213:, where an electron in the conduction band annihilates a hole in the valence band, releasing the excess energy as a photon. This is possible in a direct band gap semiconductor if the electron has a k-vector near the conduction band minimum (the hole will share the same k-vector), but not possible in an indirect band gap semiconductor, as photons cannot carry crystal momentum, and thus conservation of crystal momentum would be violated. For radiative recombination to occur in an indirect band gap material, the process must also involve the absorption or emission of a
105:
143:
126:
1374:
289:
The absorption spectrum of an indirect band gap material usually depends more on temperature than that of a direct material, because at low temperatures there are fewer phonons, and therefore it is less likely that a photon and phonon can be simultaneously absorbed to create an indirect transition.
1146:
This formula is valid only for light with photon energy larger, but not too much larger, than the band gap (more specifically, this formula assumes the bands are approximately parabolic), and ignores all other sources of absorption other than the band-to-band absorption in question, as well as the
258:
The exact reverse of radiative recombination is light absorption. For the same reason as above, light with a photon energy close to the band gap can penetrate much farther before being absorbed in an indirect band gap material than a direct band gap one (at least insofar as the light absorption is
245:
loop in the material. At the edge of the loop, the planes above and beneath the "dislocation disk" are pulled apart, creating a negative pressure, which raises the energy of the conduction band substantially, with the result that the electrons cannot pass this edge. Provided that the area directly
236:
The fact that radiative recombination is slow in indirect band gap materials also means that, under most circumstances, radiative recombinations will be a small proportion of total recombinations, with most recombinations being non-radiative, taking place at point defects or at
112:
for a semiconductor with an indirect band gap, showing that an electron cannot shift from the highest-energy state in the valence band (red) to the lowest-energy state in the conduction band (green) without a change in momentum. Here, almost all of the energy comes from a
1164:
221:, which performs essentially the same role. The involvement of the phonon makes this process much less likely to occur in a given span of time, which is why radiative recombination is far slower in indirect band gap materials than direct band gap ones. This is why
875:
530:
241:. However, if the excited electrons are prevented from reaching these recombination places, they have no choice but to eventually fall back into the valence band by radiative recombination. This can be done by creating a
266:(solar cells). Crystalline silicon is the most common solar-cell substrate material, despite the fact that it is indirect-gap and therefore does not absorb light very well. As such, they are typically hundreds of
133:
for a semiconductor with a direct band gap, showing that an electron can shift from the highest-energy state in the valence band (red) to the lowest-energy state in the conduction band (green) without a change in
397:
1369:{\displaystyle \alpha \propto {\frac {(h\nu -E_{\text{g}}+E_{\text{p}})^{2}}{\exp({\frac {E_{\text{p}}}{kT}})-1}}+{\frac {(h\nu -E_{\text{g}}-E_{\text{p}})^{2}}{1-\exp(-{\frac {E_{\text{p}}}{kT}})}}}
74:; an electron can directly emit a photon. In an "indirect" gap, a photon cannot be emitted because the electron must pass through an intermediate state and transfer momentum to the crystal lattice.
250:
possible), the electrons will fall back into the valence shell by radiative recombination, thus emitting light. This is the principle on which "DELEDs" (Dislocation
Engineered LEDs) are based.
1671:
In some materials with an indirect gap, the value of the gap is negative. The top of the valence band is higher than the bottom of the conduction band in energy. Such materials are known as
1661:
779:
402:
939:
907:
714:
66:. If the k-vectors are different, the material has an "indirect gap". The band gap is called "direct" if the crystal momentum of electrons and holes is the same in both the
1602:
1053:
1407:
1085:
970:
743:
1518:
1628:
1544:
556:
338:
1136:
673:
1745:
1567:
1491:
772:
627:
1105:
651:
578:
1458:
1433:
1021:
996:
600:
209:(i.e., conservation of total k-vector). A photon with an energy near a semiconductor band gap has almost zero momentum. One important process is called
286:), which absorb the light in a much thinner region, and consequently can be made with a very thin active layer (often less than 1 micron thick).
1779:
1604:
forms a straight line, it can normally be inferred that there is an indirect band gap, measurable by extrapolating the straight line to the
1520:
forms a straight line, it can normally be inferred that there is a direct band gap, measurable by extrapolating the straight line to the
346:
270:
thick; thinner wafers would allow much of the light (particularly in longer wavelengths) to simply pass through. By comparison,
942:
170:
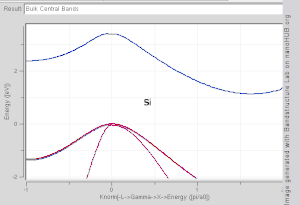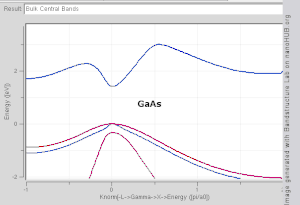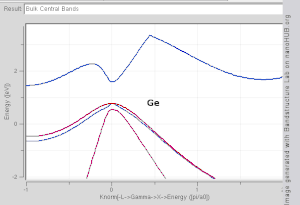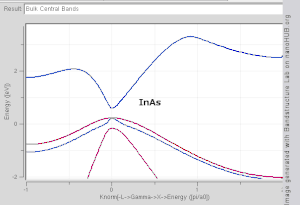
model. Note that Si and Ge are indirect band gap with minima at X and L, while GaAs and InAs are direct band gap materials.
217:, where the phonon momentum equals the difference between the electron and hole momentum. It can also, instead, involve a
1799:
17:
314:
against photon energy, one can normally tell both what value the band gap is, and whether or not it is direct.
279:
138:. Depicted is a transition in which a photon excites an electron from the valence band to the conduction band.
1633:
870:{\displaystyle m_{\text{r}}={\frac {m_{\text{h}}^{*}m_{\text{e}}^{*}}{m_{\text{h}}^{*}+m_{\text{e}}^{*}}}}
525:{\displaystyle A^{*}={\frac {q^{2}x_{vc}^{2}(2m_{\text{r}})^{3/2}}{\lambda _{0}\epsilon _{0}\hbar ^{3}n}}}
247:
912:
880:
1461:
290:
For example, silicon is opaque to visible light at room temperature, but transparent to red light at
1155:, or in the case that many of the valence band states are empty or conduction band states are full.
682:
1804:
1684:
1572:
1031:
1385:
1063:
948:
721:
1776:
1496:
1138:
is a "matrix element", with units of length and typical value the same order of magnitude as the
676:
303:
218:
210:
180:
1607:
1523:
318:
311:
202:
541:
323:
31:
1747:. However, in the Pankove version, the units / dimensional analysis appears not to work out.
1111:
658:
1723:
1549:
1473:
1152:
750:
609:
271:
222:
1090:
636:
563:
8:
1056:
1024:
302:
A common and simple method for determining whether a band gap is direct or indirect uses
229:
are almost always made of direct band gap materials, and not indirect band gap ones like
90:
1443:
1436:
1418:
1006:
981:
585:
1786:
at
Electrical and Computer Engineering Department of University of Colorado at Boulder
999:
275:
78:
1139:
206:
135:
130:
109:
59:
294:
temperatures, because red photons can only be absorbed in an indirect transition.
1783:
603:
67:
51:
238:
147:
63:
1793:
291:
263:
190:
167:
39:
973:
71:
55:
1720:
Pankove has the same equation, but with an apparently different prefactor
242:
226:
1147:
electrical attraction between the newly created electron and hole (see
1689:
1672:
307:
155:
94:
142:
186:
104:
35:
27:
Types of energy range in a solid where no electron states can exist
340:
is related to light frequency according to the following formula:
274:
are made of direct band gap materials (such as amorphous silicon,
1148:
230:
151:
97:. Some III–V materials are indirect bandgap as well, for example
1151:). It is also invalid in the case that the direct transition is
392:{\displaystyle \alpha \approx A^{*}{\sqrt {h\nu -E_{\text{g}}}}}
117:(vertical arrow), while almost all of the momentum comes from a
1467:
This formula involves the same approximations mentioned above.
1410:
630:
267:
214:
198:
194:
118:
114:
1158:
On the other hand, for an indirect band gap, the formula is:
558:
is the absorption coefficient, a function of light frequency
283:
174:
163:
159:
98:
86:
82:
77:
Examples of direct bandgap materials include hydrogenated
125:
1777:
B. Van
Zeghbroeck's Principles of Semiconductor Devices
1726:
1636:
1610:
1575:
1552:
1526:
1499:
1476:
1446:
1421:
1388:
1167:
1114:
1093:
1066:
1034:
1009:
984:
951:
915:
883:
782:
753:
724:
685:
661:
639:
612:
588:
566:
544:
405:
349:
326:
253:
1739:
1655:
1622:
1596:
1561:
1538:
1512:
1485:
1452:
1427:
1401:
1368:
1130:
1099:
1079:
1047:
1015:
990:
964:
933:
901:
869:
766:
737:
708:
667:
645:
621:
594:
572:
550:
524:
391:
332:
1791:
1087:is the vacuum wavelength for light of frequency
259:due to exciting electrons across the band gap).
246:above the dislocation loop is defect-free (no
201:, and other particles are required to satisfy
1755:
1753:
1711:, by E. Rosencher, 2002, equation (7.25).
774:is a certain constant, with formula above
297:
945:of the electron and hole, respectively (
175:Implications for radiative recombination
141:
124:
103:
14:
1792:
1750:
1546:axis. On the other hand, if a plot of
1656:{\displaystyle E_{\text{p}}\approx 0}
89:. Indirect bandgap materials include
58:are each characterized by a certain
54:and the maximal-energy state in the
50:. The minimal-energy state in the
1761:Optical Processes in Semiconductors
24:
25:
1816:
1770:
254:Implications for light absorption
81:and some III–V materials such as
1666:
934:{\displaystyle m_{\text{h}}^{*}}
902:{\displaystyle m_{\text{e}}^{*}}
262:This fact is very important for
1714:
1702:
1413:that assists in the transition
1360:
1332:
1309:
1273:
1255:
1230:
1213:
1177:
709:{\displaystyle \hbar =h/2\pi }
467:
450:
13:
1:
1695:
1597:{\displaystyle \alpha ^{1/2}}
1048:{\displaystyle \epsilon _{0}}
42:can be of two basic types, a
1402:{\displaystyle E_{\text{p}}}
1080:{\displaystyle \lambda _{0}}
965:{\displaystyle m_{\text{r}}}
738:{\displaystyle E_{\text{g}}}
7:
1678:
1513:{\displaystyle \alpha ^{2}}
317:For a direct band gap, the
248:non-radiative recombination
10:
1821:
1800:Electronic band structures
178:
1623:{\displaystyle \alpha =0}
1539:{\displaystyle \alpha =0}
1462:thermodynamic temperature
1470:Therefore, if a plot of
677:reduced Planck constant
551:{\displaystyle \alpha }
333:{\displaystyle \alpha }
308:plotting certain powers
304:absorption spectroscopy
219:crystallographic defect
211:radiative recombination
181:Radiative recombination
1741:
1657:
1624:
1598:
1563:
1540:
1514:
1487:
1454:
1429:
1403:
1370:
1132:
1131:{\displaystyle x_{vc}}
1101:
1081:
1049:
1017:
992:
966:
935:
903:
871:
768:
745:is the band gap energy
739:
710:
669:
668:{\displaystyle \hbar }
647:
623:
596:
574:
552:
526:
393:
334:
319:absorption coefficient
312:absorption coefficient
298:Formula for absorption
203:conservation of energy
171:
139:
122:
1742:
1740:{\displaystyle A^{*}}
1658:
1625:
1599:
1564:
1562:{\displaystyle h\nu }
1541:
1515:
1488:
1486:{\displaystyle h\nu }
1455:
1430:
1409:is the energy of the
1404:
1371:
1133:
1102:
1082:
1050:
1018:
993:
967:
936:
904:
872:
769:
767:{\displaystyle A^{*}}
740:
711:
670:
648:
624:
622:{\displaystyle h\nu }
597:
575:
553:
527:
394:
335:
272:thin-film solar cells
145:
128:
107:
32:semiconductor physics
1724:
1685:Moss–Burstein effect
1634:
1608:
1573:
1550:
1524:
1497:
1474:
1444:
1419:
1386:
1165:
1112:
1100:{\displaystyle \nu }
1091:
1064:
1032:
1007:
982:
949:
913:
881:
780:
751:
722:
683:
659:
646:{\displaystyle \nu }
637:
610:
586:
573:{\displaystyle \nu }
564:
542:
403:
347:
324:
1057:vacuum permittivity
1025:index of refraction
930:
898:
863:
845:
828:
813:
629:is the energy of a
449:
185:Interactions among
121:(horizontal arrow).
91:crystalline silicon
1782:2009-01-22 at the
1737:
1653:
1620:
1594:
1559:
1536:
1510:
1483:
1450:
1437:Boltzmann constant
1425:
1399:
1366:
1128:
1097:
1077:
1045:
1013:
988:
962:
931:
916:
899:
884:
867:
849:
831:
814:
799:
764:
735:
706:
665:
643:
619:
592:
580:is light frequency
570:
548:
522:
432:
389:
330:
172:
140:
123:
62:(k-vector) in the
1644:
1453:{\displaystyle T}
1428:{\displaystyle k}
1396:
1364:
1358:
1347:
1305:
1292:
1265:
1253:
1242:
1209:
1196:
1016:{\displaystyle n}
1000:elementary charge
991:{\displaystyle q}
959:
923:
891:
865:
856:
838:
821:
806:
790:
732:
595:{\displaystyle h}
520:
463:
387:
384:
79:amorphous silicon
48:indirect band gap
16:(Redirected from
1812:
1764:
1757:
1748:
1746:
1744:
1743:
1738:
1736:
1735:
1718:
1712:
1706:
1662:
1660:
1659:
1654:
1646:
1645:
1642:
1629:
1627:
1626:
1621:
1603:
1601:
1600:
1595:
1593:
1592:
1588:
1568:
1566:
1565:
1560:
1545:
1543:
1542:
1537:
1519:
1517:
1516:
1511:
1509:
1508:
1492:
1490:
1489:
1484:
1459:
1457:
1456:
1451:
1434:
1432:
1431:
1426:
1408:
1406:
1405:
1400:
1398:
1397:
1394:
1375:
1373:
1372:
1367:
1365:
1363:
1359:
1357:
1349:
1348:
1345:
1339:
1318:
1317:
1316:
1307:
1306:
1303:
1294:
1293:
1290:
1271:
1266:
1264:
1254:
1252:
1244:
1243:
1240:
1234:
1222:
1221:
1220:
1211:
1210:
1207:
1198:
1197:
1194:
1175:
1140:lattice constant
1137:
1135:
1134:
1129:
1127:
1126:
1106:
1104:
1103:
1098:
1086:
1084:
1083:
1078:
1076:
1075:
1054:
1052:
1051:
1046:
1044:
1043:
1022:
1020:
1019:
1014:
997:
995:
994:
989:
971:
969:
968:
963:
961:
960:
957:
943:effective masses
940:
938:
937:
932:
929:
924:
921:
908:
906:
905:
900:
897:
892:
889:
876:
874:
873:
868:
866:
864:
862:
857:
854:
844:
839:
836:
829:
827:
822:
819:
812:
807:
804:
797:
792:
791:
788:
773:
771:
770:
765:
763:
762:
744:
742:
741:
736:
734:
733:
730:
715:
713:
712:
707:
699:
674:
672:
671:
666:
652:
650:
649:
644:
628:
626:
625:
620:
601:
599:
598:
593:
579:
577:
576:
571:
557:
555:
554:
549:
531:
529:
528:
523:
521:
519:
515:
514:
505:
504:
495:
494:
484:
483:
482:
478:
465:
464:
461:
448:
443:
431:
430:
420:
415:
414:
398:
396:
395:
390:
388:
386:
385:
382:
367:
365:
364:
339:
337:
336:
331:
239:grain boundaries
207:crystal momentum
136:crystal momentum
131:crystal momentum
110:crystal momentum
60:crystal momentum
21:
1820:
1819:
1815:
1814:
1813:
1811:
1810:
1809:
1805:Optoelectronics
1790:
1789:
1784:Wayback Machine
1773:
1768:
1767:
1758:
1751:
1731:
1727:
1725:
1722:
1721:
1719:
1715:
1709:Optoelectronics
1707:
1703:
1698:
1681:
1669:
1641:
1637:
1635:
1632:
1631:
1630:axis (assuming
1609:
1606:
1605:
1584:
1580:
1576:
1574:
1571:
1570:
1551:
1548:
1547:
1525:
1522:
1521:
1504:
1500:
1498:
1495:
1494:
1475:
1472:
1471:
1445:
1442:
1441:
1420:
1417:
1416:
1393:
1389:
1387:
1384:
1383:
1350:
1344:
1340:
1338:
1319:
1312:
1308:
1302:
1298:
1289:
1285:
1272:
1270:
1245:
1239:
1235:
1233:
1223:
1216:
1212:
1206:
1202:
1193:
1189:
1176:
1174:
1166:
1163:
1162:
1119:
1115:
1113:
1110:
1109:
1092:
1089:
1088:
1071:
1067:
1065:
1062:
1061:
1039:
1035:
1033:
1030:
1029:
1008:
1005:
1004:
983:
980:
979:
956:
952:
950:
947:
946:
925:
920:
914:
911:
910:
893:
888:
882:
879:
878:
858:
853:
840:
835:
830:
823:
818:
808:
803:
798:
796:
787:
783:
781:
778:
777:
758:
754:
752:
749:
748:
729:
725:
723:
720:
719:
695:
684:
681:
680:
660:
657:
656:
638:
635:
634:
633:with frequency
611:
608:
607:
604:Planck constant
587:
584:
583:
565:
562:
561:
543:
540:
539:
510:
506:
500:
496:
490:
486:
485:
474:
470:
466:
460:
456:
444:
436:
426:
422:
421:
419:
410:
406:
404:
401:
400:
381:
377:
366:
360:
356:
348:
345:
344:
325:
322:
321:
300:
256:
183:
177:
166:generated with
68:conduction band
52:conduction band
44:direct band gap
28:
23:
22:
18:Direct band gap
15:
12:
11:
5:
1818:
1808:
1807:
1802:
1788:
1787:
1772:
1771:External links
1769:
1766:
1765:
1763:. Dover, 1971.
1759:J.I. Pankove,
1749:
1734:
1730:
1713:
1700:
1699:
1697:
1694:
1693:
1692:
1687:
1680:
1677:
1668:
1665:
1652:
1649:
1640:
1619:
1616:
1613:
1591:
1587:
1583:
1579:
1558:
1555:
1535:
1532:
1529:
1507:
1503:
1482:
1479:
1465:
1464:
1449:
1439:
1424:
1414:
1392:
1377:
1376:
1362:
1356:
1353:
1343:
1337:
1334:
1331:
1328:
1325:
1322:
1315:
1311:
1301:
1297:
1288:
1284:
1281:
1278:
1275:
1269:
1263:
1260:
1257:
1251:
1248:
1238:
1232:
1229:
1226:
1219:
1215:
1205:
1201:
1192:
1188:
1185:
1182:
1179:
1173:
1170:
1144:
1143:
1125:
1122:
1118:
1107:
1096:
1074:
1070:
1059:
1042:
1038:
1027:
1023:is the (real)
1012:
1002:
987:
977:
955:
928:
919:
896:
887:
861:
852:
848:
843:
834:
826:
817:
811:
802:
795:
786:
775:
761:
757:
746:
728:
717:
705:
702:
698:
694:
691:
688:
664:
654:
642:
618:
615:
591:
581:
569:
559:
547:
533:
532:
518:
513:
509:
503:
499:
493:
489:
481:
477:
473:
469:
459:
455:
452:
447:
442:
439:
435:
429:
425:
418:
413:
409:
380:
376:
373:
370:
363:
359:
355:
352:
329:
299:
296:
255:
252:
223:light-emitting
176:
173:
148:band structure
64:Brillouin zone
26:
9:
6:
4:
3:
2:
1817:
1806:
1803:
1801:
1798:
1797:
1795:
1785:
1781:
1778:
1775:
1774:
1762:
1756:
1754:
1732:
1728:
1717:
1710:
1705:
1701:
1691:
1688:
1686:
1683:
1682:
1676:
1674:
1667:Other aspects
1664:
1650:
1647:
1638:
1617:
1614:
1611:
1589:
1585:
1581:
1577:
1556:
1553:
1533:
1530:
1527:
1505:
1501:
1480:
1477:
1468:
1463:
1447:
1440:
1438:
1422:
1415:
1412:
1390:
1382:
1381:
1380:
1354:
1351:
1341:
1335:
1329:
1326:
1323:
1320:
1313:
1299:
1295:
1286:
1282:
1279:
1276:
1267:
1261:
1258:
1249:
1246:
1236:
1227:
1224:
1217:
1203:
1199:
1190:
1186:
1183:
1180:
1171:
1168:
1161:
1160:
1159:
1156:
1154:
1150:
1141:
1123:
1120:
1116:
1108:
1094:
1072:
1068:
1060:
1058:
1040:
1036:
1028:
1026:
1010:
1003:
1001:
985:
978:
975:
972:is called a "
953:
944:
926:
917:
894:
885:
859:
850:
846:
841:
832:
824:
815:
809:
800:
793:
784:
776:
759:
755:
747:
726:
718:
703:
700:
696:
692:
689:
686:
678:
662:
655:
640:
632:
616:
613:
605:
589:
582:
567:
560:
545:
538:
537:
536:
516:
511:
507:
501:
497:
491:
487:
479:
475:
471:
457:
453:
445:
440:
437:
433:
427:
423:
416:
411:
407:
378:
374:
371:
368:
361:
357:
353:
350:
343:
342:
341:
327:
320:
315:
313:
309:
305:
295:
293:
292:liquid helium
287:
285:
281:
277:
273:
269:
265:
264:photovoltaics
260:
251:
249:
244:
240:
234:
232:
228:
224:
220:
216:
212:
208:
204:
200:
196:
192:
188:
182:
169:
168:tight binding
165:
161:
157:
153:
149:
144:
137:
132:
127:
120:
116:
111:
106:
102:
100:
96:
92:
88:
84:
80:
75:
73:
69:
65:
61:
57:
53:
49:
45:
41:
40:semiconductor
37:
33:
19:
1760:
1716:
1708:
1704:
1670:
1469:
1466:
1378:
1157:
1145:
974:reduced mass
534:
316:
301:
288:
261:
257:
235:
227:laser diodes
184:
76:
72:valence band
56:valence band
47:
43:
29:
243:dislocation
129:Energy vs.
108:Energy vs.
1794:Categories
1696:References
1673:semimetals
179:See also:
1733:∗
1690:Tauc plot
1648:≈
1612:α
1578:α
1557:ν
1528:α
1502:α
1481:ν
1336:−
1330:
1324:−
1296:−
1283:−
1280:ν
1259:−
1228:
1187:−
1184:ν
1172:∝
1169:α
1153:forbidden
1095:ν
1069:λ
1037:ϵ
927:∗
895:∗
860:∗
842:∗
825:∗
810:∗
760:∗
704:π
687:ℏ
663:ℏ
641:ν
617:ν
568:ν
546:α
508:ℏ
498:ϵ
488:λ
412:∗
375:−
372:ν
362:∗
354:≈
351:α
328:α
187:electrons
1780:Archived
1679:See also
941:are the
877:, where
70:and the
36:band gap
1569:versus
1493:versus
1460:is the
1435:is the
1379:where:
1149:exciton
1055:is the
998:is the
675:is the
602:is the
535:where:
399:, with
310:of the
268:microns
231:silicon
199:photons
195:phonons
1411:phonon
631:photon
215:phonon
119:phonon
115:photon
46:or an
34:, the
306:. By
191:holes
146:Bulk
38:of a
909:and
284:CZTS
280:CIGS
276:CdTe
225:and
205:and
164:InAs
162:and
160:GaAs
150:for
99:AlSb
93:and
87:GaAs
85:and
83:InAs
1663:).
1327:exp
1225:exp
282:or
30:In
1796::
1752:^
1675:.
976:")
278:,
233:.
197:,
193:,
189:,
158:,
156:Ge
154:,
152:Si
101:.
95:Ge
1729:A
1651:0
1643:p
1639:E
1618:0
1615:=
1590:2
1586:/
1582:1
1554:h
1534:0
1531:=
1506:2
1478:h
1448:T
1423:k
1395:p
1391:E
1361:)
1355:T
1352:k
1346:p
1342:E
1333:(
1321:1
1314:2
1310:)
1304:p
1300:E
1291:g
1287:E
1277:h
1274:(
1268:+
1262:1
1256:)
1250:T
1247:k
1241:p
1237:E
1231:(
1218:2
1214:)
1208:p
1204:E
1200:+
1195:g
1191:E
1181:h
1178:(
1142:.
1124:c
1121:v
1117:x
1073:0
1041:0
1011:n
986:q
958:r
954:m
922:h
918:m
890:e
886:m
855:e
851:m
847:+
837:h
833:m
820:e
816:m
805:h
801:m
794:=
789:r
785:m
756:A
731:g
727:E
716:)
701:2
697:/
693:h
690:=
679:(
653:)
614:h
606:(
590:h
517:n
512:3
502:0
492:0
480:2
476:/
472:3
468:)
462:r
458:m
454:2
451:(
446:2
441:c
438:v
434:x
428:2
424:q
417:=
408:A
383:g
379:E
369:h
358:A
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.