger equation to predict and explain coordination geometry, however this is difficult in part because of the complex effect of environment on
164:
Metal centers, often called nodes or hubs, bond to a specific number of linkers at well defined angles. The number of linkers bound to a node is known as the
1302:
Leong, Wei Lee; Vittal, Jagadese J. (2011). "One-Dimensional
Coordination Polymers: Complexity and Diversity in Structures, Properties, and Applications".
420:
Coordination polymers are found in some commercialized as dyes.. Metal complex dyes using copper or chromium are commonly used for producing dull colors.
520:) devices. The dramatic increase in fluorescence is caused by the increase in rigidity and asymmetry of the ligand when coordinated to the metal center.
63:
Coordination polymers can be classified in a number of ways according to their structure and composition. One important classification is referred to as
948:
Knaust, J. M.; Keller, S. W. (2002). "A Mixed-Ligand
Coordination Polymer from the in Situ, Cu(I)-Mediated Isomerization of Bis(4-pyridyl)ethylene".
412:, etc.) The presence of the guest molecule can sometimes influence the structure by supporting a pore or channel, where otherwise none would exist.
280:
Coordination polymers require ligands with the ability to form multiple coordination bonds, i.e. act as bridges between metal centers. Many
46:. More formally a coordination polymer is a coordination compound with repeating coordination entities extending in 1, 2, or 3 dimensions.
779:
Robin, A. Y.; Fromm, K. M. (2006). "Coordination polymer networks with O- and N-donors: What they are, why and how they are made".
536:
Coordination polymers can have short inorganic and conjugated organic bridges in their structures, which provide pathways for
1286:
850:
714:
Chen, X; Ye, B.; Tong, M. (2005). "Metal-organic molecular architectures with 2,2′-bipyridyl-like and carboxylate ligands".
650:
Biradha, Kumar; Ramanan, Arunachalam; Vittal, Jagadese J. (2009). "Coordination
Polymers Versus Metal−Organic Frameworks".
541:
479:
in the porous coordination polymer space to decrease the pore size. Active surface guests can also be used contribute to
983:
Buvailo, Andrii I.; Gumienna-Kontecka, Elzbieta; Pavlova, Svetlana V.; Fritsky, Igor O.; Haukka, Matti (2010). "Dimeric
487:
molecules (6.83 Å in diameter) or polymers with a highly conjugated system in order to increase the surface area for H
1347:
615:
467:. The size and shapes of the pore can be controlled by the linker size and the connecting ligands' length and
180:
around it, and in general the coordination number increases with cation size. Several models, most notably
434:
Some early commercialized coordination polymers are the
Hofmann compounds, which have the formula Ni(CN)
227:, particularly copper and gold ions which as neutral atoms have full d-orbitals in their outer shells.
987:
polymeric coordination in copper(ii) cationic complexes with bis(chelating) oxime and amide ligands".
378:
Additionally, variations in the crystallization environment can also change the structure. Changes in
476:
168:, which, along with the angles they are held at, determines the dimensionality of the structure. The
472:
316:
185:
36:
496:
112:
54:
616:"Terminology of metal–organic frameworks and coordination polymers (IUPAC Recommendations 2013)"
537:
251:
224:
216:
181:
173:
145:
1218:
1063:
220:
57:, or MOFs, that are coordination networks with organic ligands containing potential voids.
50:
1163:
Hunger, K.; Mischke, P.; Rieper, W.; Raue, R.; Kunde, K.; Engel, A. (2002) "Azo Dyes." In
744:
Kitagawa, S.; Kitaura, R.; Noro, S. I. (2004). "Functional Porous
Coordination Polymers".
424:
dyes are useful because they are more stable than their bi- or mono-dentate counterparts.
8:
346:
in the salt can affect the overall structure. For example, when silver salts such as AgNO
169:
165:
108:
1222:
1067:
60:
Coordination polymers are relevant to many fields, having many potential applications.
1239:
1230:
1206:
1084:
1051:
566:
548:
and the pi* level of the bridging ligand. In some cases coordination polymers can have
148:) and in this case, dimensionality increases with cation size and polarizability. A. •H
84:
879:
127:
structure is 2-coordinated, the planar is 4-coordinated, and the 3D is 6-coordinated.
1342:
1319:
1282:
1244:
1089:
1004:
965:
846:
823:
761:
681:
513:
421:
200:
32:
1311:
1274:
1234:
1226:
1189:
1168:
1147:
1125:
1079:
1071:
1032:
996:
957:
930:
903:
875:
815:
788:
753:
723:
696:
659:
630:
468:
189:
177:
104:
592:
460:
281:
100:
471:. To modify the pore size in order to achieve effective adsorption, nonvolatile
19:
1075:
1050:
Ni, L. B.; Zhang, R. H.; Liu, Q. X.; Xia, W. S.; Wang, H.; Zhou, Z. H. (2009).
840:
574:
570:
409:
204:
141:
64:
934:
907:
792:
727:
700:
1336:
1193:
1172:
1151:
841:
Bernstein, Jeremy; Paul M. Fishbane; Stephen G. Gasiorowicz (April 3, 2000).
635:
549:
310:
is a flexible ligand, which can exist in either gauche or anti conformations.
247:
96:
80:
76:
483:. For example, the large-pore MOF-177, 11.8 Å in diameter, can be doped by C
1323:
1248:
1093:
1008:
969:
827:
819:
765:
757:
459:
Although not yet practical, porous coordination polymers have potential as
1278:
528:
118:
235:
122:
Figure 2. Shows planar geometries with 3 coordination and 6 coordination.
390:
1000:
480:
343:
292:
256:
1315:
1129:
982:
961:
663:
428:
135:
1036:
586:
Coordination polymers can also show color changes upon the change of
562:
545:
265:
1207:"Chemistry and application of flexible porous coordination polymers"
405:
613:
176:
of a metal center is determined by the nonuniform distribution of
587:
464:
261:
43:
39:
401:
285:
269:
16:
Polymer consisting of repeating units of a coordination complex
303:
1021:
865:
49:
It can also be described as a polymer whose repeat units are
1142:
Grychtol, K.; Mennicke, W. (2002) "Metal-Complex Dyes." In
208:
1204:
242:
517:
212:
778:
23:
Figure 1. An illustration of 1- 2- and 3-dimensionality.
1271:
Coordination
Polymers: Design, Analysis and Application
682:"Coordination polymer networks with s-block metal ions"
379:
920:
892:
649:
42:
structure containing metal cation centers linked by
1205:Bureekaew, S.; Shimomura, S.; Kitagawa, S. (2008).
743:
291:Very elaborate ligands have been investigated. and
1106:
713:
160:O In each case, the metal is represented in green.
1268:
1334:
1184:Atwood, J. L. (2012) "Inclusion Compounds" in
805:
203:are commonly used as nodes. Partially filled d
1186:Ullmann's Encyclopedia of Industrial Chemistry
1165:Ullmann's Encyclopedia of Industrial Chemistry
1144:Ullmann's Encyclopedia of Industrial Chemistry
373:
1273:. RSC Publishing. pp. 297–307, 396–407.
1049:
799:
1211:Science and Technology of Advanced Materials
947:
561:Coordination polymers exhibit many kinds of
540:. example of such coordination polymers are
95:Coordination polymers are often prepared by
90:
1301:
1100:
219:differently depending on environment. This
914:
739:
737:
609:
607:
523:
499:page has a detailed section dealing with H
1238:
1083:
634:
298:
1264:
1262:
1260:
1258:
834:
527:
389:
302:
223:causes some of them to exhibit multiple
134:
117:
18:
1178:
886:
746:Angewandte Chemie International Edition
734:
675:
673:
604:
243:Alkali metals and alkaline earth metals
1335:
941:
1255:
859:
679:
1198:
670:
581:
454:
195:
707:
542:conductive metal organic frameworks
463:in parallel with porous carbon and
13:
449:
385:
14:
1359:
107:with a ligand. The mechanisms of
1056:Journal of Solid State Chemistry
427:
331:
323:
130:
1295:
1157:
1136:
1043:
1015:
976:
506:
415:
923:Coordination Chemistry Reviews
845:. Prentice-Hall. p. 624.
772:
643:
230:
1:
880:10.1016/S0020-1693(01)00556-4
598:
336:
1231:10.1088/1468-6996/9/1/014108
556:
7:
1118:Crystal Growth & Design
652:Crystal Growth & Design
374:Crystallization environment
10:
1364:
1269:Batten, Stuart R. (2008).
1076:10.1016/j.jssc.2009.06.042
623:Pure and Applied Chemistry
275:
70:
935:10.1016/j.ccr.2009.09.023
908:10.1016/j.ica.2008.04.032
793:10.1016/j.ccr.2006.02.013
728:10.1016/j.ccr.2004.07.006
701:10.1016/j.ccr.2007.10.032
91:Synthesis and propagation
1194:10.1002/14356007.a14_119
1173:10.1002/14356007.a03_245
1152:10.1002/14356007.a16_299
636:10.1351/PAC-REC-12-11-20
308:1,2-Bis(4-pyridyl)ethane
186:molecular orbital theory
55:metal-organic frameworks
1188:. Wiley-VCH, Weinheim.
896:Inorganica Chimica Acta
868:Inorganica Chimica Acta
524:Electrical conductivity
497:Metal-organic framework
225:coordination geometries
113:molecular self-assembly
820:10.1002/chem.200305102
758:10.1002/anie.200300610
533:
396:
311:
299:Structural orientation
295:, have been observed.
161:
123:
51:coordination complexes
24:
1348:Coordination polymers
1279:10.1039/9781847558862
538:electrical conduction
531:
393:
306:
252:alkaline earth metals
174:coordination geometry
146:alkaline earth metals
138:
121:
22:
787:(15–16): 2127–2157.
221:electronic structure
29:coordination polymer
1223:2008STAdM...9a4108B
1068:2009JSSCh.182.2698N
989:Dalton Transactions
950:Inorganic Chemistry
170:coordination number
166:coordination number
109:crystal engineering
1001:10.1039/C0DT00008F
680:Fromm, K. (2008).
567:Antiferromagnetism
534:
397:
312:
162:
124:
85:Hofmann clathrates
25:
1316:10.1021/cr100160e
1288:978-0-85404-837-3
1130:10.1021/cg049610r
1062:(10): 2698–2706.
962:10.1021/ic025836c
852:978-0-13-955311-0
814:(21): 5361–5370.
752:(18): 2334–2375.
664:10.1021/cg801381p
582:Sensor capability
514:photoluminescence
469:functional groups
455:Molecular storage
422:Tridentate ligand
201:Transition metals
196:Transition metals
1355:
1328:
1327:
1304:Chemical Reviews
1299:
1293:
1292:
1266:
1253:
1252:
1242:
1202:
1196:
1182:
1176:
1161:
1155:
1140:
1134:
1133:
1104:
1098:
1097:
1087:
1047:
1041:
1040:
1037:10.1039/b201288j
1019:
1013:
1012:
980:
974:
973:
945:
939:
938:
918:
912:
911:
890:
884:
883:
874:(1–2): 133–137.
863:
857:
856:
838:
832:
831:
803:
797:
796:
781:Coord. Chem. Rev
776:
770:
769:
741:
732:
731:
722:(5–6): 545–565.
716:Coord. Chem. Rev
711:
705:
704:
695:(8–9): 856–885.
689:Coord. Chem. Rev
686:
677:
668:
667:
658:(7): 2969–2970.
647:
641:
640:
638:
620:
611:
461:molecular sieves
431:
282:bridging ligands
207:, either in the
190:electron density
178:electron density
1363:
1362:
1358:
1357:
1356:
1354:
1353:
1352:
1333:
1332:
1331:
1300:
1296:
1289:
1267:
1256:
1203:
1199:
1183:
1179:
1162:
1158:
1141:
1137:
1115:
1111:
1105:
1101:
1048:
1044:
1020:
1016:
995:(27): 6266–75.
981:
977:
946:
942:
919:
915:
891:
887:
864:
860:
853:
839:
835:
804:
800:
777:
773:
742:
735:
712:
708:
684:
678:
671:
648:
644:
618:
612:
605:
601:
593:tetrahydrofuran
584:
559:
526:
509:
502:
490:
486:
457:
452:
450:Research trends
445:
441:
437:
418:
388:
386:Guest molecules
376:
369:
365:
361:
357:
353:
349:
339:
334:
326:
301:
278:
245:
233:
198:
159:
155:
151:
133:
101:crystallization
93:
73:
17:
12:
11:
5:
1361:
1351:
1350:
1345:
1330:
1329:
1310:(2): 688–764.
1294:
1287:
1254:
1197:
1177:
1156:
1146:. Wiley-VCH.
1135:
1113:
1109:
1099:
1042:
1014:
975:
956:(22): 5650–2.
940:
913:
885:
858:
851:
843:Modern Physics
833:
798:
771:
733:
706:
669:
642:
602:
600:
597:
583:
580:
575:ferromagnetism
571:ferrimagnetism
558:
555:
525:
522:
508:
505:
500:
488:
484:
456:
453:
451:
448:
443:
439:
435:
417:
414:
410:carbon dioxide
387:
384:
375:
372:
367:
363:
359:
355:
351:
347:
338:
335:
333:
330:
325:
322:
300:
297:
277:
274:
244:
241:
232:
229:
197:
194:
192:distribution.
157:
153:
149:
142:periodic table
132:
129:
115:are relevant.
92:
89:
72:
69:
65:dimensionality
37:organometallic
15:
9:
6:
4:
3:
2:
1360:
1349:
1346:
1344:
1341:
1340:
1338:
1325:
1321:
1317:
1313:
1309:
1305:
1298:
1290:
1284:
1280:
1276:
1272:
1265:
1263:
1261:
1259:
1250:
1246:
1241:
1236:
1232:
1228:
1224:
1220:
1217:(1): 014108.
1216:
1212:
1208:
1201:
1195:
1191:
1187:
1181:
1174:
1170:
1167:. Wiley-VCH.
1166:
1160:
1153:
1149:
1145:
1139:
1131:
1127:
1123:
1119:
1116:(2,4-pyda)".
1103:
1095:
1091:
1086:
1081:
1077:
1073:
1069:
1065:
1061:
1057:
1053:
1046:
1038:
1034:
1030:
1026:
1018:
1010:
1006:
1002:
998:
994:
990:
986:
979:
971:
967:
963:
959:
955:
951:
944:
936:
932:
928:
924:
917:
909:
905:
901:
897:
889:
881:
877:
873:
869:
862:
854:
848:
844:
837:
829:
825:
821:
817:
813:
809:
802:
794:
790:
786:
782:
775:
767:
763:
759:
755:
751:
747:
740:
738:
729:
725:
721:
717:
710:
702:
698:
694:
690:
683:
676:
674:
665:
661:
657:
653:
646:
637:
632:
628:
624:
617:
610:
608:
603:
596:
594:
589:
579:
576:
572:
568:
564:
554:
551:
550:semiconductor
547:
543:
539:
530:
521:
519:
515:
504:
503:gas storage.
498:
492:
482:
478:
474:
470:
466:
462:
447:
432:
430:
425:
423:
413:
411:
407:
403:
392:
383:
381:
371:
345:
332:Other factors
329:
324:Ligand length
321:
318:
317:conformations
309:
305:
296:
294:
289:
287:
283:
273:
271:
267:
263:
258:
253:
249:
248:Alkali metals
240:
237:
228:
226:
222:
218:
214:
210:
206:
202:
193:
191:
187:
183:
182:hybridization
179:
175:
171:
167:
147:
143:
137:
131:Metal centers
128:
120:
116:
114:
110:
106:
102:
98:
97:self-assembly
88:
86:
82:
81:Prussian blue
78:
77:Alfred Werner
68:
66:
61:
58:
56:
52:
47:
45:
41:
38:
34:
30:
21:
1307:
1303:
1297:
1270:
1214:
1210:
1200:
1185:
1180:
1164:
1159:
1143:
1138:
1121:
1117:
1102:
1059:
1055:
1045:
1028:
1025:CrystEngComm
1024:
1017:
992:
988:
984:
978:
953:
949:
943:
929:(5–6): 661.
926:
922:
916:
899:
895:
888:
871:
867:
861:
842:
836:
811:
808:Chem. Eur. J
807:
801:
784:
780:
774:
749:
745:
719:
715:
709:
692:
688:
655:
651:
645:
626:
622:
585:
560:
535:
510:
507:Luminescence
493:
491:adsorption.
477:intercalated
458:
433:
426:
419:
416:Applications
398:
377:
340:
327:
313:
290:
279:
246:
234:
199:
163:
125:
99:, involving
94:
75:The work of
74:
62:
59:
48:
28:
26:
1031:(22): 121.
629:(8): 1715.
236:Lanthanides
231:Lanthanides
103:of a metal
1337:Categories
1124:(3): 837.
902:(2): 426.
599:References
481:adsorption
344:counterion
337:Counterion
293:phosphorus
257:counterion
184:model and
563:magnetism
557:Magnetism
546:d-orbital
366:and AgSbF
266:strontium
217:hybridize
33:inorganic
1343:Polymers
1324:20804195
1249:27877934
1094:20161370
1009:20520918
970:12401066
828:14613146
766:15114565
465:zeolites
406:nitrogen
205:orbitals
1240:5099803
1219:Bibcode
1085:2778864
1064:Bibcode
588:solvent
395:spaced.
362:, AgAsF
354:, AgClO
276:Ligands
262:calcium
152:O B. •H
71:History
44:ligands
40:polymer
1322:
1285:
1247:
1237:
1092:
1082:
1023:bpp".
1007:
985:versus
968:
849:
826:
764:
573:, and
473:guests
402:oxygen
358:, AgPF
350:, AgBF
286:ligand
270:barium
215:, can
156:O C.•H
31:is an
685:(PDF)
619:(PDF)
438:Ni(NH
1320:PMID
1283:ISBN
1245:PMID
1112:(OH)
1108:β-Cd
1090:PMID
1005:PMID
966:PMID
847:ISBN
824:PMID
762:PMID
475:are
250:and
209:atom
172:and
111:and
105:salt
83:and
1312:doi
1308:111
1275:doi
1235:PMC
1227:doi
1190:doi
1169:doi
1148:doi
1126:doi
1080:PMC
1072:doi
1060:182
1033:doi
997:doi
958:doi
931:doi
927:254
904:doi
900:362
876:doi
872:322
816:doi
789:doi
785:250
754:doi
724:doi
720:249
697:doi
693:252
660:doi
631:doi
518:LED
272:).
268:to
264:to
213:ion
211:or
35:or
1339::
1318:.
1306:.
1281:.
1257:^
1243:.
1233:.
1225:.
1213:.
1209:.
1120:.
1088:.
1078:.
1070:.
1058:.
1054:.
1027:.
1003:.
993:39
991:.
964:.
954:41
952:.
925:.
898:.
870:.
822:.
810:.
783:.
760:.
750:43
748:.
736:^
718:.
691:.
687:.
672:^
654:.
627:85
625:.
621:.
606:^
569:,
565:.
485:60
408:,
404:,
380:pH
288:.
87:.
27:A
1326:.
1314::
1291:.
1277::
1251:.
1229::
1221::
1215:9
1192::
1175:.
1171::
1154:.
1150::
1132:.
1128::
1122:5
1114:2
1110:2
1096:.
1074::
1066::
1039:.
1035::
1029:4
1011:.
999::
972:.
960::
937:.
933::
910:.
906::
882:.
878::
855:.
830:.
818::
812:9
795:.
791::
768:.
756::
730:.
726::
703:.
699::
666:.
662::
656:9
639:.
633::
501:2
489:2
444:2
442:)
440:3
436:4
368:6
364:6
360:6
356:4
352:4
348:3
158:2
154:2
150:2
144:(
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.