249:. Isotope exchange studies indicate that for aryl fluorides and, sometimes, aryl chlorides, the elimination event proceeds in two steps, deprotonation, followed by expulsion of the nucleophile. Thus, the process is formally analogous to the E1cb mechanism of aliphatic compounds. Aryl bromides and iodides, on the other hand, generally appear to undergo elimination by a concerted syn-coplanar E2 mechanism. The resulting benzyne forms addition products, usually by nucleophilic addition and protonation. Generation of the benzyne intermediate is the slow step in the reaction.
226:
263:
78:
567:
89:
253:
649:
442:
528:
638:
460:
372:
556:
297:
104:
179:
146:
165:
346:
628:
193:
503:
398:
384:
542:
423:
408:
2432:
304:
Meta substituent can afford both regioisomers as described above. Nucleophilic additions can occur with regioselectivity. Although classic explanations to explain regioselectivity refer to carbanion stability following attack by the nucleophile, this explanation has been replaced by the aryne distortion model by
644:
Multicomponent reactions of arynes are powerful transformations that allow for rapid formation of 1,2-disubstituted arenes. Despite their potential utility, examples of multicomponent aryne reactions in natural product synthesis are scarce. A four-component aryne coupling reaction was employed in the
303:
There are two possible regioisomers of benzyne with substituent (Y): triple bond can be positioned between C2 and C3 or between C3 and C4. Substituents ortho to the leaving group will lead to the triple bond between C2 and C3. Para Y and LG will lead to regioisomer with triple bond between C3 and C4.
84:
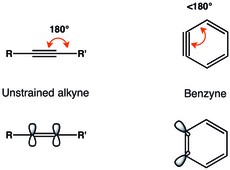
Geometric constraints on the triple bond in benzyne result in diminished overlap of in-plane p-orbitals, and thus weaker triple bond. The vibrational frequency of the triple bond in benzyne was assigned by
Radziszewski to be 1846 cm, indicating a weaker triple bond than in unstrained alkyne with
623:
A variety of natural products have been prepared using arynes as intermediates. Nucleophilic additions to arynes have been widely used in natural product total synthesis. Indeed, nucleophilic additions of arynes are some of the oldest known applications of aryne chemistry. Nucleophilic addition to
509:
Two states were proposed for 1,4-didehydrobenzene: singlet and triplet, with the singlet state lower in energy. Triplet state represents two noninteracting radical centers, and hence should abstract hydrogens at the same rate as phenyl radical. However, singlet state is more stabilized than the
379:
cycloadditions of arynes have been commonly applied to natural product total synthesis. The main limitation of such approach, however, is the need to use constrained dienes, such as furan and cyclopentadiene. In 2009 Buszek and co-workers synthesized herbindole A using aryne -cycloaddition.
393:
Due to significant byproduct formation, aryne chemistry is rarely utilized in natural product total synthesis. Nevertheless, several examples do exist. In 1982, Stevens and co-workers reported a synthesis of taxodione that utilized cycloaddition between an aryne and a ketene acetal.
99:
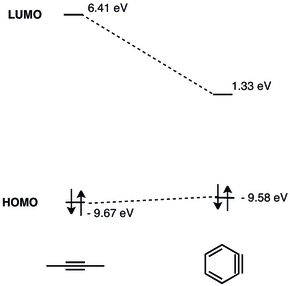
of aryne lies much lower than the LUMO of unstrained alkynes, which makes it a better energy match for the HOMO of nucleophiles. Hence, benzyne possesses electrophilic character and undergoes reactions with nucleophiles. A detailed MO analysis of benzyne was presented in 1968.
523:
The first evidence for arynes came from the work of
Stoermer and Kahlert. In 1902 they observed that upon treatment of 3-bromobenzofuran with base in ethanol 2-ethoxybenzofuran is formed. Based on this observation they postulated an aryne intermediate.
280:
When leaving group (LG) and substituent (Y) are mutually ortho or para, only one benzyne intermediate is possible. However, when LG is meta to Y, then regiochemical outcomes (A and B) are possible. If Y is electron withdrawing, then
152:
Such reactions require strong base and high temperatures. 1,2-Disubstituted arenes serve as precursors to benzynes under milder conditions. Benzyne is generated by the dehalogenation of 1-bromo-2-fluorobenzene by magnesium.
514:
for hydrogen cleavage, leading to slower hydrogen abstraction. Chen proposed the use of 1,4-didehydrobenzene analogues that have large singlet-triplet energy gaps to enhance selectivity of enediyne drug candidates.
312:. In this model, substituents cause geometric distortion of the ground state structure of the aryne, leading to regioselective reactions, consistent with reactions proceeding through early transition states.
342:
results. In this method, the concerted mechanism of the Diels-Alder reaction between benzyne and furan is shown below. Other benzyne cycloadditions are thought to proceed via a stepwise mechanism.
85:
vibrational frequency of approximately 2150 cm. Nevertheless, benzyne is more like a strained alkyne than a diradical, as seen from the large singlet–triplet gap and alkyne-like reactivity.
237:
Even at low temperatures arynes are extremely reactive. Their reactivity can be classified in three main classes: (1) nucleophilic additions, (2) pericyclic reactions, and (3) bond-insertion.
1794:
Polishchuk, A. L.; Bartlett, K. L.; Friedman, L. A.; Jones, M. Jr (2004). "A p-Benzyne to m-Benzyne
Conversion Through a 1,2-Shift of a Phenyl Group. Completion of the Benzyne Cascade".
634:
The synthesis of the tetracyclic meroterpenoid (+)-liphagal involved an aryne intermediate. Their approach employed an aryne cyclization to close the final ring of the natural product.
259:"Aryne coupling" reactions allow for generation of biphenyl compounds which are valuable in pharmaceutical industry, agriculture and as ligands in many metal-catalyzed transformations.
1824:
Richard R. Jones; Robert G. Bergman (1972). "p-Benzyne. Generation as an intermediate in a thermal isomerization reaction and trapping evidence for the 1,4-benzenediyl structure".
438:
are, respectively, 106, 122, and 138 kcal/mol (444, 510 and 577 kJ/mol). The 1,2- and 1,3- isomers have singlet ground states, whereas for 1,4-didehydrobenzene the gap is smaller.
1852:
Clauberg, H.; Minsek, D. W.; Chen, P. (1992). "Mass and photoelectron spectroscopy of C3H2. .DELTA.Hf of singlet carbenes deviate from additivity by their singlet-triplet gaps".
390:
Benzynes undergo cycloaddition with a wide range of alkenes. Due to electrophilic nature of benzyne, alkenes bearing electron-donating substituents work best for this reaction.
1879:
Blush, J. A.; Clauberg, H.; Kohn, D. W.; Minsek, D. W.; Zhang, X.; Chen, P. (1992). "Photoionization mass and photoelectron spectroscopy of radicals, carbenes, and biradicals".
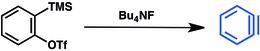
175:. This method has seen wide applicability and was reviewed in 2021. Fluoride displacement of the trimethylsilyl group induces elimination of triflate and release of benzyne:
1970:
Wittig, G.; Pieper, G.; Fuhrmann, G. (1940). "Über die
Bildung von Diphenyl aus Fluorbenzol und Phenyl-lithium (IV. Mitteil. über Austauschreaktionen mit Phenyl-lithium)".
937:
Hoffmann, R.; Imamura, A.; Hehre, W. J. (1968). "Benzynes, dehydroconjugated molecules, and the interaction of orbitals separated by a number of intervening sigma bonds".
448:
The interconversion of the 1,2-, 1,3- and 1,4-didehydrobenzenes has been studied. A 1,2- to 1,3-didehydrobenzene conversion has been postulated to occur in the
58:. Arynes are examples of didehydroarenes (1,2-didehydroarenes in this case), although 1,3- and 1,4-didehydroarenes are also known. Arynes are examples of
584:
1,3-Didehydroarenes was first demonstrated in the 1990s when it was generated from 1,3-disubstituted benzene derivatives, such as the peroxy ester 1,3-C
320:
Benzyne undergoes rapid dimerization to form biphenylene. Some routes to benzyne lead to especially rapid and high yield of this subsequent reaction.
796:
723:
694:
1307:
538:
et al. invoked zwitterionic intermediate in the reaction of fluorobenzene and phenyllithium to give biphenyl. This hypothesis was later confirmed.
2393:
Soorukram, D.; Qu, T.; Barrett, A. G. M. (2008). "Four-Component
Benzyne Coupling Reactions: A Concise Total Synthesis of Dehydroaltenuene B".
1356:
Mizukoshi, Yoshihide; Mikami, Koichiro; Uchiyama, Masanobu (2015). "Aryne
Polymerization Enabling Straightforward Synthesis of Elusive Poly(
552:
et al. showed that the reaction of chlorobenzene-1-C and potassium amide gave equal amounts of aniline with C incorporation at C-1 and C-2.
2296:
Galm, U; Hager, MH; Van Lanen, SG; Ju, J; Thorson, JS; Shen, B (Feb 2005). "Antitumor antibiotics: bleomycin, enediynes, and mitomycin".
574:
Additional evidence for the existence of benzyne came from spectroscopic studies. Benzyne has been observed in a "molecular container".
566:
555:
434:
If benzyne is 1,2-didehydrobenzene, two further isomers are possible: 1,3-didehydrobenzene and 1,4-didehydrobenzene. Their energies
74:
The alkyne representation of benzyne is the most widely encountered. Arynes are usually described as having a strained triple bond.
1040:
Logullo, Francis M.; Seitz, Arnold M.; Friedman, Lester (1968). "Benzenediazonium-2-Carboxylate and
Biphenylene (Benzenediazonium,
1626:"Use of 1,2,4,5-Tetrabromobenzene as a 1,4-Nenzadiyne Equivalent: Anti- and Syn-1,4,5,8-tetrahydroanthracene 1,4:5,8-diepoxides".
1759:
Blake, M. E.; Bartlett, K. L.; Jones, M. Jr (2003). "A m-Benzyne to o-Benzyne
Conversion Through a 1,2-Shift of a Phenyl Group".
1452:
Im, G-Yoon J.; Bronner, Sarah M.; Goetz, Adam E.; Paton, Robert S.; Cheong, Paul H.-Y.; Houk, K. N.; Garg, Neil K. (2010-12-22).
1454:"Indolyne Experimental and Computational Studies: Synthetic Applications and Origins of Selectivities of Nucleophilic Additions"
404:
Mori and co-workers performed a palladium-catalyzed -cocyclization of aryne and diyne in their total synthesis of taiwanins C.
2253:
On-surface; Pérez, E.Guitián; Peña, L.Gross (2015). "On-surface generation and imaging of arynes by atomic force microscopy".
2166:
289:
resulting in regioisomer B being generated. Analogously, if Y is electron donating, regioisomer A is generated, since now H
1283:
897:
Radziszewski, J. G.; Hess, B. A. Jr.; Zahradnik, R. (1992). "Infrared
Spectrum of o-Benzyne: Experiment and Theory".
17:
1655:"Concise Total Synthesis of (±)-cis-Trikentrin A and (±)-Herbindole A via Intermolecular Indole Aryne Cycloaddition"
670:
456:
substituted aryne precursors as shown below. Extremely high temperatures are required for benzyne interconversion.
186:
157:
can be converted to 2-diazoniobenzene-1-carboxylate by diazotization and neutralization. Although explosive, this
2452:
1395:
Cheong, Paul H.-Y.; Paton, Robert S.; Bronner, Sarah M.; Im, G-Yoon J.; Garg, Neil K.; Houk, K. N. (2010-02-03).
994:
Sullivan, John M. (1971-06-01). "Explosion during preparation of benzenediazonium-2-carboxylate hydrochloride".
2462:
353:
1084:
Tadross, P. M.; Stoltz, B. M. (2012). "A Comprehensive
History of Arynes in Natural Product Total Synthesis".
578:
499:
is formed can only be explained by a presence of a cyclic and symmetrical intermediate–1,4-didehydrobenzene.
211:
1997:
Wittig, Georg (1942). "Phenyl-lithium, der Schlüssel zu einer neuen Chemie metallorganischer Verbindungen".
527:
245:
Upon treatment with basic nucleophiles, aryl halides deprotonate alpha to the leaving group, resulting in
270:
371:
2078:
Roberts, John D. (1953). "Rearrangement in the Reaction of Chlorobenzene-1-C14With Potassium Amide1".
1511:"The Role of Aryne Distortions, Steric Effects, and Charges in Regioselectivities of Aryne Reactions"
357:
277:
as the initiator to add to the first aryne yielded polymers containing up to about 100 arene units.
1222:(1969). "Reactive intermediates. Part I. Synthesis and oxidation of 1- and 2-aminobenzotriazole".
380:
6,7-indolyne undergoes cycloaddition with cyclopentadiene to afford complex tetracyclic product.
2436:
43:
2344:
Day, J. J.; McFadden, R. M.; Virgil, S. C.; Kolding, H.; Alleva, J. L.; Stoltz, B. M. (2011).
1275:
1268:
2457:
862:
Gampe, C. M.; Carreira, E. M. (2012). "Arynes and Cyclohexyne in Natural Product Synthesis".
762:
Hans Henning Wenk; Michael Winkler; Wolfram Sander (2003). "One Century of Aryne Chemistry".
218:
generates an intermediate which can be oxidised to benzyne in almost quantitative yield with
120:
2262:
2049:
2006:
1171:
1003:
419:
The first example of aryne σ-bond insertion reaction is the synthesis of melleine in 1973.
332:
2073:
2071:
225:
8:
666:
662:
608:
548:
In 1953 C labeling experiments provided strong support for the intermediacy of benzyne.
246:
135:
2266:
2053:
2010:
1175:
1007:
510:
triplet, and therefore some of the stabilizing energy will be lost in order to form the
2370:
2345:
2068:
2022:
1679:
1654:
1543:
1486:
1429:
1301:
1192:
1159:
1120:
1274:. Richardson, Kathleen Schueller (3rd ed.). New York: Harper & Row. pp.
624:
aryne was used in the attempted synthesis of cryptaustoline (1) and cryptowoline (2).
607:
Breakthroughs on 1,4-didehydrobenzene came in the 1960s, followed from studies on the
2410:
2375:
2313:
2278:
2235:
2162:
1826:
1776:
1684:
1572:
1548:
1530:
1491:
1473:
1434:
1416:
1377:
1289:
1279:
1197:
1140:
1101:
1019:
879:
779:
274:
124:
31:
2132:
Roberts, John D. (1956). "Orientation in Aminations of Substituted Halobenzenes 1".
2026:
806:
733:
704:
2402:
2365:
2357:
2305:
2270:
2227:
2187:
2141:
2114:
2087:
2057:
2014:
1979:
1952:
1919:
1888:
1861:
1834:
1803:
1768:
1674:
1666:
1635:
1608:
1538:
1522:
1481:
1465:
1424:
1408:
1369:
1233:
1187:
1179:
1132:
1093:
1053:
1011:
976:
946:
906:
871:
810:
801:
771:
737:
728:
708:
699:
511:
219:
154:
47:
611:. This theme became topical with the discovery of enediyne "cytostatics", such as
1224:
549:
1567:
1255:. Pasadena, CA: California Institute of Technology (Ph.D. Thesis). pp. 4–5.
1136:
2161:. Edited by Lutz Ackermann 2009 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
2040:
Wittig, G (1954). "Fortschritte auf dem Gebiet der organischen Aniono-Chemie".
896:
361:
761:
563:
Wittig and Pohmer found that benzyne participate in cycloaddition reactions.
2446:
2061:
1983:
1956:
1639:
1612:
1534:
1510:
1477:
1453:
1420:
1396:
1144:
1057:
1023:
980:
805:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
732:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
703:, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
612:
321:
203:
96:
51:
1293:
814:
741:
712:
2414:
2379:
2361:
2317:
2282:
2239:
1924:
1907:
1780:
1688:
1552:
1509:
Medina, Jose M.; Mackey, Joel L.; Garg, Neil K.; Houk, K. N. (2014-11-05).
1495:
1438:
1381:
1219:
1201:
1105:
883:
875:
783:
775:
535:
325:
305:
262:
139:
2218:
Warmuth, R.; Yoon (2001). "Recent highlights in hemicarcerand chemistry".
2105:
Roberts, John D. (1956). "The Mechanism of Aminations of Halobenzenes 1".
296:
77:
1237:
1158:
Hoye, T. R.; Baire, B.; Niu, D.; Willoughby, P. H.; Woods, B. P. (2012).
128:
63:
55:
2191:
2145:
2118:
2091:
1892:
1865:
1838:
1253:
The Elimination-Addition Mechanism of Nucleophilic Aromatic Substitution
1183:
1015:
950:
910:
471:
In classical 1,4-didehydrobenzene experiments, heating to 300 °C, -
88:
2274:
2018:
441:
339:
335:
158:
2406:
2309:
2231:
1772:
1670:
1526:
1469:
1412:
1373:
1097:
927:. Patai, S.; Rappaport, Z. Eds., John Wiley & Sons, New York, 1983
345:
252:
827:
648:
449:
435:
309:
2392:
1940:
1807:
27:
Organic compound made by removing substituents from an aromatic ring
1397:"Indolyne and Aryne Distortions and Nucleophilic Regioselectivites"
676:
172:
1990:
637:
459:
269:
The metal–arene product can also add to another aryne, leading to
1823:
1121:"o -Silylaryl Triflates: A Journey of Kobayashi Aryne Precursors"
2346:"The catalytic enantioselective total synthesis of (+)-liphagal"
103:
2431:
1972:
Berichte der Deutschen Chemischen Gesellschaft (A and B Series)
1793:
453:
178:
164:
145:
59:
839:-benzyne" as erroneous terms for 1,3- and 1,4-didehydrobenzene
161:
species is a convenient and inexpensive precursor to benzyne.
365:
2178:
Heaney, H. (1962). "The Benzyne and Related Intermediates".
627:
1083:
861:
559:
C labeling experiment shows equal distribution of products.
192:
618:
502:
397:
115:
Due to their extreme reactivity, arynes must be generated
570:
Capture of benzyne as dienophile in Diels-Alder reaction.
541:
383:
334:
cyclization reactions. When generated in the presence of
2343:
925:
The Chemistry of Triple Bonded Functional Groups, Part 1
487:. The simultaneous migration of deuterium atoms to form
422:
1908:"Design of Diradical-based Hydrogen Abstraction Agents"
1878:
1355:
407:
189:(HDDA) involves cycloaddition of 1,3-diyne and alkyne.
1157:
414:
2295:
1969:
1508:
1394:
1339:
Diemer, V.; Begaut, M.; Leroux, F. R.; Colobert, F.
1039:
936:
2252:
1451:
1119:Shi, Jiarong; Li, Lianggui; Li, Yang (2021-04-14).
240:
1851:
1758:
1267:
1217:
315:
2444:
1938:
1213:
1211:
577:In 2015, a single aryne molecule was imaged by
1945:Berichte der Deutschen Chemischen Gesellschaft
1652:
171:Another method is based on trimethylsilylaryl
1319:
1317:
1565:
1306:: CS1 maint: multiple names: authors list (
1208:
2217:
1754:
1752:
1750:
1035:
1033:
1653:Buszek, K. R.; Brown, N.; Kuo, D. (2009).
1314:
1079:
1077:
1075:
1073:
1071:
1069:
1067:
615:, which generates a 1,4-didehydrobenzene.
2369:
1923:
1678:
1542:
1485:
1428:
1270:Mechanism and theory in organic chemistry
1191:
962:
960:
757:
755:
753:
751:
749:
2134:Journal of the American Chemical Society
2107:Journal of the American Chemical Society
2080:Journal of the American Chemical Society
1747:
1515:Journal of the American Chemical Society
1458:Journal of the American Chemical Society
1401:Journal of the American Chemical Society
1030:
993:
429:
123:, benzyne must be trapped, otherwise it
2131:
2104:
2077:
1819:
1817:
1118:
1064:
852:, University Science Books, 2006, p612.
619:Examples of benzynes in total synthesis
110:
14:
2445:
2177:
2039:
1996:
1160:"The hexadehydro-Diels–Alder reaction"
966:
957:
746:
661:More examples use of aryne chemistry:
466:
352:A classic example is the synthesis of
232:
1250:
1905:
1814:
1044:-carboxy-, hydroxide, inner salt)".
967:Wittig, Georg (1959). "Triptycene".
69:
24:
1939:Stoermer, R.; Kahlert, B. (1902).
1599:"1,2,3,4-Tetraphenylnaphthalene".
1566:Heaney, H.; Millar, I. T. (1960).
1325:Modern Physical Organic Chemistry.
1265:
802:Compendium of Chemical Terminology
729:Compendium of Chemical Terminology
700:Compendium of Chemical Terminology
647:
636:
626:
565:
554:
540:
526:
501:
458:
440:
421:
415:Bond-insertion reactions of arynes
406:
396:
382:
370:
344:
295:
261:
251:
224:
191:
177:
163:
144:
102:
87:
76:
25:
2474:
2424:
1941:"Ueber das 1- und 2-Brom-cumaron"
1587:, vol. 5, 1973, p. 1120
850:Modern Physical Organic Chemistry
848:Anslyn, E. V.; Dougherty, D. A.:
831:entry for "benzynes" identifies "
645:synthesis of dehydroaltenuene B.
134:Early routes to benzyne involved
2430:
1714:Stevens, R. V.; Bisacchi, G. S.
1323:Anslyn, E. V.; Dougherty, D. A.
671:Transition metal benzyne complex
479:, but does not equilibrate with
241:Nucleophilic additions to arynes
187:hexadehydro Diels-Alder reaction
2386:
2337:
2330:Kametani, T.; Ogasawara, K. J.
2324:
2289:
2246:
2211:
2198:
2171:
2152:
2125:
2098:
2033:
1963:
1932:
1899:
1872:
1845:
1787:
1734:
1721:
1708:
1695:
1646:
1619:
1592:
1559:
1502:
1445:
1388:
1349:
1333:
1259:
1244:
1151:
1112:
987:
930:
923:Gilchrist, T. L. Supplement C:
368:to form a tetrahydroanthracene
1727:Sato, Y.; Tamura,T.; Mori, M.
917:
890:
855:
842:
819:
790:
717:
688:
354:1,2,3,4-tetraphenylnaphthalene
316:Pericyclic reactions of arynes
13:
1:
1701:Pellissier, H.; Santelli, M.
996:Journal of Chemical Education
682:
531:First indication of benzyne.
491:, and the fact that none of
7:
1137:10.1021/acs.chemrev.0c01011
655:
475:readily equilibrates with -
375:diaryne reaction with furan
293:is the more acidic proton.
271:chain-growth polymerization
10:
2479:
1912:Angew. Chem. Int. Ed. Engl
1327:University Science Books,
1266:H., Lowry, Thomas (1987).
864:Angew. Chem. Int. Ed. Engl
518:
2159:Modern Arylation Methods
2062:10.1002/ange.19540660103
1984:10.1002/cber.19400731113
1957:10.1002/cber.19020350286
1640:10.15227/orgsyn.075.0201
1613:10.15227/orgsyn.046.0107
1058:10.15227/orgsyn.048.0012
981:10.15227/orgsyn.039.0075
675:The pyridine equivalent
2204:Wittig, G.; Pohmer, L.
1999:Die Naturwissenschaften
815:10.1351/goldbook.D01574
742:10.1351/goldbook.B00634
713:10.1351/goldbook.A00465
2453:Reactive intermediates
2362:10.1002/anie.201101842
1925:10.1002/anie.199614781
1251:Panar, Manuel (1961).
876:10.1002/anie.201107485
776:10.1002/anie.200390151
652:
641:
631:
571:
560:
545:
532:
506:
463:
445:
426:
411:
401:
387:
376:
349:
300:
266:
256:
229:
196:
182:
168:
149:
121:reactive intermediates
107:
92:
81:
42:are a class of highly
2463:Aromatic hydrocarbons
1740:Guyot, M.; Molho, D.
1729:Angew. Chem. Int. Ed.
651:
640:
630:
569:
558:
544:
530:
505:
462:
452:(900 °C) of the
444:
430:Other dehydrobenzenes
425:
410:
400:
386:
374:
348:
331:Benzynes can undergo
299:
285:is more acidic than H
265:
255:
228:
195:
181:
167:
148:
106:
91:
80:
2439:at Wikimedia Commons
2350:Angew. Chem. Int. Ed
1238:10.1039/J39690000742
764:Angew. Chem. Int. Ed
111:Generation of arynes
2267:2015NatCh...7..623P
2192:10.1021/cr60216a001
2146:10.1021/ja01584a025
2119:10.1021/ja01584a024
2092:10.1021/ja01109a523
2054:1954AngCh..66...10W
2011:1942NW.....30..696W
1893:10.1021/ar00021a001
1866:10.1021/ja00027a014
1839:10.1021/ja00757a071
1521:(44): 15798–15805.
1464:(50): 17933–17944.
1184:10.1038/nature11518
1176:2012Natur.490..208H
1016:10.1021/ed048p419.3
1008:1971JChEd..48..419S
951:10.1021/ja01008a018
911:10.1021/ja00027a007
667:in-methylcyclophane
663:tricyclobutabenzene
609:Bergman cyclization
467:1,4-Didehydroarenes
247:dehydrohalogenation
233:Reactions of arynes
136:dehydrohalogenation
119:. Typical of other
2275:10.1038/nchem.2300
2208:1955; 67(13), 348.
2019:10.1007/BF01489519
2005:(46–47): 696–703.
1796:J. Phys. Org. Chem
1341:Eur. J. Org. Chem.
653:
642:
632:
572:
561:
546:
533:
507:
464:
446:
427:
412:
402:
388:
377:
350:
301:
267:
257:
230:
197:
183:
169:
150:
108:
93:
82:
54:by removal of two
2435:Media related to
2407:10.1021/ol8015435
2401:(17): 3833–3835.
2310:10.1021/cr030117g
2232:10.1021/ar980082k
2167:978-3-527-31937-4
2086:(13): 3290–3291.
2042:Angewandte Chemie
1978:(11): 1193–1197.
1827:J. Am. Chem. Soc.
1773:10.1021/ja0213672
1742:Tetrahedron Lett.
1671:10.1021/ol802425m
1628:Organic Syntheses
1601:Organic Syntheses
1585:Collected Volumes
1573:Organic Syntheses
1527:10.1021/ja5099935
1470:10.1021/ja1086485
1413:10.1021/ja9098643
1374:10.1021/ja5112207
1170:(7419): 208–212.
1098:10.1021/cr200478h
358:Tetrabromobenzene
275:copper(I) cyanide
70:Bonding in arynes
32:organic chemistry
18:Benzyne mechanism
16:(Redirected from
2470:
2434:
2419:
2418:
2390:
2384:
2383:
2373:
2341:
2335:
2332:J. Chem. Soc., C
2328:
2322:
2321:
2298:Chemical Reviews
2293:
2287:
2286:
2255:Nature Chemistry
2250:
2244:
2243:
2215:
2209:
2202:
2196:
2195:
2180:Chemical Reviews
2175:
2169:
2156:
2150:
2149:
2129:
2123:
2122:
2102:
2096:
2095:
2075:
2066:
2065:
2037:
2031:
2030:
1994:
1988:
1987:
1967:
1961:
1960:
1951:(2): 1633–1640.
1936:
1930:
1929:
1927:
1906:Chen, P (1996).
1903:
1897:
1896:
1876:
1870:
1869:
1854:J. Am. Chem. Soc
1849:
1843:
1842:
1821:
1812:
1811:
1791:
1785:
1784:
1761:J. Am. Chem. Soc
1756:
1745:
1738:
1732:
1725:
1719:
1712:
1706:
1699:
1693:
1692:
1682:
1650:
1644:
1643:
1623:
1617:
1616:
1596:
1590:
1588:
1581:
1563:
1557:
1556:
1546:
1506:
1500:
1499:
1489:
1449:
1443:
1442:
1432:
1407:(4): 1267–1269.
1392:
1386:
1385:
1362:J. Am. Chem. Soc
1353:
1347:
1337:
1331:
1321:
1312:
1311:
1305:
1297:
1273:
1263:
1257:
1256:
1248:
1242:
1241:
1218:Campbell, C.D.;
1215:
1206:
1205:
1195:
1155:
1149:
1148:
1131:(7): 3892–4044.
1125:Chemical Reviews
1116:
1110:
1109:
1092:(6): 3550–3577.
1081:
1062:
1061:
1037:
1028:
1027:
991:
985:
984:
964:
955:
954:
939:J. Am. Chem. Soc
934:
928:
921:
915:
914:
899:J. Am. Chem. Soc
894:
888:
887:
859:
853:
846:
840:
823:
817:
794:
788:
787:
759:
744:
721:
715:
692:
512:transition state
220:lead(IV) acetate
155:Anthranilic acid
50:derived from an
21:
2478:
2477:
2473:
2472:
2471:
2469:
2468:
2467:
2443:
2442:
2427:
2422:
2391:
2387:
2342:
2338:
2329:
2325:
2294:
2290:
2251:
2247:
2216:
2212:
2203:
2199:
2176:
2172:
2157:
2153:
2130:
2126:
2103:
2099:
2076:
2069:
2038:
2034:
1995:
1991:
1968:
1964:
1937:
1933:
1904:
1900:
1877:
1873:
1850:
1846:
1822:
1815:
1808:10.1002/poc.797
1792:
1788:
1767:(21): 6485–90.
1757:
1748:
1739:
1735:
1726:
1722:
1713:
1709:
1700:
1696:
1651:
1647:
1625:
1624:
1620:
1598:
1597:
1593:
1583:
1564:
1560:
1507:
1503:
1450:
1446:
1393:
1389:
1354:
1350:
1338:
1334:
1322:
1315:
1299:
1298:
1286:
1264:
1260:
1249:
1245:
1225:J. Chem. Soc. C
1216:
1209:
1156:
1152:
1117:
1113:
1082:
1065:
1038:
1031:
992:
988:
965:
958:
935:
931:
922:
918:
895:
891:
870:(16): 3766–78.
860:
856:
847:
843:
835:-benzyne" and "
824:
820:
795:
791:
760:
747:
722:
718:
693:
689:
685:
658:
621:
603:
599:
595:
591:
587:
550:John D. Roberts
521:
469:
432:
417:
360:can react with
318:
292:
288:
284:
243:
235:
113:
72:
28:
23:
22:
15:
12:
11:
5:
2476:
2466:
2465:
2460:
2455:
2441:
2440:
2426:
2425:External links
2423:
2421:
2420:
2385:
2356:(30): 6814–8.
2336:
2323:
2288:
2245:
2220:Acc. Chem. Res
2210:
2197:
2170:
2151:
2140:(3): 611–614.
2124:
2113:(3): 601–611.
2097:
2067:
2032:
1989:
1962:
1931:
1918:(1314): 1478.
1898:
1881:Acc. Chem. Res
1871:
1844:
1833:(2): 660–661.
1813:
1802:(9): 798–806.
1786:
1746:
1744:1973; 14, 3433
1733:
1731:2004; 43, 2436
1720:
1718:1982; 47, 2396
1707:
1694:
1665:(1): 201–204.
1645:
1618:
1591:
1568:"Triphenylene"
1558:
1501:
1444:
1387:
1348:
1332:
1313:
1284:
1258:
1243:
1232:(5): 742–747.
1207:
1150:
1111:
1063:
1029:
986:
956:
929:
916:
889:
854:
841:
818:
789:
770:(5): 502–528.
745:
716:
686:
684:
681:
680:
679:
673:
657:
654:
620:
617:
601:
597:
593:
589:
585:
520:
517:
468:
465:
431:
428:
416:
413:
317:
314:
290:
286:
282:
242:
239:
234:
231:
216:-sulfonic acid
212:hydroxylamine-
208:-benzotriazole
202:-amination of
112:
109:
71:
68:
26:
9:
6:
4:
3:
2:
2475:
2464:
2461:
2459:
2456:
2454:
2451:
2450:
2448:
2438:
2433:
2429:
2428:
2416:
2412:
2408:
2404:
2400:
2396:
2389:
2381:
2377:
2372:
2367:
2363:
2359:
2355:
2351:
2347:
2340:
2333:
2327:
2319:
2315:
2311:
2307:
2304:(2): 739–58.
2303:
2299:
2292:
2284:
2280:
2276:
2272:
2268:
2264:
2260:
2256:
2249:
2241:
2237:
2233:
2229:
2225:
2221:
2214:
2207:
2201:
2193:
2189:
2185:
2181:
2174:
2168:
2164:
2160:
2155:
2147:
2143:
2139:
2135:
2128:
2120:
2116:
2112:
2108:
2101:
2093:
2089:
2085:
2081:
2074:
2072:
2063:
2059:
2055:
2051:
2047:
2043:
2036:
2028:
2024:
2020:
2016:
2012:
2008:
2004:
2000:
1993:
1985:
1981:
1977:
1973:
1966:
1958:
1954:
1950:
1946:
1942:
1935:
1926:
1921:
1917:
1913:
1909:
1902:
1894:
1890:
1886:
1882:
1875:
1867:
1863:
1859:
1855:
1848:
1840:
1836:
1832:
1829:
1828:
1820:
1818:
1809:
1805:
1801:
1797:
1790:
1782:
1778:
1774:
1770:
1766:
1762:
1755:
1753:
1751:
1743:
1737:
1730:
1724:
1717:
1716:J. Org, Chem.
1711:
1705:2003; 59, 701
1704:
1698:
1690:
1686:
1681:
1676:
1672:
1668:
1664:
1660:
1656:
1649:
1641:
1637:
1634:: 201. 1998.
1633:
1629:
1622:
1614:
1610:
1607:: 107. 1966.
1606:
1602:
1595:
1586:
1579:
1575:
1574:
1569:
1562:
1554:
1550:
1545:
1540:
1536:
1532:
1528:
1524:
1520:
1516:
1512:
1505:
1497:
1493:
1488:
1483:
1479:
1475:
1471:
1467:
1463:
1459:
1455:
1448:
1440:
1436:
1431:
1426:
1422:
1418:
1414:
1410:
1406:
1402:
1398:
1391:
1383:
1379:
1375:
1371:
1367:
1363:
1360:-arylene)s".
1359:
1352:
1345:
1342:
1336:
1330:
1326:
1320:
1318:
1309:
1303:
1295:
1291:
1287:
1285:0-06-044084-8
1281:
1277:
1272:
1271:
1262:
1254:
1247:
1239:
1235:
1231:
1227:
1226:
1221:
1214:
1212:
1203:
1199:
1194:
1189:
1185:
1181:
1177:
1173:
1169:
1165:
1161:
1154:
1146:
1142:
1138:
1134:
1130:
1126:
1122:
1115:
1107:
1103:
1099:
1095:
1091:
1087:
1080:
1078:
1076:
1074:
1072:
1070:
1068:
1059:
1055:
1051:
1047:
1043:
1036:
1034:
1025:
1021:
1017:
1013:
1009:
1005:
1001:
997:
990:
982:
978:
974:
970:
963:
961:
952:
948:
944:
940:
933:
926:
920:
912:
908:
904:
900:
893:
885:
881:
877:
873:
869:
865:
858:
851:
845:
838:
834:
830:
829:
822:
816:
812:
808:
807:Dehydroarenes
804:
803:
798:
793:
785:
781:
777:
773:
769:
765:
758:
756:
754:
752:
750:
743:
739:
735:
731:
730:
725:
720:
714:
710:
706:
702:
701:
696:
691:
687:
678:
674:
672:
668:
664:
660:
659:
650:
646:
639:
635:
629:
625:
616:
614:
613:calicheamicin
610:
605:
582:
580:
575:
568:
564:
557:
553:
551:
543:
539:
537:
529:
525:
516:
513:
504:
500:
498:
494:
490:
486:
482:
478:
474:
461:
457:
455:
451:
443:
439:
437:
424:
420:
409:
405:
399:
395:
391:
385:
381:
373:
369:
367:
363:
359:
355:
347:
343:
341:
337:
333:
329:
327:
323:
322:Trimerization
313:
311:
307:
298:
294:
278:
276:
272:
264:
260:
254:
250:
248:
238:
227:
223:
221:
217:
215:
209:
207:
201:
194:
190:
188:
180:
176:
174:
166:
162:
160:
156:
147:
143:
141:
137:
132:
130:
126:
122:
118:
105:
101:
98:
90:
86:
79:
75:
67:
65:
61:
57:
53:
52:aromatic ring
49:
45:
41:
37:
33:
19:
2458:Cycloalkynes
2398:
2394:
2388:
2353:
2349:
2339:
2331:
2326:
2301:
2297:
2291:
2261:(8): 623–8.
2258:
2254:
2248:
2223:
2219:
2213:
2206:Angew. Chem.
2205:
2200:
2186:(2): 81–97.
2183:
2179:
2173:
2158:
2154:
2137:
2133:
2127:
2110:
2106:
2100:
2083:
2079:
2048:(1): 10–17.
2045:
2041:
2035:
2002:
1998:
1992:
1975:
1971:
1965:
1948:
1944:
1934:
1915:
1911:
1901:
1884:
1880:
1874:
1857:
1853:
1847:
1830:
1825:
1799:
1795:
1789:
1764:
1760:
1741:
1736:
1728:
1723:
1715:
1710:
1703:Tetrahedron,
1702:
1697:
1662:
1658:
1648:
1631:
1627:
1621:
1604:
1600:
1594:
1584:
1577:
1571:
1561:
1518:
1514:
1504:
1461:
1457:
1447:
1404:
1400:
1390:
1368:(1): 74–77.
1365:
1361:
1357:
1351:
1343:
1340:
1335:
1328:
1324:
1269:
1261:
1252:
1246:
1229:
1223:
1167:
1163:
1153:
1128:
1124:
1114:
1089:
1085:
1049:
1045:
1041:
999:
995:
989:
972:
968:
942:
938:
932:
924:
919:
902:
898:
892:
867:
863:
857:
849:
844:
836:
832:
826:
821:
800:
792:
767:
763:
727:
719:
698:
690:
643:
633:
622:
606:
583:
576:
573:
562:
547:
534:
522:
508:
496:
492:
488:
484:
480:
476:
472:
470:
447:
433:
418:
403:
392:
389:
378:
362:butyllithium
351:
330:
326:triphenylene
319:
302:
279:
268:
258:
244:
236:
213:
205:
199:
198:
184:
170:
159:zwitterionic
151:
140:aryl halides
133:
116:
114:
94:
83:
73:
56:substituents
39:
35:
29:
945:(6): 1499.
129:biphenylene
62:under high
2447:Categories
2334:1967, 2208
1887:(9): 385.
1046:Org. Synth
1002:(6): 419.
969:Org. Synth
683:References
340:trypticene
336:anthracene
2395:Org. Lett
2226:(2): 96.
1659:Org. Lett
1535:0002-7863
1478:0002-7863
1421:0002-7863
1302:cite book
1220:C.W. Rees
1145:0009-2665
1086:Chem. Rev
1024:0021-9584
828:Gold Book
450:pyrolysis
436:in silico
173:triflates
125:dimerises
46:chemical
2415:18672878
2380:21671325
2318:15700963
2283:26201737
2240:11263868
2027:37148502
1781:12785789
1689:19055375
1553:25303232
1496:21114321
1439:20058924
1382:25459083
1294:14214254
1202:23060191
1106:22443517
884:22422638
784:12569480
734:Benzynes
677:pyridyne
656:See also
273:. Using
44:reactive
40:benzynes
2371:3361906
2263:Bibcode
2050:Bibcode
2007:Bibcode
1680:2723800
1544:4221504
1487:3075889
1430:2819077
1193:3538845
1172:Bibcode
1004:Bibcode
519:History
117:in situ
60:alkynes
48:species
2437:Arynes
2413:
2378:
2368:
2316:
2281:
2238:
2165:
2025:
1860:: 99.
1779:
1687:
1677:
1551:
1541:
1533:
1494:
1484:
1476:
1437:
1427:
1419:
1380:
1292:
1282:
1200:
1190:
1164:Nature
1143:
1104:
1052:: 12.
1022:
975:: 75.
905:: 52.
882:
825:IUPAC
782:
596:C(O)CH
536:Wittig
454:phenyl
324:gives
64:strain
36:arynes
2023:S2CID
1580:: 105
1358:ortho
1346:, 341
797:IUPAC
724:IUPAC
705:Aryne
695:IUPAC
366:furan
210:with
2411:PMID
2376:PMID
2314:PMID
2279:PMID
2236:PMID
2163:ISBN
1777:PMID
1685:PMID
1549:PMID
1531:ISSN
1492:PMID
1474:ISSN
1435:PMID
1417:ISSN
1378:PMID
1344:2011
1329:2006
1308:link
1290:OCLC
1280:ISBN
1230:1969
1198:PMID
1141:ISSN
1102:PMID
1020:ISSN
880:PMID
780:PMID
364:and
310:Garg
308:and
306:Houk
97:LUMO
95:The
38:and
2403:doi
2366:PMC
2358:doi
2306:doi
2302:105
2271:doi
2228:doi
2188:doi
2142:doi
2115:doi
2088:doi
2058:doi
2015:doi
1980:doi
1953:doi
1920:doi
1889:doi
1862:doi
1858:114
1835:doi
1804:doi
1769:doi
1765:125
1675:PMC
1667:doi
1636:doi
1609:doi
1539:PMC
1523:doi
1519:136
1482:PMC
1466:doi
1462:132
1425:PMC
1409:doi
1405:132
1370:doi
1366:137
1276:643
1234:doi
1188:PMC
1180:doi
1168:490
1133:doi
1129:121
1094:doi
1090:112
1054:doi
1012:doi
977:doi
947:doi
907:doi
903:114
872:doi
811:doi
809:".
772:doi
738:doi
736:".
709:doi
707:".
579:STM
495:or
483:or
138:of
127:to
30:In
2449::
2409:.
2399:10
2397:.
2374:.
2364:.
2354:50
2352:.
2348:.
2312:.
2300:.
2277:.
2269:.
2257:.
2234:.
2224:34
2222:.
2184:62
2182:.
2138:78
2136:.
2111:78
2109:.
2084:75
2082:.
2070:^
2056:.
2046:66
2044:.
2021:.
2013:.
2003:30
2001:.
1976:73
1974:.
1949:35
1947:.
1943:.
1916:35
1914:.
1910:.
1885:25
1883:.
1856:.
1831:94
1816:^
1800:17
1798:.
1775:.
1763:.
1749:^
1683:.
1673:.
1663:11
1661:.
1657:.
1632:75
1630:.
1605:46
1603:.
1582:;
1578:40
1576:.
1570:.
1547:.
1537:.
1529:.
1517:.
1513:.
1490:.
1480:.
1472:.
1460:.
1456:.
1433:.
1423:.
1415:.
1403:.
1399:.
1376:.
1364:.
1316:^
1304:}}
1300:{{
1288:.
1278:.
1228:.
1210:^
1196:.
1186:.
1178:.
1166:.
1162:.
1139:.
1127:.
1123:.
1100:.
1088:.
1066:^
1050:48
1048:.
1032:^
1018:.
1010:.
1000:48
998:.
973:39
971:.
959:^
943:90
941:.
901:.
878:.
868:51
866:.
799:,
778:.
768:42
766:.
748:^
726:,
697:,
669:,
665:,
604:.
592:(O
581:.
356:.
338:,
328:.
222:.
185:A
142::
131:.
66:.
34:,
2417:.
2405::
2382:.
2360::
2320:.
2308::
2285:.
2273::
2265::
2259:7
2242:.
2230::
2194:.
2190::
2148:.
2144::
2121:.
2117::
2094:.
2090::
2064:.
2060::
2052::
2029:.
2017::
2009::
1986:.
1982::
1959:.
1955::
1928:.
1922::
1895:.
1891::
1868:.
1864::
1841:.
1837::
1810:.
1806::
1783:.
1771::
1691:.
1669::
1642:.
1638::
1615:.
1611::
1589:.
1555:.
1525::
1498:.
1468::
1441:.
1411::
1384:.
1372::
1310:)
1296:.
1240:.
1236::
1204:.
1182::
1174::
1147:.
1135::
1108:.
1096::
1060:.
1056::
1042:o
1026:.
1014::
1006::
983:.
979::
953:.
949::
913:.
909::
886:.
874::
837:p
833:m
813::
786:.
774::
740::
711::
602:2
600:)
598:3
594:2
590:4
588:H
586:6
497:D
493:C
489:B
485:D
481:C
477:B
473:A
291:A
287:A
283:B
281:H
214:O
206:H
204:1
200:N
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.