52:
306:
189:
34:
494:
43:
543:
1104:(After having tried so vainly to decompose Prussian blue by acids, I made recourse to alkalies. I put a half ounce of this blue in a flask, and I poured on it ten ounces of a solution of nitre fixed by tartar . As soon as these two substances had been mixed together, I saw with astonishment that, without the aid of heat, the blue color had entirely disappeared; the powder at the bottom of the flask had only a rather gray color: having put this vessel on a
1306:
669:
548:
1307:
Peter Aggett, Fernando
Aguilar, Riccardo Crebelli, Birgit Dusemund, Metka Filipič, Maria Jose Frutos, Pierre Galtier, David Gott, Ursula Gundert-Remy, Gunter Georg Kuhnle, Claude Lambré, Jean-Charles Leblanc, Inger Therese Lillegaard, Peter Moldeus, Alicja Mortensen, Agneta Oskarsson, Ivan Stankovic,
902:
are widely used as anticaking agents for both road salt and table salt. The potassium and sodium hexacyanidoferrates(II) are also used in the purification of tin and the separation of copper from molybdenum ores. Potassium hexacyanidoferrate(II) is used in the production of wine and citric acid.
546:
932:, giving a bright blue visualization where an antibody (or other molecule), conjugated to Beta-gal, has bonded to its target. On reacting with Fe(3) it gives a Prussian blue colour. Thus it is used as an identifying reagent for iron in labs.
975:
Like other metal cyanides, solid potassium hexacyanidoferrate(II), both as the hydrate and anhydrous salts, has a complicated polymeric structure. The polymer consists of octahedral centers crosslinked with K ions that are bound to the CN
502:
1108:
in order to heat the solution until it simmered, this gray color also disappeared entirely, and all that was contained in the flask, both the powder and the solution, had only a yellow color a little
474:
146:
1198:
547:
1037:
Five
Hundred Useful and Amusing Experiments in Chemistry, and in the Arts and Manufactures: With Observations on the Properties Employed, and Their Application to Useful Purposes
899:
2572:
2588:
549:
2417:
1144:
Gail, E.; Gos, S.; Kulzer, R.; Lorösch, J.; Rubo, A.; Sauer, M.; Kellens, R.; Reddy, J.; Steier, N.; Hasenpusch, W. (October 2011). "Cyano
Compounds, Inorganic".
2401:
2009:
1962:
1974:
1816:
1235:
2552:
1771:
1558:
682:
924:
reactions. Potassium hexacyanidoferrate(II) is used in a mixture with potassium ferricyanide and phosphate buffered solution to provide a buffer for
1359:
1424:
571:
1035:
1457:
1214:
345:
1102:"Après avoir essayé ainsi inutilement de décomposer le bleu de Prusse par les acides, … n'avoit plus qu'une couleur jaune un peu rousse."
1054:
677:
1310:"Re-evaluation of sodium ferrocyanide (E 535), potassium ferrocyanide (E 536) and calcium ferrocyanide (E 538) as food additives"
1262:"Polymorphism of Potassium Ferrocyanide Trihydrate as Studied by Solid-State Multinuclear NMR Spectroscopy and X-ray Diffraction"
807:, or dried blood. It was also obtained commercially from gasworks spent oxide (purification of city gas from hydrogen cyanide).
906:
In the EU, hexacyanidoferrates(II) (E 535–538) were, as of 2017, solely authorised in two food categories as salt additives.
791:
Historically, the compound was manufactured from nitrogenous organic material, iron filings, and potassium carbonate. Common
51:
1375:"Polarization dependence of XANES of square-planar Ni(CN)2−4 ion. A comparison with octahedral Fe(CN)4−6 and Fe(CN)3−6 ions"
1161:
1450:
1010:
839:
320:
689:
2433:
1239:
557:
2116:
739:(1718–1784) first reported the preparation of Potassium hexacyanidoferrate(II), which he achieved by reacting
2250:
2154:
1946:
2620:
2202:
2140:
1443:
254:
184:
775:
O. This solution is then treated with potassium salts to precipitate the mixed calcium-potassium salt CaK
493:
2635:
2625:
2178:
2166:
1690:
284:
2480:
2212:
2085:
2226:
196:
301:
2630:
2376:
2364:
2057:
1091:
2645:
1934:
1898:
1714:
1120:
457:
2340:
2332:
2288:
2276:
1986:
1910:
1645:
1503:
913:
632:
20:
912:
In the laboratory, potassium hexacyanidoferrate(II) is used to determine the concentration of
166:
2640:
2037:
1922:
1730:
1698:
1578:
1516:
1353:
516:
486:
64:
1308:
Ine
Waalkens-Berendsen, Rudolf Antonius Woutersen, Matthew Wright and Maged Younes. (2018).
2385:
1746:
1620:
1485:
828:
271:
263:
121:
113:
1096:
Histoire de l'Académie Royale des
Sciences …, § Mémoires de l'Académie royale des Sciences
8:
2508:
2320:
2069:
2025:
1889:
1804:
1795:
1682:
1662:
1603:
1568:
1550:
1466:
1181:
780:
744:
717:
647:
103:
1058:
305:
188:
2536:
2352:
2128:
2045:
1954:
1828:
1783:
1762:
1608:
1508:
1498:
1480:
1336:
925:
705:
1416:
862:
This reaction can be used to remove potassium hexacyanidoferrate(II) from a solution.
33:
2520:
2492:
2468:
2460:
2308:
1881:
1873:
1848:
1670:
1650:
1636:
1628:
1573:
1563:
1529:
1524:
1493:
1394:
1390:
1341:
1309:
1289:
1281:
1206:
1157:
950:
764:
760:
1078:
898:
Potassium hexacyanidoferrate(II) finds many niche applications in industry. It and
1542:
1537:
1386:
1331:
1321:
1273:
1149:
824:
756:
424:
368:
243:
1374:
1153:
1865:
1857:
1594:
939:
946:
736:
660:
607:
2614:
1398:
1326:
1285:
1261:
866:
740:
651:
522:
449:
413:
222:
177:
1260:
Willans, Mathew J.; Wasylishen, Roderick E.; McDonald, Robert (2009-05-18).
1345:
1293:
1015:
1005:
996:) at 6400 mg/kg. The kidneys are the organ for ferrocyanide toxicity.
943:
800:
570:
989:
935:
Potassium hexacyanidoferrate(II) can be used as a fertilizer for plants.
816:
583:
329:
InChI=1S/6CN.Fe.4K.3H2O/c6*1-2;;;;;;;;/h;;;;;;;;;;;3*1H2/q6*-1;+2;4*+1;;;
42:
556:
721:
563:
439:
383:
197:
157:
1435:
1277:
1205:. Vol. 2 (2nd ed.). New York: Academic Press. p. 1768.
1055:"POTASSIUM FERROCYANIDE MSDS Number: P5763 - Effective Date: 12/08/96"
1105:
917:
887:
1143:
942:, potassium hexacyanidoferrate(II) was the most important source of
659:
Except where otherwise noted, data are given for materials in their
980:. The K---NC linkages break when the solid is dissolved in water.
835:
792:
529:
214:
145:
1373:
Kosugi, Nobuhiro; Yokoyama, Toshihiko; Kuroda, Haruo (May 1986).
724:
564:
445:
403:
230:
1210:
977:
865:
A famous reaction involves treatment with ferric salts to give
796:
755:
Potassium hexacyanidoferrate(II) is produced industrially from
953:
was produced by decomposing potassium hexacyanidoferrate(II):
929:
921:
886:, this insoluble but deeply coloured material is the blue of
804:
135:
1425:"Potassium Ferrocyanide in Salt Is Entirely Safe To Consume"
1236:"EuSalt Expert Meeting on E 535 and E 536 as Feed Additives"
289:
1259:
641:
626:
1186:. New York: D. Appleton & Co. p. 474 & 477.
215:
1372:
82:
Potassium hexacyanoferrate (II) trihydrate
815:Treatment of potassium hexacyanidoferrate(II) with
838:gas, potassium hexacyanidoferrate(II) converts to
823:. After neutralization of this intermediate with
1094:[Chemical examination of Prussian blue].
1079:https://chem.nlm.nih.gov/chemidplus/rn/13943-58-3
2612:
242:
88:Ferrate hexacyano tetrapotassium trihydrate
1119:Munroe, Charles E.; Chatard, Thomas M. (1902).
545:
120:
112:
1146:Ullmann's Encyclopedia of Industrial Chemistry
1125:Twelfth Census of the United States: Bulletins
1451:
1121:"Manufactures: Chemicals and Allied Products"
1118:
1358:: CS1 maint: multiple names: authors list (
1203:Handbook of Preparative Inorganic Chemistry
938:Prior to 1900, before the invention of the
85:Tetrapotassium ferrocyanide trihydrate
1458:
1444:
1417:"Cyanide (inorganic) compounds fact sheet"
1179:
304:
187:
165:
1419:. National Pollutant Inventory Australia.
1335:
1325:
1139:
1137:
1135:
1049:
1047:
270:
262:
1196:
1175:
1173:
786:
390:422.388 g/mol (trihydrate)
1465:
1089:
300:
2613:
1132:
1044:
178:
1439:
1170:
810:
767:, the combination of which affords Ca
332:Key: UTYXJYFJPBYDKY-UHFFFAOYSA-N
909:It can also be used in animal feed.
750:
1092:"Éxamen chymique de bleu de Prusse"
716:O. It is the potassium salt of the
398:Light yellow, crystalline granules
233:
13:
988:The toxicity in rats is low, with
541:
14:
2657:
1409:
1057:. J. T. Baker Inc. Archived from
1011:Potassium hexacyanidoferrate(III)
840:potassium hexacyanidoferrate(III)
831:can be selectively crystallized.
783:to give the tetrapotassium salt.
27:Potassium hexacyanidoferrate(II)
779:, which in turn is treated with
702:Potassium hexacyanidoferrate(II)
667:
492:
68:Potassium hexacyanidoferrate(II)
50:
41:
32:
1366:
1300:
1253:
893:
720:. This salt forms lemon-yellow
663:(at 25 °C , 100 kPa).
1228:
1199:"Sodium nitrosyl cyanoferrate"
1190:
1112:
1083:
1072:
1028:
949:. In this historical process,
743:(iron(III) ferrocyanide) with
353:..N#C(C#N)(C#N)(C#N)(C#N)C#N..
1:
1183:Manual of chemical technology
1154:10.1002/14356007.a08_159.pub3
1021:
1391:10.1016/0301-0104(86)85034-0
970:
735:In 1752, the French chemist
730:
79:(Yellow) Prussiate of Potash
7:
1180:Von Wagner, Rudolf (1897).
999:
983:
916:, a compound often used in
596:or concentration (LD, LC):
434:28.9 g/100 mL (20 °C)
10:
2662:
928:, which is used to cleave
18:
2453:
2109:
2002:
1841:
1587:
1473:
869:. With the composition Fe
657:
618:
592:
473:
468:
361:
341:
316:
223:(acidity regulators, ...)
96:
73:
63:
58:
49:
40:
31:
1327:10.2903/j.efsa.2018.5374
1238:. EUSalt. Archived from
388:368.35 g/mol (anhydrous)
19:Not to be confused with
1201:. In Brauer, G. (ed.).
1148:. Weinheim: Wiley-VCH.
900:the related sodium salt
614:6400 mg/kg (oral, rat)
464:−130.0·10 cm/mol
458:Magnetic susceptibility
408:1.85 g/cm (trihydrate)
914:potassium permanganate
633:Potassium ferricyanide
552:
21:potassium ferricyanide
803:horn, leather scrap,
787:Historical production
737:Pierre Joseph Macquer
551:
1040:. Thomas Tegg. 1825.
834:Upon treatment with
829:sodium nitroprusside
718:coordination complex
534:(fire diamond)
2621:Potassium compounds
1474:H, (pseudo)halogens
1467:Potassium compounds
1266:Inorganic Chemistry
1098:(in French): 60–77.
781:potassium carbonate
745:potassium hydroxide
648:Sodium ferrocyanide
425:Solubility in water
28:
2636:E-number additives
2626:Iron(II) compounds
926:beta-galactosidase
827:, red crystals of
811:Chemical reactions
706:inorganic compound
690:Infobox references
619:Related compounds
553:
275: (trihydrate)
125: (trihydrate)
26:
2608:
2607:
2110:transition metals
1278:10.1021/ic802134j
1272:(10): 4342–4353.
1197:Seel, F. (1965).
1100:From pp. 63-64:
951:potassium cyanide
765:calcium hydroxide
761:iron(II) chloride
751:Modern production
698:Chemical compound
696:
695:
517:Hazard statements
285:CompTox Dashboard
267: (anhydrous)
147:Interactive image
117: (anhydrous)
16:Chemical compound
2653:
2260:
2246:
2245:
2244:
2236:
2235:
2222:
2208:
2198:
2197:
2196:
2188:
2187:
2150:
1460:
1453:
1446:
1437:
1436:
1432:
1420:
1403:
1402:
1379:Chemical Physics
1370:
1364:
1363:
1357:
1349:
1339:
1329:
1304:
1298:
1297:
1257:
1251:
1250:
1248:
1247:
1232:
1226:
1225:
1223:
1222:
1213:. Archived from
1194:
1188:
1187:
1177:
1168:
1167:
1141:
1130:
1128:
1116:
1110:
1099:
1090:Macquer (1752).
1087:
1081:
1076:
1070:
1069:
1067:
1066:
1051:
1042:
1041:
1032:
885:
884:
877:
876:
825:sodium carbonate
757:hydrogen cyanide
680:
674:
671:
670:
573:
566:
559:
544:
524:
496:
369:Chemical formula
309:
308:
293:
291:
274:
266:
246:
235:
217:
199:
191:
180:
169:
149:
124:
116:
54:
45:
36:
29:
25:
2661:
2660:
2656:
2655:
2654:
2652:
2651:
2650:
2631:Cyano complexes
2611:
2610:
2609:
2604:
2600:
2596:
2592:
2584:
2580:
2576:
2568:
2564:
2560:
2556:
2548:
2544:
2540:
2532:
2528:
2524:
2516:
2512:
2504:
2500:
2496:
2488:
2484:
2476:
2472:
2464:
2449:
2445:
2441:
2437:
2429:
2425:
2421:
2413:
2409:
2405:
2397:
2393:
2389:
2380:
2372:
2368:
2360:
2356:
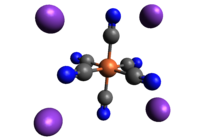
2348:
2344:
2336:
2328:
2324:
2316:
2312:
2304:
2300:
2296:
2292:
2284:
2280:
2272:
2268:
2259:
2255:
2251:
2243:
2240:
2239:
2238:
2234:
2231:
2230:
2229:
2227:
2221:
2217:
2213:
2207:
2203:
2195:
2192:
2191:
2190:
2186:
2183:
2182:
2181:
2179:
2174:
2170:
2162:
2158:
2149:
2145:
2141:
2136:
2132:
2124:
2120:
2105:
2101:
2097:
2093:
2089:
2081:
2077:
2073:
2065:
2061:
2053:
2049:
2041:
2033:
2029:
2021:
2017:
2013:
1998:
1994:
1990:
1982:
1978:
1970:
1966:
1958:
1950:
1942:
1938:
1930:
1926:
1918:
1914:
1906:
1902:
1893:
1885:
1877:
1869:
1861:
1852:
1837:
1832:
1824:
1820:
1812:
1808:
1799:
1791:
1787:
1779:
1775:
1766:
1758:
1754:
1750:
1742:
1738:
1734:
1726:
1722:
1718:
1710:
1706:
1702:
1694:
1686:
1678:
1674:
1666:
1658:
1654:
1640:
1632:
1624:
1616:
1612:
1598:
1583:
1554:
1546:
1533:
1520:
1512:
1489:
1469:
1464:
1423:
1415:
1412:
1407:
1406:
1371:
1367:
1351:
1350:
1305:
1301:
1258:
1254:
1245:
1243:
1234:
1233:
1229:
1220:
1218:
1195:
1191:
1178:
1171:
1164:
1142:
1133:
1117:
1113:
1088:
1084:
1077:
1073:
1064:
1062:
1053:
1052:
1045:
1034:
1033:
1029:
1024:
1002:
995:
986:
973:
967:
963:
959:
940:Castner process
896:
883:
880:
879:
878:
875:
872:
871:
870:
857:
853:
849:
822:
813:
789:
778:
774:
770:
753:
733:
715:
711:
699:
692:
687:
686:
685: ?)
676:
672:
668:
664:
650:
644:
629:
611:
605:
578:
577:
576:
575:
568:
561:
554:
550:
542:
519:
505:
489:
461:
433:
427:
389:
377:
371:
357:
354:
349:
348:
337:
334:
333:
330:
324:
323:
312:
294:
287:
278:
249:
236:
209:
172:
152:
139:
128:
106:
92:
91:
69:
24:
17:
12:
11:
5:
2659:
2649:
2648:
2646:Iron complexes
2643:
2638:
2633:
2628:
2623:
2606:
2605:
2603:
2602:
2598:
2594:
2590:
2586:
2582:
2578:
2574:
2570:
2566:
2562:
2558:
2554:
2550:
2546:
2542:
2538:
2534:
2530:
2526:
2522:
2518:
2514:
2510:
2506:
2502:
2498:
2494:
2490:
2486:
2482:
2478:
2474:
2470:
2466:
2462:
2457:
2455:
2451:
2450:
2448:
2447:
2443:
2439:
2435:
2431:
2427:
2423:
2419:
2415:
2411:
2407:
2403:
2399:
2395:
2391:
2387:
2383:
2378:
2374:
2370:
2366:
2362:
2358:
2354:
2350:
2346:
2342:
2338:
2334:
2330:
2326:
2322:
2318:
2314:
2310:
2306:
2302:
2298:
2294:
2290:
2286:
2282:
2278:
2274:
2270:
2266:
2262:
2257:
2253:
2248:
2241:
2232:
2224:
2219:
2215:
2210:
2205:
2200:
2193:
2184:
2176:
2172:
2168:
2164:
2160:
2156:
2152:
2147:
2143:
2138:
2134:
2130:
2126:
2122:
2118:
2113:
2111:
2107:
2106:
2104:
2103:
2099:
2095:
2091:
2087:
2083:
2079:
2075:
2071:
2067:
2063:
2059:
2055:
2051:
2047:
2043:
2039:
2035:
2031:
2027:
2023:
2019:
2015:
2011:
2006:
2004:
2000:
1999:
1997:
1996:
1992:
1988:
1984:
1980:
1976:
1972:
1968:
1964:
1960:
1956:
1952:
1948:
1944:
1940:
1936:
1932:
1928:
1924:
1920:
1916:
1912:
1908:
1904:
1900:
1896:
1891:
1887:
1883:
1879:
1875:
1871:
1867:
1863:
1859:
1855:
1850:
1845:
1843:
1839:
1838:
1836:
1835:
1830:
1826:
1822:
1818:
1814:
1810:
1806:
1802:
1797:
1793:
1789:
1785:
1781:
1777:
1773:
1769:
1764:
1760:
1756:
1752:
1748:
1744:
1740:
1736:
1732:
1728:
1724:
1720:
1716:
1712:
1708:
1704:
1700:
1696:
1692:
1688:
1684:
1680:
1676:
1672:
1668:
1664:
1660:
1656:
1652:
1648:
1643:
1638:
1634:
1630:
1626:
1622:
1618:
1614:
1610:
1606:
1601:
1596:
1591:
1589:
1585:
1584:
1582:
1581:
1576:
1571:
1566:
1561:
1556:
1552:
1548:
1544:
1540:
1535:
1531:
1527:
1522:
1518:
1514:
1510:
1506:
1501:
1496:
1491:
1487:
1483:
1477:
1475:
1471:
1470:
1463:
1462:
1455:
1448:
1440:
1434:
1433:
1421:
1411:
1410:External links
1408:
1405:
1404:
1385:(3): 449–453.
1365:
1299:
1252:
1227:
1189:
1169:
1163:978-3527306732
1162:
1131:
1111:
1082:
1071:
1043:
1026:
1025:
1023:
1020:
1019:
1018:
1013:
1008:
1001:
998:
993:
985:
982:
972:
969:
965:
961:
957:
895:
892:
881:
873:
860:
859:
855:
851:
847:
820:
812:
809:
788:
785:
776:
772:
768:
752:
749:
732:
729:
713:
709:
708:with formula K
697:
694:
693:
688:
666:
665:
661:standard state
658:
655:
654:
645:
639:
636:
635:
630:
624:
621:
620:
616:
615:
612:
603:
601:
598:
597:
590:
589:
588:Non-flammable
586:
580:
579:
569:
562:
555:
540:
539:
538:
537:
535:
526:
525:
520:
515:
512:
511:
506:
501:
498:
497:
490:
485:
482:
481:
471:
470:
466:
465:
462:
456:
453:
452:
442:
436:
435:
428:
423:
420:
419:
416:
410:
409:
406:
400:
399:
396:
392:
391:
386:
380:
379:
375:
372:
367:
364:
363:
359:
358:
356:
355:
352:
344:
343:
342:
339:
338:
336:
335:
331:
328:
327:
319:
318:
317:
314:
313:
311:
310:
302:DTXSID60892423
297:
295:
283:
280:
279:
277:
276:
268:
259:
257:
251:
250:
248:
247:
239:
237:
229:
226:
225:
219:
211:
210:
208:
207:
203:
201:
193:
192:
182:
174:
173:
171:
170:
162:
160:
154:
153:
151:
150:
142:
140:
133:
130:
129:
127:
126:
118:
109:
107:
102:
99:
98:
94:
93:
90:
89:
86:
83:
80:
76:
75:
71:
70:
67:
61:
60:
56:
55:
47:
46:
38:
37:
15:
9:
6:
4:
3:
2:
2658:
2647:
2644:
2642:
2639:
2637:
2634:
2632:
2629:
2627:
2624:
2622:
2619:
2618:
2616:
2601:
2587:
2585:
2571:
2569:
2551:
2549:
2535:
2533:
2519:
2517:
2507:
2505:
2491:
2489:
2479:
2477:
2467:
2465:
2459:
2458:
2456:
2452:
2446:
2432:
2430:
2416:
2414:
2400:
2398:
2384:
2382:
2375:
2373:
2363:
2361:
2351:
2349:
2339:
2337:
2331:
2329:
2319:
2317:
2307:
2305:
2287:
2285:
2275:
2273:
2263:
2261:
2249:
2247:
2225:
2223:
2211:
2209:
2201:
2199:
2177:
2175:
2165:
2163:
2153:
2151:
2139:
2137:
2127:
2125:
2115:
2114:
2112:
2108:
2102:
2084:
2082:
2068:
2066:
2056:
2054:
2044:
2042:
2036:
2034:
2024:
2022:
2008:
2007:
2005:
2001:
1995:
1985:
1983:
1973:
1971:
1961:
1959:
1953:
1951:
1945:
1943:
1933:
1931:
1921:
1919:
1909:
1907:
1897:
1895:
1888:
1886:
1880:
1878:
1872:
1870:
1864:
1862:
1856:
1854:
1847:
1846:
1844:
1840:
1834:
1827:
1825:
1815:
1813:
1803:
1801:
1794:
1792:
1782:
1780:
1770:
1768:
1761:
1759:
1745:
1743:
1729:
1727:
1713:
1711:
1697:
1695:
1689:
1687:
1681:
1679:
1669:
1667:
1661:
1659:
1649:
1647:
1644:
1642:
1635:
1633:
1627:
1625:
1619:
1617:
1607:
1605:
1602:
1600:
1593:
1592:
1590:
1586:
1580:
1577:
1575:
1572:
1570:
1567:
1565:
1562:
1560:
1557:
1555:
1549:
1547:
1541:
1539:
1536:
1534:
1528:
1526:
1523:
1521:
1515:
1513:
1507:
1505:
1502:
1500:
1497:
1495:
1492:
1490:
1484:
1482:
1479:
1478:
1476:
1472:
1468:
1461:
1456:
1454:
1449:
1447:
1442:
1441:
1438:
1430:
1426:
1422:
1418:
1414:
1413:
1400:
1396:
1392:
1388:
1384:
1380:
1376:
1369:
1361:
1355:
1347:
1343:
1338:
1333:
1328:
1323:
1319:
1315:
1311:
1303:
1295:
1291:
1287:
1283:
1279:
1275:
1271:
1267:
1263:
1256:
1242:on 2019-05-12
1241:
1237:
1231:
1217:on 2010-03-07
1216:
1212:
1208:
1204:
1200:
1193:
1185:
1184:
1176:
1174:
1165:
1159:
1155:
1151:
1147:
1140:
1138:
1136:
1127:(210): 1–306.
1126:
1122:
1115:
1107:
1103:
1097:
1093:
1086:
1080:
1075:
1061:on 2015-11-21
1060:
1056:
1050:
1048:
1039:
1038:
1031:
1027:
1017:
1014:
1012:
1009:
1007:
1004:
1003:
997:
991:
981:
979:
968:
960:→ 4 KCN + FeC
954:
952:
948:
945:
941:
936:
933:
931:
927:
923:
919:
915:
910:
907:
904:
901:
891:
889:
868:
867:Prussian blue
863:
845:
844:
843:
841:
837:
832:
830:
826:
818:
808:
806:
802:
799:sources were
798:
794:
784:
782:
766:
762:
758:
748:
746:
742:
741:Prussian blue
738:
728:
726:
723:
719:
707:
703:
691:
684:
679:
662:
656:
653:
652:Prussian blue
649:
646:
643:
638:
637:
634:
631:
628:
623:
622:
617:
613:
609:
600:
599:
595:
591:
587:
585:
582:
581:
574:
567:
560:
536:
533:
532:
528:
527:
521:
518:
514:
513:
510:
507:
504:
500:
499:
495:
491:
488:
484:
483:
479:
477:
472:
467:
463:
459:
455:
454:
451:
447:
444:insoluble in
443:
441:
438:
437:
432:
429:
426:
422:
421:
418:(decomposes)
417:
415:
414:Boiling point
412:
411:
407:
405:
402:
401:
397:
394:
393:
387:
385:
382:
381:
373:
370:
366:
365:
360:
351:
350:
347:
340:
326:
325:
322:
315:
307:
303:
299:
298:
296:
286:
282:
281:
273:
269:
265:
261:
260:
258:
256:
253:
252:
245:
241:
240:
238:
232:
228:
227:
224:
220:
218:
213:
212:
205:
204:
202:
200:
195:
194:
190:
186:
183:
181:
179:ECHA InfoCard
176:
175:
168:
164:
163:
161:
159:
156:
155:
148:
144:
143:
141:
137:
132:
131:
123:
119:
115:
111:
110:
108:
105:
101:
100:
95:
87:
84:
81:
78:
77:
72:
66:
62:
57:
53:
48:
44:
39:
35:
30:
22:
2641:Nephrotoxins
2264:
1428:
1382:
1378:
1368:
1354:cite journal
1317:
1314:EFSA Journal
1313:
1302:
1269:
1265:
1255:
1244:. Retrieved
1240:the original
1230:
1219:. Retrieved
1215:the original
1202:
1192:
1182:
1145:
1129:; see p. 31.
1124:
1114:
1101:
1095:
1085:
1074:
1063:. Retrieved
1059:the original
1036:
1030:
1016:Ferricyanide
1006:Ferrocyanide
987:
974:
955:
944:alkali metal
937:
934:
911:
908:
905:
897:
894:Applications
888:blueprinting
864:
861:
833:
814:
790:
754:
734:
701:
700:
593:
530:
508:
475:
430:
97:Identifiers
74:Other names
1320:(7): 5374.
990:lethal dose
817:nitric acid
608:median dose
594:Lethal dose
584:Flash point
503:Signal word
395:Appearance
362:Properties
185:100.034.279
2615:Categories
2003:B, C group
1842:pnictogens
1588:chalcogens
1429:rediff.com
1246:2018-12-06
1221:2017-09-10
1065:2012-04-08
1022:References
918:titrations
722:monoclinic
487:Pictograms
440:Solubility
431:trihydrate
384:Molar mass
272:961WP42S65
264:GTP1P30292
158:ChemSpider
134:3D model (
122:14459-95-1
114:13943-58-3
104:CAS Number
65:IUPAC name
1399:0301-0104
1286:0020-1669
1106:sand bath
971:Structure
920:based on
801:torrified
731:Synthesis
478:labelling
206:237-722-2
198:EC Number
1346:32626000
1294:19425611
1211:63-14307
1000:See also
984:Toxicity
947:cyanides
836:chlorine
793:nitrogen
725:crystals
531:NFPA 704
469:Hazards
460:(χ)
216:E number
167:20162028
2454:organic
1337:7009536
978:ligands
858:+ 2 KCl
819:gives H
704:is the
683:what is
681: (
642:cations
509:Warning
446:ethanol
404:Density
378:
231:PubChem
2281:Fe(CN)
2269:Fe(CN)
2133:Pt(CN)
1397:
1344:
1334:
1292:
1284:
1209:
1160:
797:carbon
763:, and
678:verify
675:
640:Other
627:anions
625:Other
346:SMILES
244:161067
59:Names
1109:red.)
930:X-Gal
922:redox
854:→ 2 K
805:offal
450:ether
321:InChI
221:E536
136:JSmol
2461:KHCO
2377:KCrO
2333:KMnO
2293:Fe(C
2218:ReBr
2204:KAsF
2171:ReCl
2159:PtCl
2121:PtCl
2038:KHCO
1979:HAsO
1955:KAsO
1691:KHSO
1683:KHSO
1663:KHSO
1579:KSCN
1574:KOCN
1569:KCNO
1530:KBrO
1517:KClO
1509:KClO
1504:KClO
1395:ISSN
1360:link
1342:PMID
1290:PMID
1282:ISSN
1207:LCCN
1158:ISBN
850:+ Cl
795:and
771:·11H
523:H411
255:UNII
2509:KHC
2481:KCF
2469:KCH
2369:CrO
2357:CrO
2345:CrO
2325:MnO
2313:FeO
2256:ZrF
2237:ReI
2189:ReF
2146:TiF
2062:SiF
2050:SiO
1991:AsO
1967:AsO
1947:KPF
1927:HPO
1882:KNO
1874:KNO
1858:KNH
1821:TeO
1809:TeO
1788:SeO
1776:SeO
1646:KHS
1604:KOH
1564:KCN
1559:KAt
1551:KIO
1543:KIO
1525:KBr
1499:KCl
1486:KHF
1387:doi
1383:104
1332:PMC
1322:doi
1274:doi
1150:doi
992:(LD
964:+ N
846:2 K
759:,
712:·3H
476:GHS
290:EPA
234:CID
2617::
2597:KO
2581:KO
2543:35
2539:18
2537:KC
2527:23
2523:12
2521:KC
2485:CO
2473:CO
2442:Cl
2438:Mo
2428:13
2422:Cr
2412:10
2406:Cr
2390:Cr
2381:Cl
2090:Al
2074:Al
2030:CO
1987:KH
1939:PO
1935:KH
1915:PO
1903:PO
1899:KH
1866:KN
1833:Po
1800:Te
1767:Se
1675:SO
1655:SO
1629:KO
1621:KO
1538:KI
1494:KH
1481:KF
1427:.
1393:.
1381:.
1377:.
1356:}}
1352:{{
1340:.
1330:.
1318:16
1316:.
1312:.
1288:.
1280:.
1270:48
1268:.
1264:.
1172:^
1156:.
1134:^
1123:.
1046:^
994:50
890:.
842::
747:.
727:.
604:50
602:LD
480::
448:,
2599:4
2595:7
2593:H
2591:5
2589:C
2583:4
2579:6
2577:H
2575:4
2573:C
2567:4
2565:O
2563:2
2561:K
2559:2
2557:H
2555:3
2553:C
2547:2
2545:O
2541:H
2531:2
2529:O
2525:H
2515:4
2513:O
2511:2
2503:4
2501:O
2499:2
2497:C
2495:2
2493:K
2487:2
2483:3
2475:2
2471:3
2463:2
2444:8
2440:2
2436:4
2434:K
2426:O
2424:4
2420:2
2418:K
2410:O
2408:3
2404:2
2402:K
2396:7
2394:O
2392:2
2388:2
2386:K
2379:3
2371:8
2367:3
2365:K
2359:4
2355:2
2353:K
2347:4
2343:3
2341:K
2335:4
2327:4
2323:2
2321:K
2315:4
2311:2
2309:K
2303:3
2301:)
2299:4
2297:O
2295:2
2291:3
2289:K
2283:6
2279:3
2277:K
2271:6
2267:4
2265:K
2258:6
2254:2
2252:K
2242:6
2233:2
2228:K
2220:6
2216:2
2214:K
2206:6
2194:6
2185:2
2180:K
2173:6
2169:2
2167:K
2161:6
2157:2
2155:K
2148:6
2144:2
2142:K
2135:4
2131:2
2129:K
2123:4
2119:2
2117:K
2100:7
2098:O
2096:2
2094:B
2092:2
2088:2
2086:K
2080:4
2078:O
2076:2
2072:2
2070:K
2064:6
2060:2
2058:K
2052:3
2048:2
2046:K
2040:3
2032:3
2028:2
2026:K
2020:7
2018:O
2016:2
2014:K
2012:4
2010:B
1993:4
1989:2
1981:4
1977:2
1975:K
1969:4
1965:3
1963:K
1957:2
1949:6
1941:4
1937:2
1929:4
1925:2
1923:K
1917:4
1913:3
1911:K
1905:3
1901:2
1894:P
1892:3
1890:K
1884:3
1876:2
1868:3
1860:2
1853:N
1851:3
1849:K
1831:2
1829:K
1823:4
1819:2
1817:K
1811:3
1807:2
1805:K
1798:2
1796:K
1790:4
1786:2
1784:K
1778:3
1774:2
1772:K
1765:2
1763:K
1757:8
1755:O
1753:2
1751:S
1749:2
1747:K
1741:7
1739:O
1737:2
1735:S
1733:2
1731:K
1725:5
1723:O
1721:2
1719:S
1717:2
1715:K
1709:3
1707:O
1705:2
1703:S
1701:2
1699:K
1693:5
1685:4
1677:4
1673:2
1671:K
1665:3
1657:3
1653:2
1651:K
1641:S
1639:2
1637:K
1631:3
1623:2
1615:2
1613:O
1611:2
1609:K
1599:O
1597:2
1595:K
1553:4
1545:3
1532:3
1519:4
1511:3
1488:2
1459:e
1452:t
1445:v
1431:.
1401:.
1389::
1362:)
1348:.
1324::
1296:.
1276::
1249:.
1224:.
1166:.
1152::
1068:.
966:2
962:2
958:4
956:K
882:3
874:4
856:3
852:2
848:4
821:2
777:2
773:2
769:2
714:2
710:4
673:N
610:)
606:(
572:0
565:0
558:1
376:4
374:K
292:)
288:(
138:)
23:.
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.