269:
72 hours at room temperature, SAMs of alkanethiolates form within minutes. Special attention is essential in some cases, such as that of dithiol SAMs to avoid problems due to oxidation or photoinduced processes, which can affect terminal groups and lead to disorder and multilayer formation. In this case appropriate choice of solvents, their degassing by inert gasses and preparation in the absence of light is crucial and allows formation of "standing up" SAMs with free –SH groups. Self-assembled monolayers can also be adsorbed from the vapor phase. In some cases when obtaining an ordered assembly is difficult or when different density phases need to be obtained substitutional self-assembly is used. Here one first forms the SAM of a given type of molecules, which give rise to ordered assembly and then a second assembly phase is performed (e.g. by immersion into a different solution). This method has also been used to give information on relative binding strengths of SAMs with different head groups and more generally on self-assembly characteristics.
502:
across a large area the molecules support each other into forming their SAM shape seen in Figure 1. The orientation of the molecules can be described with two parameters: α and β. α is the angle of tilt of the backbone from the surface normal. In typical applications α varies from 0 to 60 degrees depending on the substrate and type of SAM molecule. β is the angle of rotation along the long axis of tee molecule. β is usually between 30 and 40 degrees. In some cases existence of kinetic traps hindering the final ordered orientation has been pointed out. Thus in case of dithiols formation of a "lying down" phase was considered an impediment to formation of "standing up" phase, however various recent studies indicate this is not the case.
724:(MEMS), and everyday household goods. SAMs can serve as models for studying membrane properties of cells and organelles and cell attachment on surfaces. SAMs can also be used to modify the surface properties of electrodes for electrochemistry, general electronics, and various NEMS and MEMS. For example, the properties of SAMs can be used to control electron transfer in electrochemistry. They can serve to protect metals from harsh chemicals and etchants. SAMs can also reduce sticking of NEMS and MEMS components in humid environments. In the same way, SAMs can alter the properties of glass. A common household product,
317:(STM). STM has been able to help understand the mechanisms of SAM formation as well as determine the important structural features that lend SAMs their integrity as surface-stable entities. In particular STM can image the shape, spatial distribution, terminal groups and their packing structure. AFM offers an equally powerful tool without the requirement of the SAM being conducting or semi-conducting. AFM has been used to determine chemical functionality, conductance, magnetic properties, surface charge, and frictional forces of SAMs. The
321:(SVET) is a further scanning probe microscopy which has been used to characterize SAMs, with defect free SAMs showing homogeneous activity in SVET. More recently, however, diffractive methods have also been used. The structure can be used to characterize the kinetics and defects found on the monolayer surface. These techniques have also shown physical differences between SAMs with planar substrates and nanoparticle substrates. An alternative characterisation instrument for measuring the self-assembly in real time is
35:
163:
68:
80:
243:(NEMS). Additionally, it can withstand harsh chemical cleaning treatments. Recently other chalcogenide SAMs: selenides and tellurides have attracted attention in a search for different bonding characteristics to substrates affecting the SAM characteristics and which could be of interest in some applications such as molecular electronics. Silanes are generally used on nonmetallic oxide surfaces; however monolayers formed from
562:
the SAMs. The transfer of the SAMs is a complex diffusion process that depends on the type of molecule, concentration, duration of contact, and pressure applied. Typical stamps use PDMS because its elastomeric properties, E = 1.8 MPa, allow it to fit the contour of micro surfaces and its low surface energy, γ = 21.6 dyn/cm². This is a parallel process and can thus place nanoscale objects over a large area in a short time.
175:
at higher molecular coverage, over a period of minutes to hours, begin to form three-dimensional crystalline or semicrystalline structures on the substrate surface. The "head groups" assemble together on the substrate, while the tail groups assemble far from the substrate. Areas of close-packed molecules nucleate and grow until the surface of the substrate is covered in a single monolayer.
849:
There has been considerable interest in use of SAMs for new materials e.g. via formation of two- or three-dimensional metal organic superlattices by assembly of SAM capped nanoparticles or layer by layer SAM-nanoparticle arrays using dithiols. A detailed review on this subject using dithiols is given
674:
The final strategy focuses not on the deposition or removal of SAMS, but the modification of terminal groups. In the first case the terminal group can be modified to remove functionality so that SAM molecule will be inert. In the same regards the terminal group can be modified to add functionality so
501:
overcome the surrounding force. The forces between the molecules orient them so they are in their straight, optimal, configuration. Then as other molecules come close by they interact with these already organized molecules in the same fashion and become a part of the conformed group. When this occurs
480:
The phase transitions in which a SAM forms depends on the temperature of the environment relative to the triple point temperature, the temperature in which the tip of the low-density phase intersects with the intermediate-phase region. At temperatures below the triple point the growth goes from phase
268:
or electroless deposition. Thiol or selenium SAMs produced by adsorption from solution are typically made by immersing a substrate into a dilute solution of alkane thiol in ethanol, though many different solvents can be used besides use of pure liquids. While SAMs are often allowed to form over 12 to
618:
can remove SAM molecules in many different ways. The first is to remove them mechanically by dragging the tip across the substrate surface. This is not the most desired technique as these tips are expensive and dragging them causes a lot of wear and reduction of the tip quality. The second way is to
523:
The final structure of the SAM is also dependent on the chain length and the structure of both the adsorbate and the substrate. Steric hindrance and metal substrate properties, for example, can affect the packing density of the film, while chain length affects SAM thickness. Longer chain length also
505:
Many of the SAM properties, such as thickness, are determined in the first few minutes. However, it may take hours for defects to be eliminated via annealing and for final SAM properties to be determined. The exact kinetics of SAM formation depends on the adsorbate, solvent and substrate properties.
496:
with some hydrogen bonding. The small size of the SAM molecules are important here because Van der Waals forces arise from the dipoles of molecules and are thus much weaker than the surrounding surface forces at larger scales. The assembly process begins with a small group of molecules, usually two,
381:
There is evidence that SAM formation occurs in two steps: an initial fast step of adsorption and a second slower step of monolayer organization. Adsorption occurs at the liquid–liquid, liquid–vapor, and liquid-solid interfaces. The transport of molecules to the surface occurs due to a combination of
581:
to transfer molecules on the tip to a substrate. Initially the tip is dipped into a reservoir with an ink. The ink on the tip evaporates and leaves the desired molecules attached to the tip. When the tip is brought into contact with the surface a water meniscus forms between the tip and the surface
221:
and interfacial properties. An appropriate substrate is chosen to react with the head group. Substrates can be planar surfaces, such as silicon and metals, or curved surfaces, such as nanoparticles. Alkanethiols are the most commonly used molecules for SAMs. Alkanethiols are molecules with an alkyl
174:
of "head groups" onto a substrate from either the vapor or liquid phase followed by a slow organization of "tail groups". Initially, at small molecular density on the surface, adsorbate molecules form either a disordered mass of molecules or form an ordered two-dimensional "lying down phase", and
561:
is analogous to printing ink with a rubber stamp. The SAM molecules are inked onto a pre-shaped elastomeric stamp with a solvent and transferred to the substrate surface by stamping. The SAM solution is applied to the entire stamp but only areas that make contact with the surface allow transfer of
484:
At temperatures above the triple point the growth is more complex and can take two paths. In the first path the heads of the SAM organize to their near final locations with the tail groups loosely formed on top. Then as they transit to phase 3, the tail groups become ordered and straighten out. In
351:
Defects due to both external and intrinsic factors may appear. External factors include the cleanliness of the substrate, method of preparation, and purity of the adsorbates. SAMs intrinsically form defects due to the thermodynamics of formation, e.g. thiol SAMs on gold typically exhibit etch pits
238:
SH)). Interest in such dithiols stems from the possibility of linking the two sulfur ends to metallic contacts, which was first used in molecular conduction measurements. Thiols are frequently used on noble metal substrates because of the strong affinity of sulfur for these metals. The sulfur gold
334:
measurements can be used to determine the surface free-energy which reflects the average composition of the surface of the SAM and can be used to probe the kinetics and thermodynamics of the formation of SAMs. The kinetics of adsorption and temperature induced desorption as well as information on
222:
chain, (C-C)ⁿ chain, as the back bone, a tail group, and a S-H head group. Other types of interesting molecules include aromatic thiols, of interest in molecular electronics, in which the alkane chain is (partly) replaced by aromatic rings. An example is the dithiol 1,4-Benzenedimethanethiol (SHCH
715:
SAMs are an inexpensive and versatile surface coating for applications including control of wetting and adhesion, chemical resistance, bio compatibility, sensitization, and molecular recognition for sensors and nano fabrication. Areas of application for SAMs include biology, electrochemistry and
352:(monatomic vacancy islands) likely due to extraction of adatoms from the substrate and formation of adatom-adsorbate moieties. Recently, a new type of fluorosurfactants have found that can form nearly perfect monolayer on gold substrate due to the increase of mobility of gold surface atoms.
664:
In this process, UV light is projected onto the surface with a SAM through a pattern of apertures in a chromium film. This leads to photo oxidation of the SAM molecules. These can then be washed away in a polar solvent. This process has 100 nm resolutions and requires exposure time of
251:
because they do not form reversibly. Self-assembled monolayers of thiolates on noble metals are a special case because the metal-metal bonds become reversible after the formation of the thiolate-metal complex. This reversibility is what gives rise to vacancy islands and it is why SAMs of
2298:
Laibinis, Paul E.; Whitesides, George M.; Allara, David L.; Tao, Yu Tai; Parikh, Atul N.; Nuzzo, Ralph G. (1 September 1991). "Comparison of the structures and wetting properties of self-assembled monolayers of n-alkanethiols on the coinage metal surfaces, copper, silver, and gold".
1777:
Hamoudi, H.; Guo Z., Prato M., Dablemont C., Zheng W.Q., Bourguignon B., Canepa M., Esaulov, V. A.; Prato, Mirko; Dablemont, Céline; Zheng, Wan Quan; Bourguignon, Bernard; Canepa, Maurizio; Esaulov, Vladimir A. (2008). "On the self assembly of short chain alkanedithiols".
2113:
Tang, Yongan; Yan, Jiawei; Zhu, Feng; Sun, Chunfeng; Mao, Bingwei (2011). "Comparative electrochemical scanning tunneling microscopy study of nonionic fluorosurfactant zonyl FSN self-assembled monolayers on Au(111) and Au(100) a potential-induced structural transition".
688:
Exposure to electron beams and UV light changes the terminal group chemistry. Some of the changes that can occur include the cleavage of bonds, the forming of double carbon bonds, cross-linking of adjacent molecules, fragmentation of molecules, and confromational
771:
is coated with a SAM that binds to the fungus. As the contaminated blood is filtered through a MEMS device the magnetic nanoparticles are inserted into the blood where they bind to the fungus and are then magnetically driven out of the blood stream into a nearby
195:
group on a substrate, and forms very stable, covalent bond with an energy of 452 kJ/mol. Thiol-metal bonds are on the order of 100 kJ/mol, making them fairly stable in a variety of temperatures, solvents, and potentials. The monolayer packs tightly due to
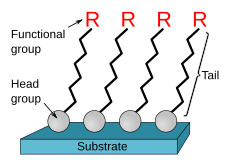
147:(HOPG). In other cases the molecules possess a head group that has a strong affinity to the substrate and anchors the molecule to it. Such a SAM consisting of a head group, tail and functional end group is depicted in Figure 1. Common head groups include
788:, because each adsorbate molecule can be tailored to attract two different materials. Current techniques utilize the head to attract to a surface, like a plate of gold. The terminal group is then modified to attract a specific material like a particular
582:
resulting in the diffusion of molecules from the tip to the surface. These tips can have radii in the tens of nanometers, and thus SAM molecules can be very precisely deposited onto a specific location of the surface. This process was discovered by
481:
1 to phase 2 where many islands form with the final SAM structure, but are surrounded by random molecules. Similar to nucleation in metals, as these islands grow larger they intersect forming boundaries until they end up in phase 3, as seen below.
178:
Adsorbate molecules adsorb readily because they lower the surface free-energy of the substrate and are stable due to the strong chemisorption of the "head groups." These bonds create monolayers that are more stable than the physisorbed bonds of
239:
interaction is semi-covalent and has a strength of approximately 45 kcal/mol. In addition, gold is an inert and biocompatible material that is easy to acquire. It is also easy to pattern via lithography, a useful feature for applications in
134:
and are organized into more or less large ordered domains. In some cases molecules that form the monolayer do not interact strongly with the substrate. This is the case for instance of the two-dimensional supramolecular networks of e.g.
216:
Selecting the type of head group depends on the application of the SAM. Typically, head groups are connected to a molecular chain in which the terminal end can be functionalized (i.e. adding –OH, –NH2, –COOH, or –SH groups) to vary the
804:, SWNTs. Dip pen nanolithography was used to pattern a 16-mercaptohexadecanoic acid (MHA)SAM and the rest of the surface was passivated with 1-octadecanethiol (ODT) SAM. The polar solvent that is carrying the SWNTs is attracted to the
2262:
Lud, S.Q; Neppl, S; Xu, F; Feulner, P; Stutzmann, M; Jordan, Rainer; Feulner, Peter; Stutzmann, Martin; Garrido, Jose A. (2010). "Controlling
Surface Functionality through Generation of Thiol Groups in a Self-Assembled Monolayer".
2077:
Yan, Jiawei; Tang, Yongan; Sun, Chunfeng; Su, Yuzhuan; Mao, Bingwei (2010). "STM Study on
Nonionic Fluorosurfactant Zonyl FSN Self-Assembly on Au(100) (3/1/-1/1) Molecular Lattice, Corrugations, and Adsorbate-Enhanced Mobility".
506:
In general, however, the kinetics are dependent on both preparations conditions and material properties of the solvent, adsorbate and substrate. Specifically, kinetics for adsorption from a liquid solution are dependent on:
836:
so that binding of these molecules can be detected. The ability to pattern these SAMs allows them to be placed in configurations that increase sensitivity and do not damage or interfere with other components of the
103:
545:
will later be located. This strategy is advantageous because it involves high throughput methods that generally involve fewer steps than the other two strategies. The major techniques that use this strategy are:
364:
and nanocrystals, "stabilize the reactive surface of the particle and present organic functional groups at the particle-solvent interface". These organic functional groups are useful for applications, such as
1398:
Andres, R.P.; Bein T.; Dorogi M.; Feng S.; Henderson J.I.; Kubiak C.P.; Mahoney W.; Osifchin R.G.; Reifenberger R. (1996). "Coulomb
Staircase at Room Temperature in a Self-Assembled Molecular Nanostructure".
446:
2041:
Tang, Yongan; Yan, Jiawei; Zhou, Xiaoshun; Fu, Yongchun; Mao, Bingwei (2008). "An STM study on nonionic fluorosurfactant zonyl FSN self-assembly on Au(111) large domains, few defects, and good stability".
816:, Schatz and their co-workers were able to make complex two-dimensional shapes, a representation of a shape created is shown to the right. Another application of patterned SAMs is the functionalization of
325:
where the refractive index, thickness, mass and birefringence of the self assembled layer are quantified at high resolution. Another method that can be used to measure the self-assembly in real-time is
2377:
Lud, S.Q; Steenackers, M; Bruno, P; Gruen, D.M; Feulner, P; Garrido, J.A; Stutzmann, M; Stutzmann, M (2006). "Chemical
Grafting of Biphenyl Self-Assembled Monolayers on Ultrananocrystalline Diamond".
1998:
Laffineur, F.; Auguste, D.; Plumier, F.; Pirlot, C.; Hevesi, L.; Delhalle, J.; Mekhalif, Z. (2004). "Comparison between CH3(CH2)15SH and CF3(CF2)3(CH2)11SH Monolayers on
Electrodeposited Silver".
1838:
Alarcon, L.S.; Chen L., Esaulov, V. A., Gayone J.E., Sanchez E., Grizzi O. (2010). "Thiol
Terminated 1,4-Benzenedimethanethiol Self-Assembled Monolayers on Au(111) and InP(110) from Vapor Phase".
2631:
Vijaya
Sarathy, K.; John Thomas P., Kulkarni G.U., Rao C.N.R. (1999). "Superlattices of Metal and Metal−Semiconductor Quantum Dots Obtained by Layer-by-Layer Deposition of Nanoparticle Arrays".
1879:
Chaudhari, V.; Harish N.M.K.; Sampath S.; Esaulov V.A. (2011). "Substitutional Self-Assembly of
Alkanethiol and Selenol SAMs from a Lying-Down Doubly Tethered Butanedithiol SAM on Gold".
1488:
Subramanian, S.; Sampath S. (2007). "Enhanced stability of short- and long-chain diselenide self-assembled monolayers on gold probed by electrochemistry, spectroscopy, and microscopy".
2334:
Noyhouzer, Tomer; Mandler, Daniel (17 January 2011). "Determination of low levels of cadmium ions by the under potential deposition on a self-assembled monolayer on gold electrode".
2752:
Faucheux, N.; Schweiss, R.; Lützow, K.; Werner, C.; Groth, T. (2004). "Self-assembled monolayers with different terminating groups as model substrates for cell adhesion studies".
2781:
Wasserman, S. R.; Tao, Y. T.; Whitesides, G. M. (1989). "Structure and
Reactivity of Alkylsiloxane Monolayers Formed by Reaction of Alkyltrichlorosilanes on Silicon Substrates".
382:
diffusion and convective transport. According to the
Langmuir or Avrami kinetic model the rate of deposition onto the surface is proportional to the free space of the surface.
1531:
Bucher, Jean-Pierre; Santesson, Lars, Kern, Klaus (31 March 1994). "Thermal Healing of Self-Assembled Organic Monolayers: Hexane- and Octadecanethiol on Au(111) and Ag(111)".
603:
is not desired. The result is the same as in the locally attract strategy, the difference being in the way this is achieved. The major techniques that use this strategy are:
485:
the second path the molecules start in a lying down position along the surface. These then form into islands of ordered SAMs, where they grow into phase 3, as seen below.
110:
1906:
Prato, M.; Moroni R.; Bisio F.; Rolandi R.; Mattera L.; Cavalleri O.; Canepa M. (2008). "Optical Characterization of Thiolate Self-Assembled Monolayers on Au(111)".
1721:
Hamoudi, H.; Prato M., Dablemont C., Cavalleri O., Canepa M., Esaulov, V. A. (2010). "Self-Assembly of 1,4-Benzenedimethanethiol Self-Assembled Monolayers on Gold".
2834:
599:
The locally remove strategy begins with covering the entire surface with a SAM. Then individual SAM molecules are removed from locations where the deposition of
327:
1617:
981:
Elemans, J.A.A.W.; Lei S., De Feyter S. (2009). "Molecular and Supramolecular Networks on Surfaces: From Two-Dimensional Crystal Engineering to Reactivity".
1450:
Shaporenko, A.; Muller J.; Weidner T.; Terfort A.; Zharnikov M. (2007). "Balance of Structure-Building Forces in Selenium-Based Self-Assembled Monolayers".
1167:
Schwartz, D.K., Mechanisms and Kinetics of Self-Assembled Monolayer Formation (2001). "Mechanisms and kinetics of self-assembled monolayer formation".
675:
it can accept different materials or have different properties than the original SAM terminal group. The major techniques that use this strategy are:
459:
is the rate constant. Although this model is robust it is only used for approximations because it fails to take into account intermediate processes.
728:, utilizes SAMs to create a hydrophobic monolayer on car windshields to keep them clear of rain. Another application is an anti-adhesion coating on
2658:
2617:
1865:
1821:
1756:
1602:
1558:
1331:
1283:
1210:
488:
The nature in which the tail groups organize themselves into a straight ordered monolayer is dependent on the inter-molecular attraction, or
2806:; Behm, R.J. (2007). "Structure Formation in Bis(terpyridine)Derivative Adlayers – Molecule-Substrate vs. Molecule-Molecule Interactions".
2574:
Kiely, C.J.; Fink J., Brust M., Bethell D? Schiffrin D.J. (1999). "Spontaneous ordering of bimodal ensembles of nanoscopic gold clusters".
513:
Concentration of adsorbate in the solution – low concentrations require longer immersion times and often create highly crystalline domains.
1776:
492:, between the tail groups. To minimize the free energy of the organic layer the molecules adopt conformations that allow high degree of
96:
388:
1061:
De Feyter, S.; De Schreyer F.C. (2003). "Two-dimensional supramolecular self-assembly probed by scanning tunneling microscopy".
318:
1700:
1676:
Bain, Colin D.; Troughton, E. Barry; Tao, Yu Tai; Evall, Joseph; Whitesides, George M.; Nuzzo, Ralph G. (31 December 1988).
285:, which also give information on interfacial properties. The order in the SAM and orientation of molecules can be probed by
759:
or other MEMS devices that need to separate one type of molecule from its environment. One example is the use of magnetic
293:
in Reflection Absorption Infrared Spectroscopy (RAIRS) studies. Numerous other spectroscopic techniques are used such as
136:
1026:
Witte, G.; Wöll Ch. (2004). "Growth of aromatic molecules on solid substrates for applications in organic electronics".
1575:
Schlenoff, Joseph B.; Li, Ming, Ly, Hiep (30 November 1995). "Stability and Self-Exchange in Alkanethiol Monolayers".
463:
being a real time technique with ~10 Hz resolution can measure the kinetics of monolayer self-assembly directly.
2859:
302:
2447:
Yung; Fiering, J; Mueller, AJ; Ingber, DE; et al. (2009). "Micromagnetic–microfluidic blood cleansing device".
946:
Barlow, S.M.; Raval R.. (2003). "Complex organic molecules at metal surfaces: bonding, organisation and chirality".
1933:
Kato, H.; Noh J.; Hara M.; Kawai M. (2002). "An HREELS Study of Alkanethiol Self-Assembled Monolayers on Au(111)".
1646:
Nuzzo, Ralph G.; Allara, David L. (31 May 1983). "Adsorption of bifunctional organic disulfides on gold surfaces".
1624:
460:
322:
282:
2724:
Sagiv, J.; Polymeropoulos, E.E. (1978). "Adsorbed monolayers - molecular-organization and electrical-properties".
648:
473:
2. An intermediate-density phase with conformational disordered molecules or molecules lying flat on the surface.
721:
204:
if lateral interactions are neglected. If they cannot be neglected, the adsorption is better described by the
1166:
717:
620:
615:
607:
314:
240:
639:
The most common use of this technique is to remove the SAM molecules in a process called shaving, where the
360:
The structure of SAMs is also dependent on the curvature of the substrate. SAMs on nanoparticles, including
737:
201:
911:
Love; et al. (2005). "Self-Assembled Monolayers of Thiolates on Metals as a Form of Nanotechnology".
476:
3. A high-density phase with close-packed order and molecules standing normal to the substrate's surface.
2864:
2412:
Cech J; Taboryski R (2012). "Stability of FDTS monolayer coating on aluminum injection molding tools".
294:
265:
197:
180:
2529:
Garcia, R.; Martinez, R.V; Martinez, J (2005). "Nano Chemistry and Scanning Probe Nanolithographies".
1720:
538:
340:
336:
309:. The structures of SAMs are commonly determined using scanning probe microscopy techniques such as
261:
2543:
1678:"Formation of monolayer films by the spontaneous assembly of organic thiols from solution onto gold"
1075:
298:
1837:
2849:
808:
MHA; as the solvent evaporates, the SWNTs are close enough to the MHA SAM to attach to it due to
729:
694:
644:
640:
632:
587:
578:
574:
567:
310:
49:
26:
2538:
1107:
1070:
701:
A conductive AFM tip can create an electrochemical reaction that can change the terminal group.
2744:
I. Rubinstein, E. Sabatani, R. Maoz and J. Sagiv, Organized Monolayers on Gold Electrodes, in
2482:
Carroll, Gregory T.; Wang, Denong; Turro, Nicholas J.; Koberstein, Jeffrey T. (1 March 2006).
800:
attached to the tail groups. One example is the use of two types of SAMs to align single wall
2652:
2611:
1859:
1815:
1750:
1596:
1552:
1204:
784:
Photolithographic methods are useful in patterning SAMs. SAMs are also useful in depositing
2803:
2685:
2583:
2421:
2343:
2175:
1972:
1787:
1497:
1408:
1242:
1176:
1035:
955:
809:
498:
493:
489:
370:
2802:
Hoster, H.E.; Roos, M.; Breitruck, A.; Meier, C.; Tonigold, K.; Waldmann, T.; Ziener, U.;
1106:
Carroll, Gregory T.; Pollard, Michael M.; van Delden, Richard A.; Feringa, Ben L. (2010).
755:
can now selectively attach itself to other molecules or SAMs. This technique is useful in
8:
2854:
2689:
2587:
2425:
2347:
2179:
1976:
1791:
1501:
1412:
1246:
1180:
1039:
959:
516:
Purity of the adsorbate – impurities can affect the final physical properties of the SAM
2765:
2703:
2630:
2599:
2244:
2196:
2163:
1432:
1325:
1277:
1233:
Schreiber, F (30 November 2000). "Structure and growth of self-assembling monolayers".
1130:
54:
1254:
1188:
967:
2823:
2769:
2707:
2556:
2511:
2503:
2464:
2394:
2359:
2316:
2280:
2236:
2201:
2131:
2095:
2059:
2023:
2015:
1803:
1738:
1513:
1467:
1424:
1192:
1088:
1008:
928:
733:
205:
84:
2248:
1436:
1134:
1108:"Controlled rotary motion of light-driven molecular motors assembled on a gold film"
466:
Once the molecules are at the surface the self-organization occurs in three phases:
130:) of organic molecules are molecular assemblies formed spontaneously on surfaces by
2815:
2790:
2761:
2733:
2693:
2640:
2603:
2591:
2548:
2495:
2456:
2429:
2386:
2351:
2308:
2272:
2228:
2219:
Piner, R.D; Zhu, J; Xu, F; Hong, S; Mirkin, C.A (1999). "Dip-Pen Nanolithography".
2191:
2183:
2123:
2087:
2051:
2007:
1984:
1980:
1942:
1915:
1888:
1847:
1795:
1730:
1692:
1655:
1584:
1540:
1505:
1459:
1416:
1250:
1184:
1122:
1080:
1043:
998:
990:
963:
920:
801:
624:
558:
550:
519:
Dirt or contamination on the substrate – imperfections can cause defects in the SAM
1420:
1385:
Self-assembled monolayers (SAMs). Fundamentals of BioMEMS and Medical Microdevices
2573:
2433:
2232:
1315:
510:
Temperature – room-temperature preparation improves kinetics and reduces defects.
335:
structure can also be obtained in real time by ion scattering techniques such as
252:
alkanethiolates can be thermally desorbed and undergo exchange with free thiols.
184:
619:
degrade or desorb the SAM molecules by shooting them with an electron beam. The
1677:
1675:
1509:
821:
797:
744:
600:
244:
200:, thereby reducing its own free energy. The adsorption can be described by the
72:
34:
2674:"Selfassembly of α, ω-dithiols on surfaces and metal dithiol heterostructures"
2355:
2187:
2843:
2737:
2507:
2483:
2320:
2019:
812:. The nanotubes thus line up with the MHA-ODT boundary. Using this technique
793:
785:
760:
752:
748:
542:
331:
248:
171:
1878:
2827:
2773:
2698:
2673:
2560:
2515:
2468:
2398:
2363:
2284:
2205:
2135:
2099:
2063:
2027:
1807:
1742:
1618:"Self-Assembled Monolayers (SAMs) as Collision Surfaces for Ion Activation"
1517:
1471:
1196:
1092:
1012:
994:
932:
789:
773:
768:
278:
162:
156:
2240:
1428:
1047:
470:
1. A low-density phase with random dispersion of molecules on the surface.
330:
where the mass and viscoelastic properties of the adlayer are quantified.
813:
805:
680:
656:
643:
tip is dragged along the surface mechanically removing the molecules. An
583:
366:
2794:
2376:
2312:
2011:
1696:
1659:
1588:
1544:
796:. In this way, wherever the SAM is patterned to a surface there will be
1449:
1126:
817:
756:
140:
131:
2819:
2644:
2499:
2390:
2276:
2127:
2091:
2055:
1946:
1919:
1892:
1851:
1734:
1463:
1003:
924:
67:
2552:
2460:
1799:
1084:
838:
833:
829:
1963:
Smith; et al. (2004). "Patterning Self-Assembled Monolayers".
825:
361:
192:
2595:
2528:
2164:"Strategies for Controlled Placement of Nanoscale Building Blocks"
1530:
328:
Quartz Crystal Microbalance with Dissipation monitoring technology
79:
218:
152:
1905:
1350:
Fundamentals of Microfabrication: The Science of Miniaturization
2261:
1105:
820:. The tail groups can be modified so they have an affinity for
764:
725:
441:{\displaystyle \mathbf {k(1-\theta )} ={\frac {d\theta }{dt}}.}
306:
286:
148:
2484:"Photochemical Micropatterning of Carbohydrates on a Surface"
1997:
1397:
373:, that are dependent on chemical composition of the surface.
2751:
2218:
2297:
1574:
980:
290:
188:
144:
2481:
1316:
Vos, Johannes G., Robert J. Forster, Tia E. Keyes (2003).
1267:
307:
High-resolution electron energy loss spectroscopy (HREELS)
247:
between silicon and carbon or oxygen cannot be considered
2835:
Sigma-Aldrich "Material Matters", Molecular Self-Assembly
2748:, C.K.N. Li (Ed.), The Electrochemical Society 1986: 175.
1932:
1060:
260:
Metal substrates for use in SAMs can be produced through
2801:
2726:
Berichte der Bunsengesellschaft für Physikalische Chemie
2446:
1270:
Encyclopedia of Biomaterials and Biomedical Engineering
910:
2780:
1645:
1487:
391:
2746:
Electrochemical Sensors for Biomedical Applications
2723:
1483:
1481:
1382:
440:
341:time of flight direct recoil spectroscopy (TOFDRS)
2411:
455:is the proportional amount of area deposited and
408:
396:
287:Near Edge Xray Absorption Fine Structure (NEXAFS)
2841:
2333:
537:This first strategy involves locally depositing
1962:
1478:
945:
2672:Hamoudi, Hicham; Esaulov, Vladimir A. (2016).
2671:
2161:
1833:
1831:
1772:
1770:
1768:
1766:
1716:
1714:
1712:
1710:
1443:
844:
277:The thicknesses of SAMs can be measured using
2112:
2040:
1872:
1370:Supramolecular Electrochemistry. Coral Gables
1367:
1025:
104:
2657:: CS1 maint: multiple names: authors list (
2616:: CS1 maint: multiple names: authors list (
2076:
1864:: CS1 maint: multiple names: authors list (
1820:: CS1 maint: multiple names: authors list (
1755:: CS1 maint: multiple names: authors list (
1601:: CS1 maint: multiple names: authors list (
1570:
1568:
1557:: CS1 maint: multiple names: authors list (
1330:: CS1 maint: multiple names: authors list (
1282:: CS1 maint: multiple names: authors list (
1209:: CS1 maint: multiple names: authors list (
2665:
2624:
1828:
1763:
1707:
1347:
1228:
1226:
1224:
1222:
1220:
166:Figure 1. Representation of a SAM structure
1391:
1162:
1160:
1158:
1156:
1154:
1152:
1150:
1148:
1146:
1144:
732:(NIL) tools and stamps. One can also coat
355:
111:
97:
2697:
2567:
2542:
2195:
1899:
1565:
1363:
1361:
1359:
1272:. Informa Healthcare. pp. 1331–1333.
1232:
1074:
1054:
1002:
974:
2405:
2379:Journal of the American Chemical Society
2301:Journal of the American Chemical Society
2157:
2155:
2153:
2151:
2149:
2147:
2145:
1958:
1956:
1926:
1685:Journal of the American Chemical Society
1648:Journal of the American Chemical Society
1641:
1639:
1637:
1577:Journal of the American Chemical Society
1490:Journal of Colloid and Interface Science
1452:Journal of the American Chemical Society
1343:
1341:
1217:
1019:
939:
669:
161:
1703:from the original on 23 September 2017.
1671:
1669:
1609:
1141:
906:
904:
902:
900:
898:
896:
894:
892:
890:
888:
886:
884:
882:
524:increases the thermodynamic stability.
291:Fourier Transform Infrared Spectroscopy
2842:
1356:
1311:
1309:
1307:
1305:
1303:
1301:
1299:
1297:
1295:
1293:
1099:
880:
878:
876:
874:
872:
870:
868:
866:
864:
862:
319:scanning vibrating electrode technique
283:X-ray photoelectron spectroscopy (XPS)
2142:
1953:
1634:
1338:
1318:Interfacial Supramolecular Assemblies
747:. In this way they functionalize the
743:Thin film SAMs can also be placed on
736:tools for polymer replication with a
627:and field enhanced surface diffusion.
532:
187:based "head group", for example in a
1666:
832:. The SAM can then be placed onto a
594:
1780:Physical Chemistry Chemical Physics
1615:
1290:
1268:Wnek, Gary, Gary L. Bowlin (2004).
1261:
859:
751:. This is advantageous because the
272:
137:perylenetetracarboxylic dianhydride
13:
2766:10.1016/j.biomaterials.2003.09.069
2717:
145:highly oriented pyrolitic graphite
14:
2876:
1189:10.1146/annurev.physchem.52.1.107
779:
710:
647:can also remove SAM molecules by
303:Surface-enhanced Raman scattering
461:Dual polarisation interferometry
402:
399:
393:
337:low energy ion scattering (LEIS)
323:dual polarisation interferometry
78:
66:
33:
2522:
2475:
2440:
2370:
2327:
2291:
2255:
2212:
2106:
2070:
2034:
1991:
1935:Journal of Physical Chemistry C
1908:Journal of Physical Chemistry C
1881:Journal of Physical Chemistry C
1840:Journal of Physical Chemistry C
1524:
1376:
705:
649:local oxidation nanolithography
21:Part of a series of articles on
1985:10.1016/j.progsurf.2003.12.001
1372:. Wiley VCH. pp. 191–193.
722:microelectromechanical systems
541:on the surface only where the
497:getting close enough that the
255:
1:
2633:Journal of Physical Chemistry
1421:10.1126/science.272.5266.1323
1387:. SPIE Press. pp. 94–96.
1255:10.1016/S0079-6816(00)00024-1
1028:Journal of Materials Research
968:10.1016/S0167-5729(03)00015-3
853:
718:nanoelectromechanical systems
621:scanning tunneling microscope
616:scanning tunneling microscope
608:Scanning tunneling microscope
527:
315:scanning tunneling microscopy
241:nanoelectromechanical systems
2434:10.1016/j.apsusc.2012.07.078
2233:10.1126/science.283.5402.661
738:Perfluordecyltrichlorosilane
202:Langmuir adsorption isotherm
7:
1965:Progress in Surface Science
1383:Saliterman, Steven (2006).
1235:Progress in Surface Science
845:Metal organic superlattices
376:
139:(PTCDA) on gold or of e.g.
10:
2881:
1510:10.1016/j.jcis.2007.03.021
679:Focused electron beam and
577:is a process that uses an
557:Micro-contact printing or
346:
295:Second-harmonic generation
198:van der Waals interactions
2356:10.1016/j.aca.2010.10.021
2188:10.1007/s11671-007-9091-3
792:, wire, ribbon, or other
767:from a blood stream. The
539:self-assembled monolayers
262:physical vapor deposition
124:Self-assembled monolayers
2860:Supramolecular chemistry
2738:10.1002/bbpc.19780820917
2531:Chemical Society Reviews
1320:. Wiley. pp. 88–94.
1063:Chemical Society Reviews
623:can also remove SAMs by
299:Sum-frequency generation
211:
191:molecule, reacts with a
170:SAMs are created by the
45:Self-assembled monolayer
2414:Applied Surface Science
2162:Seong, Jin Koh (2007).
948:Surface Science Reports
850:by Hamoudi and Esaulov
730:nanoimprint lithography
695:Atomic force microscope
645:atomic force microscope
641:atomic force microscope
633:Atomic force microscope
588:Northwestern University
579:atomic force microscope
575:Dip-pen nanolithography
568:Dip-pen nanolithography
356:Nanoparticle properties
311:atomic force microscopy
181:Langmuir–Blodgett films
50:Supramolecular assembly
27:Molecular self-assembly
2699:10.1002/andp.201500280
2336:Analytica Chimica Acta
1368:Kaifer, Angel (2001).
1352:. CRC. pp. 62–63.
995:10.1002/anie.200806339
551:Micro-contact printing
442:
167:
1169:Annu. Rev. Phys. Chem
1048:10.1557/JMR.2004.0251
670:3. Modify tail groups
443:
165:
85:Technology portal
1348:Madou, Marc (2002).
983:Angew. Chem. Int. Ed
810:Van der Waals forces
499:Van der Waals forces
494:Van der Waals forces
490:van der Waals forces
389:
2814:(23): 11570–11579.
2795:10.1021/la00088a035
2690:2016AnP...528..242H
2588:1998Natur.396..444K
2426:2012ApSS..259..538C
2385:(51): 16884–16891.
2348:2011AcAC..684....1N
2313:10.1021/ja00019a011
2180:2007NRL.....2..519K
2050:(23): 13245–13249.
1977:2004PrSS...75....1S
1887:(33): 16518–16523.
1846:(47): 19993–19999.
1792:2008PCCP...10.6836H
1697:10.1021/ja00183a049
1660:10.1021/ja00351a063
1589:10.1021/ja00155a016
1583:(50): 12528–12536.
1545:10.1021/la00016a001
1502:2007JCIS..312..413S
1413:1996Sci...272.1323A
1407:(5266): 1323–1325.
1247:2000PrSS...65..151S
1181:2001ARPC...52..107S
1040:2004JMatR..19.1889W
960:2003SurSR..50..201B
665:15–20 minutes.
305:(SERS), as well as
2678:Annalen der Physik
2168:Nanoscale Res Lett
1127:10.1039/C0SC00162G
586:and co-workers at
533:1. Locally attract
438:
168:
73:Science portal
55:DNA nanotechnology
2865:Self-organization
2820:10.1021/la701382n
2760:(14): 2721–2730.
2645:10.1021/jp983836l
2500:10.1021/la0531042
2391:10.1021/ja0657049
2307:(19): 7152–7167.
2277:10.1021/la102225r
2271:(20): 15895–900.
2227:(5402): 661–663.
2128:10.1021/la103812v
2092:10.1021/la903250m
2056:10.1021/la802682n
2012:10.1021/la035851+
1947:10.1021/jp020968c
1941:(37): 9655–9658.
1920:10.1021/jp711194s
1914:(10): 3899–3906.
1893:10.1021/jp2042922
1852:10.1021/jp1044157
1786:(45): 6836–6841.
1735:10.1021/la904317b
1729:(10): 7242–7247.
1654:(13): 4481–4483.
1464:10.1021/ja068916e
989:(40): 7298–7332.
925:10.1021/cr0300789
734:injection molding
595:2. Locally remove
433:
266:electrodeposition
121:
120:
2872:
2831:
2798:
2789:(4): 1074–1087.
2777:
2741:
2712:
2711:
2701:
2684:(3–4): 242–263.
2669:
2663:
2662:
2656:
2648:
2628:
2622:
2621:
2615:
2607:
2571:
2565:
2564:
2553:10.1039/b501599p
2546:
2526:
2520:
2519:
2494:(6): 2899–2905.
2479:
2473:
2472:
2461:10.1039/b816986a
2455:(9): 1171–1177.
2444:
2438:
2437:
2409:
2403:
2402:
2374:
2368:
2367:
2331:
2325:
2324:
2295:
2289:
2288:
2259:
2253:
2252:
2216:
2210:
2209:
2199:
2159:
2140:
2139:
2110:
2104:
2103:
2086:(6): 3829–3834.
2074:
2068:
2067:
2038:
2032:
2031:
2006:(8): 3240–3245.
1995:
1989:
1988:
1960:
1951:
1950:
1930:
1924:
1923:
1903:
1897:
1896:
1876:
1870:
1869:
1863:
1855:
1835:
1826:
1825:
1819:
1811:
1800:10.1039/B809760G
1774:
1761:
1760:
1754:
1746:
1718:
1705:
1704:
1682:
1673:
1664:
1663:
1643:
1632:
1631:
1630:on 22 June 2010.
1629:
1623:. Archived from
1622:
1613:
1607:
1606:
1600:
1592:
1572:
1563:
1562:
1556:
1548:
1528:
1522:
1521:
1485:
1476:
1475:
1458:(8): 2232–2233.
1447:
1441:
1440:
1395:
1389:
1388:
1380:
1374:
1373:
1365:
1354:
1353:
1345:
1336:
1335:
1329:
1321:
1313:
1288:
1287:
1281:
1273:
1265:
1259:
1258:
1241:(5–8): 151–257.
1230:
1215:
1214:
1208:
1200:
1164:
1139:
1138:
1112:
1103:
1097:
1096:
1085:10.1039/b206566p
1078:
1058:
1052:
1051:
1034:(7): 1889–1916.
1023:
1017:
1016:
1006:
978:
972:
971:
954:(6–8): 201–341.
943:
937:
936:
919:(4): 1103–1170.
908:
802:carbon nanotubes
625:field desorption
559:soft lithography
447:
445:
444:
439:
434:
432:
424:
416:
411:
273:Characterization
113:
106:
99:
83:
82:
71:
70:
37:
18:
17:
2880:
2879:
2875:
2874:
2873:
2871:
2870:
2869:
2840:
2839:
2720:
2718:Further reading
2715:
2670:
2666:
2650:
2649:
2629:
2625:
2609:
2608:
2572:
2568:
2544:10.1.1.454.2979
2527:
2523:
2480:
2476:
2445:
2441:
2410:
2406:
2375:
2371:
2332:
2328:
2296:
2292:
2260:
2256:
2217:
2213:
2174:(11): 519–545.
2160:
2143:
2111:
2107:
2075:
2071:
2039:
2035:
1996:
1992:
1961:
1954:
1931:
1927:
1904:
1900:
1877:
1873:
1857:
1856:
1836:
1829:
1813:
1812:
1775:
1764:
1748:
1747:
1719:
1708:
1680:
1674:
1667:
1644:
1635:
1627:
1620:
1614:
1610:
1594:
1593:
1573:
1566:
1550:
1549:
1529:
1525:
1486:
1479:
1448:
1444:
1396:
1392:
1381:
1377:
1366:
1357:
1346:
1339:
1323:
1322:
1314:
1291:
1275:
1274:
1266:
1262:
1231:
1218:
1202:
1201:
1165:
1142:
1110:
1104:
1100:
1076:10.1.1.467.5727
1059:
1055:
1024:
1020:
979:
975:
944:
940:
909:
860:
856:
847:
782:
713:
708:
672:
597:
535:
530:
425:
417:
415:
392:
390:
387:
386:
379:
358:
349:
275:
258:
237:
233:
229:
225:
214:
185:trichlorosilane
117:
77:
65:
12:
11:
5:
2878:
2868:
2867:
2862:
2857:
2852:
2850:Nanotechnology
2838:
2837:
2832:
2804:Landfester, K.
2799:
2778:
2749:
2742:
2719:
2716:
2714:
2713:
2664:
2639:(3): 399–401.
2623:
2582:(3): 444–446.
2566:
2521:
2474:
2439:
2404:
2369:
2326:
2290:
2254:
2211:
2141:
2122:(3): 943–947.
2105:
2069:
2033:
1990:
1952:
1925:
1898:
1871:
1827:
1762:
1706:
1691:(1): 321–335.
1665:
1633:
1608:
1564:
1539:(4): 979–983.
1523:
1496:(2): 413–424.
1477:
1442:
1390:
1375:
1355:
1337:
1289:
1260:
1216:
1140:
1098:
1069:(3): 139–150.
1053:
1018:
973:
938:
857:
855:
852:
846:
843:
798:nanostructures
786:nanostructures
781:
780:Patterned SAMs
778:
776:waste stream.
745:nanostructures
712:
711:Thin-film SAMs
709:
707:
704:
703:
702:
698:
697:
691:
690:
685:
684:
671:
668:
667:
666:
661:
660:
653:
652:
636:
635:
629:
628:
611:
610:
601:nanostructures
596:
593:
592:
591:
571:
570:
564:
563:
554:
553:
534:
531:
529:
526:
521:
520:
517:
514:
511:
478:
477:
474:
471:
449:
448:
437:
431:
428:
423:
420:
414:
410:
407:
404:
401:
398:
395:
378:
375:
357:
354:
348:
345:
274:
271:
257:
254:
249:self assembled
245:covalent bonds
235:
231:
227:
223:
213:
210:
119:
118:
116:
115:
108:
101:
93:
90:
89:
88:
87:
75:
60:
59:
58:
57:
52:
47:
39:
38:
30:
29:
23:
22:
9:
6:
4:
3:
2:
2877:
2866:
2863:
2861:
2858:
2856:
2853:
2851:
2848:
2847:
2845:
2836:
2833:
2829:
2825:
2821:
2817:
2813:
2809:
2805:
2800:
2796:
2792:
2788:
2784:
2779:
2775:
2771:
2767:
2763:
2759:
2755:
2750:
2747:
2743:
2739:
2735:
2731:
2727:
2722:
2721:
2709:
2705:
2700:
2695:
2691:
2687:
2683:
2679:
2675:
2668:
2660:
2654:
2646:
2642:
2638:
2634:
2627:
2619:
2613:
2605:
2601:
2597:
2596:10.1038/24808
2593:
2589:
2585:
2581:
2577:
2570:
2562:
2558:
2554:
2550:
2545:
2540:
2536:
2532:
2525:
2517:
2513:
2509:
2505:
2501:
2497:
2493:
2489:
2485:
2478:
2470:
2466:
2462:
2458:
2454:
2450:
2449:Lab on a Chip
2443:
2435:
2431:
2427:
2423:
2419:
2415:
2408:
2400:
2396:
2392:
2388:
2384:
2380:
2373:
2365:
2361:
2357:
2353:
2349:
2345:
2341:
2337:
2330:
2322:
2318:
2314:
2310:
2306:
2302:
2294:
2286:
2282:
2278:
2274:
2270:
2266:
2258:
2250:
2246:
2242:
2238:
2234:
2230:
2226:
2222:
2215:
2207:
2203:
2198:
2193:
2189:
2185:
2181:
2177:
2173:
2169:
2165:
2158:
2156:
2154:
2152:
2150:
2148:
2146:
2137:
2133:
2129:
2125:
2121:
2117:
2109:
2101:
2097:
2093:
2089:
2085:
2081:
2073:
2065:
2061:
2057:
2053:
2049:
2045:
2037:
2029:
2025:
2021:
2017:
2013:
2009:
2005:
2001:
1994:
1986:
1982:
1978:
1974:
1971:(1–2): 1–68.
1970:
1966:
1959:
1957:
1948:
1944:
1940:
1936:
1929:
1921:
1917:
1913:
1909:
1902:
1894:
1890:
1886:
1882:
1875:
1867:
1861:
1853:
1849:
1845:
1841:
1834:
1832:
1823:
1817:
1809:
1805:
1801:
1797:
1793:
1789:
1785:
1781:
1773:
1771:
1769:
1767:
1758:
1752:
1744:
1740:
1736:
1732:
1728:
1724:
1717:
1715:
1713:
1711:
1702:
1698:
1694:
1690:
1686:
1679:
1672:
1670:
1661:
1657:
1653:
1649:
1642:
1640:
1638:
1626:
1619:
1612:
1604:
1598:
1590:
1586:
1582:
1578:
1571:
1569:
1560:
1554:
1546:
1542:
1538:
1534:
1527:
1519:
1515:
1511:
1507:
1503:
1499:
1495:
1491:
1484:
1482:
1473:
1469:
1465:
1461:
1457:
1453:
1446:
1438:
1434:
1430:
1426:
1422:
1418:
1414:
1410:
1406:
1402:
1394:
1386:
1379:
1371:
1364:
1362:
1360:
1351:
1344:
1342:
1333:
1327:
1319:
1312:
1310:
1308:
1306:
1304:
1302:
1300:
1298:
1296:
1294:
1285:
1279:
1271:
1264:
1256:
1252:
1248:
1244:
1240:
1236:
1229:
1227:
1225:
1223:
1221:
1212:
1206:
1198:
1194:
1190:
1186:
1182:
1178:
1174:
1170:
1163:
1161:
1159:
1157:
1155:
1153:
1151:
1149:
1147:
1145:
1136:
1132:
1128:
1124:
1121:(1): 97–101.
1120:
1116:
1109:
1102:
1094:
1090:
1086:
1082:
1077:
1072:
1068:
1064:
1057:
1049:
1045:
1041:
1037:
1033:
1029:
1022:
1014:
1010:
1005:
1000:
996:
992:
988:
984:
977:
969:
965:
961:
957:
953:
949:
942:
934:
930:
926:
922:
918:
914:
907:
905:
903:
901:
899:
897:
895:
893:
891:
889:
887:
885:
883:
881:
879:
877:
875:
873:
871:
869:
867:
865:
863:
858:
851:
842:
840:
835:
831:
827:
823:
819:
815:
811:
807:
803:
799:
795:
794:nanostructure
791:
787:
777:
775:
770:
766:
762:
761:nanoparticles
758:
754:
753:nanostructure
750:
749:nanostructure
746:
741:
739:
735:
731:
727:
723:
719:
716:electronics,
700:
699:
696:
693:
692:
687:
686:
682:
678:
677:
676:
663:
662:
658:
655:
654:
650:
646:
642:
638:
637:
634:
631:
630:
626:
622:
617:
613:
612:
609:
606:
605:
604:
602:
589:
585:
580:
576:
573:
572:
569:
566:
565:
560:
556:
555:
552:
549:
548:
547:
544:
543:nanostructure
540:
525:
518:
515:
512:
509:
508:
507:
503:
500:
495:
491:
486:
482:
475:
472:
469:
468:
467:
464:
462:
458:
454:
435:
429:
426:
421:
418:
412:
405:
385:
384:
383:
374:
372:
368:
363:
353:
344:
342:
338:
333:
332:Contact angle
329:
324:
320:
316:
312:
308:
304:
300:
296:
292:
288:
284:
280:
270:
267:
263:
253:
250:
246:
242:
220:
209:
207:
203:
199:
194:
190:
186:
182:
176:
173:
172:chemisorption
164:
160:
158:
154:
150:
146:
142:
138:
133:
129:
125:
114:
109:
107:
102:
100:
95:
94:
92:
91:
86:
81:
76:
74:
69:
64:
63:
62:
61:
56:
53:
51:
48:
46:
43:
42:
41:
40:
36:
32:
31:
28:
25:
24:
20:
19:
16:
2811:
2807:
2786:
2782:
2757:
2754:Biomaterials
2753:
2745:
2729:
2725:
2681:
2677:
2667:
2653:cite journal
2636:
2632:
2626:
2612:cite journal
2579:
2575:
2569:
2537:(1): 29–38.
2534:
2530:
2524:
2491:
2487:
2477:
2452:
2448:
2442:
2417:
2413:
2407:
2382:
2378:
2372:
2342:(1–2): 1–7.
2339:
2335:
2329:
2304:
2300:
2293:
2268:
2264:
2257:
2224:
2220:
2214:
2171:
2167:
2119:
2115:
2108:
2083:
2079:
2072:
2047:
2043:
2036:
2003:
1999:
1993:
1968:
1964:
1938:
1934:
1928:
1911:
1907:
1901:
1884:
1880:
1874:
1860:cite journal
1843:
1839:
1816:cite journal
1783:
1779:
1751:cite journal
1726:
1722:
1688:
1684:
1651:
1647:
1625:the original
1611:
1597:cite journal
1580:
1576:
1553:cite journal
1536:
1532:
1526:
1493:
1489:
1455:
1451:
1445:
1404:
1400:
1393:
1384:
1378:
1369:
1349:
1317:
1269:
1263:
1238:
1234:
1205:cite journal
1172:
1168:
1118:
1114:
1101:
1066:
1062:
1056:
1031:
1027:
1021:
986:
982:
976:
951:
947:
941:
916:
912:
848:
790:nanoparticle
783:
769:nanoparticle
763:to remove a
742:
714:
706:Applications
673:
598:
536:
522:
504:
487:
483:
479:
465:
456:
452:
450:
380:
367:immunoassays
359:
350:
279:ellipsometry
276:
264:techniques,
259:
215:
177:
169:
157:phosphonates
127:
123:
122:
44:
15:
2420:: 538–541.
814:Chad Mirkin
806:hydrophilic
720:(NEMS) and
683:irradiation
681:ultraviolet
659:irradiation
657:Ultraviolet
584:Chad Mirkin
256:Preparation
2855:Thin films
2844:Categories
2732:(9): 883.
1175:: 107–37.
1004:2066/75325
854:References
818:biosensors
757:biosensors
528:Patterning
313:(AFM) and
208:isotherm.
141:porphyrins
132:adsorption
2708:124513678
2539:CiteSeerX
2508:0743-7463
2321:0002-7863
2020:0743-7463
1616:Wysocki.
1326:cite book
1278:cite book
1115:Chem. Sci
1071:CiteSeerX
913:Chem. Rev
839:biosensor
834:biosensor
830:molecules
689:disorder.
422:θ
406:θ
403:−
2828:17914848
2808:Langmuir
2783:Langmuir
2774:14962551
2561:16365640
2516:16519501
2488:Langmuir
2469:19370233
2399:17177439
2364:21167979
2285:20845943
2265:Langmuir
2249:27011581
2206:21794185
2136:21214202
2116:Langmuir
2100:20058870
2080:Langmuir
2064:18980356
2044:Langmuir
2028:15875853
2000:Langmuir
1808:19015788
1743:20199099
1723:Langmuir
1701:Archived
1533:Langmuir
1518:17451727
1472:17274618
1437:24880913
1197:11326061
1135:97346507
1093:12792937
1013:19746490
933:15826011
826:proteins
377:Kinetics
362:colloids
193:hydroxyl
2686:Bibcode
2604:4420426
2584:Bibcode
2422:Bibcode
2344:Bibcode
2241:9924019
2221:Science
2197:3246612
2176:Bibcode
1973:Bibcode
1788:Bibcode
1498:Bibcode
1429:8662464
1409:Bibcode
1401:Science
1243:Bibcode
1177:Bibcode
1036:Bibcode
956:Bibcode
774:laminar
371:sensors
347:Defects
301:(SFG),
297:(SHG),
219:wetting
206:Frumkin
159:, etc.
153:silanes
2826:
2772:
2706:
2602:
2576:Nature
2559:
2541:
2514:
2506:
2467:
2397:
2362:
2319:
2283:
2247:
2239:
2204:
2194:
2134:
2098:
2062:
2026:
2018:
1806:
1741:
1516:
1470:
1435:
1427:
1195:
1133:
1091:
1073:
1011:
931:
765:fungus
726:Rain-X
451:Where
149:thiols
2704:S2CID
2600:S2CID
2245:S2CID
1681:(PDF)
1628:(PDF)
1621:(PDF)
1433:S2CID
1131:S2CID
1111:(PDF)
828:, or
822:cells
740:SAM.
212:Types
2824:PMID
2770:PMID
2659:link
2618:link
2557:PMID
2512:PMID
2504:ISSN
2465:PMID
2395:PMID
2360:PMID
2317:ISSN
2281:PMID
2237:PMID
2202:PMID
2132:PMID
2096:PMID
2060:PMID
2024:PMID
2016:ISSN
1866:link
1822:link
1804:PMID
1757:link
1739:PMID
1603:link
1559:link
1514:PMID
1468:PMID
1425:PMID
1332:link
1284:link
1211:link
1193:PMID
1089:PMID
1009:PMID
929:PMID
614:The
339:and
289:and
281:and
189:FDTS
183:. A
2816:doi
2791:doi
2762:doi
2734:doi
2694:doi
2682:528
2641:doi
2637:103
2592:doi
2580:396
2549:doi
2496:doi
2457:doi
2430:doi
2418:259
2387:doi
2383:128
2352:doi
2340:684
2309:doi
2305:113
2273:doi
2229:doi
2225:283
2192:PMC
2184:doi
2124:doi
2088:doi
2052:doi
2008:doi
1981:doi
1943:doi
1939:106
1916:doi
1912:112
1889:doi
1885:115
1848:doi
1844:114
1796:doi
1731:doi
1693:doi
1689:111
1656:doi
1652:105
1585:doi
1581:117
1541:doi
1506:doi
1494:312
1460:doi
1456:129
1417:doi
1405:272
1251:doi
1185:doi
1123:doi
1081:doi
1044:doi
999:hdl
991:doi
964:doi
921:doi
917:105
369:or
143:on
128:SAM
2846::
2822:.
2812:23
2810:.
2785:.
2768:.
2758:25
2756:.
2730:82
2728:.
2702:.
2692:.
2680:.
2676:.
2655:}}
2651:{{
2635:.
2614:}}
2610:{{
2598:.
2590:.
2578:.
2555:.
2547:.
2535:35
2533:.
2510:.
2502:.
2492:22
2490:.
2486:.
2463:.
2451:.
2428:.
2416:.
2393:.
2381:.
2358:.
2350:.
2338:.
2315:.
2303:.
2279:.
2269:26
2267:.
2243:.
2235:.
2223:.
2200:.
2190:.
2182:.
2170:.
2166:.
2144:^
2130:.
2120:27
2118:.
2094:.
2084:26
2082:.
2058:.
2048:24
2046:.
2022:.
2014:.
2004:20
2002:.
1979:.
1969:75
1967:.
1955:^
1937:.
1910:.
1883:.
1862:}}
1858:{{
1842:.
1830:^
1818:}}
1814:{{
1802:.
1794:.
1784:10
1782:.
1765:^
1753:}}
1749:{{
1737:.
1727:26
1725:.
1709:^
1699:.
1687:.
1683:.
1668:^
1650:.
1636:^
1599:}}
1595:{{
1579:.
1567:^
1555:}}
1551:{{
1537:10
1535:.
1512:.
1504:.
1492:.
1480:^
1466:.
1454:.
1431:.
1423:.
1415:.
1403:.
1358:^
1340:^
1328:}}
1324:{{
1292:^
1280:}}
1276:{{
1249:.
1239:65
1237:.
1219:^
1207:}}
1203:{{
1191:.
1183:.
1173:52
1171:.
1143:^
1129:.
1117:.
1113:.
1087:.
1079:.
1067:32
1065:.
1042:.
1032:19
1030:.
1007:.
997:.
987:48
985:.
962:.
952:50
950:.
927:.
915:.
861:^
841:.
824:,
343:.
234:CH
155:,
151:,
2830:.
2818::
2797:.
2793::
2787:5
2776:.
2764::
2740:.
2736::
2710:.
2696::
2688::
2661:)
2647:.
2643::
2620:)
2606:.
2594::
2586::
2563:.
2551::
2518:.
2498::
2471:.
2459::
2453:9
2436:.
2432::
2424::
2401:.
2389::
2366:.
2354::
2346::
2323:.
2311::
2287:.
2275::
2251:.
2231::
2208:.
2186::
2178::
2172:2
2138:.
2126::
2102:.
2090::
2066:.
2054::
2030:.
2010::
1987:.
1983::
1975::
1949:.
1945::
1922:.
1918::
1895:.
1891::
1868:)
1854:.
1850::
1824:)
1810:.
1798::
1790::
1759:)
1745:.
1733::
1695::
1662:.
1658::
1605:)
1591:.
1587::
1561:)
1547:.
1543::
1520:.
1508::
1500::
1474:.
1462::
1439:.
1419::
1411::
1334:)
1286:)
1257:.
1253::
1245::
1213:)
1199:.
1187::
1179::
1137:.
1125::
1119:1
1095:.
1083::
1050:.
1046::
1038::
1015:.
1001::
993::
970:.
966::
958::
935:.
923::
651:.
590:.
457:k
453:θ
436:.
430:t
427:d
419:d
413:=
409:)
400:1
397:(
394:k
236:2
232:4
230:H
228:6
226:C
224:2
126:(
112:e
105:t
98:v
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.