363:
26:
428:
2355:
395:, the critical regulatory subunit of myosin), is the smallest and most mysterious subunit. Currently little is known about M20, except that it is not necessary for catalysis, as removing the subunit does not affect turnover or selectivity. While some believe it could have regulatory function, nothing has been determined yet.
383:
as well as smooth muscle contraction. Because it is so important to basic cellular functions, and because there are far fewer protein phosphatases than kinases in cells, PP1’s structure and function is highly conserved (though the specific isoform used in myosin phosphatase is the δ isoform, PP1δ).
465:
When the regulatory systems of myosin phosphatase begin to fail, there can be major health consequences. Since smooth muscle is found in the respiratory, circulatory, and reproductive systems of humans (as well as other places), if the smooth muscle can no longer relax because of faulty regulation,
441:
The regulatory pathways of MLC kinase have been well-established, but until the late 1980s, it was assumed that myosin phosphatase was not regulated, and contraction/relaxation was entirely dependent on MLC kinase activity. However, since the 1980s, the inhibiting effect of rho-associated protein
321:
and myosin, two proteins that interact together to produce muscle contraction and relaxation. Myosin II, also known as conventional myosin, has two heavy chains that consist of the head and tail domains and four light chains (two per head) that bind to the heavy chains in the “neck” region. When
387:
Surrounding these ions is a Y-shaped cleft with three grooves: a hydrophobic, an acidic, and a C-terminal groove. When PP1 is not bonded to any other subunit, it is not particularly specific. However, when it bonds to the second subunit of myosin phosphatase, MYPT1 (MW ~130 kDa), this catalytic
354:. Without the group, the myosin reverts to its original conformation, in which it cannot interact with the actin and hold the muscle tense, so the muscle relaxes. The muscle will remain in this relaxed position until myosin is phosphorylated by MLC kinase and undergoes a conformational change.
290:
ions as catalysts to dephosphorylate the light-chains on myosin, which causes a conformational change in the myosin and relaxes the muscle. The enzyme is highly conserved and is found in all organisms’ smooth muscle tissue. While it is known that myosin phosphatase is regulated by
346:
and causing the muscle to contract. Because myosin undergoes a conformational change, the muscle will stay contracted even if calcium and activated MLC kinase concentrations are brought to normal levels. The conformational change must be undone to relax the muscle.
457:
One other proposed regulatory strategy involves arachidonic acid. When arachidonic acid is added to tensed muscle tissue, the acid decreases the rate of dephosphorylation (and thus relaxation) of myosin. However, it is unclear how arachidonic acid functions as an
415:
and a free water molecule are stabilized by the hydrogen-bonding residues in the active site, as well as the positively charged ions (which interact strongly with the negative phosphate group). His-125 (on myosin phosphatase) donates a proton to Ser-19
450:, which phosphorylates the MYPT1 at two major inhibitory sites, Thr-696 and Thr-866. This fully demonstrates the value of the MYPT1, not only to increase reaction rate and specificity, but also to greatly slow down the reaction. However, when
431:
The mechanism of PP1 for myosin phosphatase, with critical enzyme residues shown. The substrates and products are bold and in red, and the manganese ions are in blue. The alcohol group shown on myosin after dephosphorylation is the alcohol on
388:
cleft changes configuration. This results in a dramatic increase in myosin specificity. Thus, it is clear that MYPT1 has great regulatory power over PP1 and myosin phosphatase, even without the presence of other activators or inhibitors.
366:
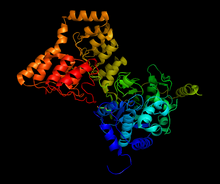
A 3D representation of PP1 (shown in red) and a portion of MYPT1 (shown in blue), with the manganese ion catalysts shown in white. The yellow lines mark the grooves that are critical for enzyme binding and
634:. Authors: Stephen M. Schwartz, Robert P. Mecham. Editors: Stephen M. Schwartz, Robert P. Mecham. Contributors: Stephen M. Schwartz, Robert P. Mecham. Publisher: Academic Press, 1995.
1136:"Purification and characterization of a multisubunit phosphatase from turkey gizzard smooth muscle. The effect of calmodulin binding to myosin light chain kinase on dephosphorylation"
777:
Fujioka, M; Takahashi, N (April 1, 1998). "A New
Isoform of Human Myosin Phosphatase Targeting/Regulatory Subunit (MYPT2): cDNA Cloning, Tissue Expression, and Chromosomal Mapping".
424:. After shuffling protons to stabilize (which happens rapidly compared to the attack on phosphorus), the phosphate and alcohol are formed, and both leave the active site.
462:. Two competing theories are that either arachidonic acid acts as a co-messenger in the rho-kinase cascade mentioned above, or that it binds to the c-terminal of MYPT1.
1836:
411:
binds to both the hydrophobic and acid grooves of PP1 and MYPT1, the regulatory site on myosin phosphatase. Once in the proper configuration, both the phyosphorylated
1782:
191:
990:"Molecular Mechanism of Telokin-Mediated Disinhibition of Myosin Light Chain Phosphatase and cAMP/cGMP-Induced Relaxation of Gastrointestinal Smooth Muscle"
403:
The mechanism of removing the phosphate from Ser-19 is very similar to other dephosphorylation reactions in the cell, such as the activation of
1444:
1605:
210:
1262:
1701:
1412:
1429:
203:
371:
Myosin phosphatase is made of three subunits. The catalytic subunit, PP1, is one of the more important Ser/Thr phosphatases in
1676:
647:
154:
1590:
243:
2380:
1691:
1685:
1486:
1236:
1534:
823:
585:
Hartshorne, DJ; Ito, M (May 1998). "Myosin Light Chain
Phosphatase: Subunit Composition, Interactions and Regulation".
2074:
1645:
1434:
639:
1752:
1640:
1041:"Signal Transduction by G-Proteins, Rho-Kinase and Protein Phosphatase to Smooth Muscle and Non-Muscle Myosin II"
362:
1169:
Kimura K; et al. (1996). "Regulation of Myosin phosphatase by Rho and Rho-associated kinase (Rho-kinase)".
148:
1561:
1471:
1424:
300:
130:
2230:
1983:
1924:
292:
135:
627:
2028:
1595:
1585:
1988:
1880:
1505:
454:
is added, it effectively undoes the effect of Rho-kinase, even though it does not dephosphorylate MYPT1.
215:
342:) at the Ser-19 residue. This phosphorylation causes a conformational change in the myosin, activating
123:
2345:
1574:
1570:
1566:
1482:
1315:
25:
58:
2331:
2318:
2305:
2292:
2279:
2266:
2253:
2215:
1742:
1298:
492:
335:
275:
2225:
2179:
2122:
1490:
1339:
1253:
151:
41:
75:
2127:
1359:
1229:
938:"ROCK Isoform Regulation of Myosin Phosphatase and Contractility in Vascular Smooth Muscle Cells"
1915:
1554:
742:
Cohen, Patricia T. W. (January 15, 2002). "Protein
Phosphatase 1-Targeted in Many Directions".
632:
The vascular smooth muscle cell: molecular and biological responses to the extracellular matrix
331:
270:
and initiates the relaxation process of the muscle cells. Thus, myosin phosphatase undoes the
2148:
2067:
1850:
1747:
1539:
1500:
1364:
1286:
279:
2220:
2033:
1868:
1863:
1796:
1456:
1281:
1178:
1056:
544:
475:
312:
267:
111:
384:
PP1 works by using two manganese ions as catalysts for the dephosphorylation (see below).
8:
2184:
1888:
1858:
1662:
1657:
1581:
1517:
1303:
87:
53:
1182:
889:
548:
46:
2117:
2021:
1873:
1354:
1344:
1222:
1202:
1065:
1040:
1016:
989:
962:
937:
870:
610:
427:
286:(MYPT1), and a third subunit (M20) of unknown function. The catalytic region uses two
271:
1152:
1135:
166:
2375:
1822:
1767:
1735:
1610:
1329:
1194:
1157:
1111:
1070:
1021:
967:
911:
862:
819:
794:
759:
724:
683:
643:
635:
602:
562:
404:
247:
142:
1206:
874:
614:
2163:
2158:
2132:
2060:
1898:
1478:
1383:
1378:
1334:
1186:
1147:
1101:
1060:
1052:
1011:
1001:
957:
949:
901:
852:
786:
751:
714:
675:
594:
552:
459:
296:
1090:"Physiological Pathways and Molecular Mechanisms Regulating Uterine Contractility"
953:
2210:
2194:
2107:
2000:
1814:
1730:
1725:
1720:
1633:
1628:
1388:
1266:
1190:
1214:
170:
2359:
2248:
2189:
1973:
1968:
1963:
1349:
1324:
1320:
1293:
1276:
857:
840:
186:
666:
Webb, R. Clinton (November 2003). "Smooth Muscle
Contraction and Relaxation".
598:
2369:
2153:
2112:
1714:
1623:
1373:
813:
679:
380:
161:
1106:
1089:
1006:
30:
Structure of complex between PP1 and a portion of MYPT1, generated from 1s70
2102:
1841:
1772:
1495:
1416:
1115:
1074:
1025:
971:
915:
866:
790:
763:
728:
719:
702:
687:
566:
471:
1198:
1161:
798:
755:
606:
2326:
2261:
2097:
2016:
1787:
1681:
1544:
1510:
1448:
703:"Structural Basis for Regulation of Protein Phosphatase 1 by Inhibitor-2"
343:
557:
532:
99:
1993:
502:
421:
2300:
2274:
1906:
1705:
1618:
1245:
372:
351:
287:
338:(MLC kinase). MLC kinase phosphorylates the myosin light chain (MLC
2354:
1951:
1946:
1941:
1762:
1397:
1249:
376:
283:
278:. The enzyme is composed of three subunits: the catalytic region (
1958:
1936:
1931:
1549:
936:
Wang, Yuepeng; Riddick, Nadeen; et al. (February 27, 2009).
451:
327:
323:
295:, there is current debate about whether other molecules, such as
118:
906:
531:
Terrak, Mohammed; Kerff, Frederic; et al. (June 17, 2004).
2313:
2083:
1978:
1911:
1804:
1402:
1310:
888:
Lee, Ernest Y.C.; Zhang, Lifang; et al. (March 15, 1999).
487:
467:
412:
251:
239:
198:
94:
82:
70:
2287:
1920:
1671:
1667:
447:
318:
841:"Serine/Threonine Phosphatases: Mechanism through Structure"
379:
metabolism, intracellular transport, protein synthesis, and
1652:
1529:
1522:
1466:
1461:
890:"Phosphorylase Phosphatase: New Horizons for an Old Enzyme"
497:
443:
106:
2052:
334:, where they activate calmodulin, which in turn activates
1039:
Somlyo, Andrew P.; Somlyo, Avril V. (November 10, 1999).
988:
Khromov, ES; Momotani, K.; et al. (April 27, 2012).
442:
kinase has been discovered and thoroughly investigated.
701:
Hurley, Thomas; Yang, Jie; et al. (July 18, 2007).
350:
When myosin phosphatase binds to myosin, it removes the
533:"Structural Basis of Protein Phosphatase 1 Regulation"
2343:
580:
578:
576:
391:
The third subunit, M20 (not to be confused with MLC
526:
524:
522:
520:
518:
1540:Fructose 6-P,2-kinase:fructose 2,6-bisphosphatase
1244:
573:
2367:
776:
1088:Aguilar, Hector; Mitchell, B.F. (May 7, 2010).
1087:
987:
515:
436:
2068:
1230:
1133:
584:
244:serine/threonine-specific protein phosphatase
1038:
466:then a wide number of problems ranging from
983:
981:
935:
530:
2075:
2061:
1237:
1223:
700:
621:
266:This dephosphorylation reaction occurs in
24:
1151:
1105:
1064:
1015:
1005:
961:
905:
856:
718:
661:
659:
657:
655:
556:
1168:
978:
887:
812:Gomperts, Bastein D. (August 19, 2009).
811:
426:
361:
1430:Ubiquitin carboxy-terminal hydrolase L1
317:Smooth muscle tissue is mostly made of
2368:
1057:10.1111/j.1469-7793.2000.t01-2-00177.x
770:
652:
420:), and the water molecule attacks the
2056:
2010:either deoxy- or ribo-
1218:
741:
1591:Protein serine/threonine phosphatase
665:
1692:Cyclic nucleotide phosphodiesterase
1686:Clostridium perfringens alpha toxin
1487:Tartrate-resistant acid phosphatase
838:
13:
1535:Pyruvate dehydrogenase phosphatase
1126:
407:. Myosin's regulatory subunit MLC
14:
2392:
1435:4-hydroxybenzoyl-CoA thioesterase
2353:
839:Shi, Yigong (October 30, 2009).
815:Signal Transduction: 2nd Edition
668:Advances in Physiology Education
1753:N-acetylglucosamine-6-sulfatase
1641:Sphingomyelin phosphodiesterase
1081:
1032:
929:
881:
1562:Inositol-phosphate phosphatase
1425:Palmitoyl protein thioesterase
1134:Pato MD, Adelstein RS (1983).
832:
805:
735:
694:
322:the muscle needs to contract,
293:rho-associated protein kinases
250:the regulatory light chain of
234:(EC 3.1.3.53; systematic name
228:Myosin light-chain phosphatase
19:Myosin Light-Chain Phosphatase
1:
1925:RNA-induced silencing complex
1153:10.1016/S0021-9258(18)32330-5
954:10.1161/circresaha.108.188524
508:
2029:Serratia marcescens nuclease
1596:Dual-specificity phosphatase
1586:Protein tyrosine phosphatase
1191:10.1126/science.273.5272.245
398:
357:
303:, also regulate the enzyme.
7:
2082:
1506:Fructose 1,6-bisphosphatase
481:
437:Regulation and Human Health
306:
236:-phosphate phosphohydrolase
10:
2397:
2381:Enzymes of known structure
858:10.1016/j.cell.2009.10.006
818:. London: Academic Press.
310:
2239:
2231:Michaelis–Menten kinetics
2203:
2172:
2141:
2090:
2009:
1897:
1849:
1835:
1813:
1795:
1781:
1761:
1743:Galactosamine-6 sulfatase
1700:
1604:
1443:
1411:
1299:6-phosphogluconolactonase
1261:
1094:Human Reproduction Update
493:Myosin light-chain kinase
336:myosin light-chain kinase
276:myosin light-chain kinase
209:
197:
185:
180:
176:
160:
141:
129:
117:
105:
93:
81:
69:
64:
52:
40:
35:
23:
18:
2123:Diffusion-limited enzyme
1491:Purple acid phosphatases
680:10.1152/advan.00025.2003
375:, as it plays a role in
1007:10.1074/jbc.m112.341479
894:Frontiers in Bioscience
599:10.1023/A:1005385302064
587:J Muscle Res Cell Motil
1916:Microprocessor complex
1555:Beta-propeller phytase
791:10.1006/geno.1998.5222
720:10.1074/jbc.m703472200
433:
368:
332:sarcoplasmic reticulum
284:myosin binding subunit
2216:Eadie–Hofstee diagram
2149:Allosteric regulation
1851:Endodeoxyribonuclease
1748:Iduronate-2-sulfatase
1501:Glucose 6-phosphatase
1287:Butyrylcholinesterase
1107:10.1093/humupd/dmq016
1045:Journal of Physiology
756:10.1242/jcs.115.2.241
430:
365:
280:protein phosphatase 1
274:process initiated by
2226:Lineweaver–Burk plot
2034:Micrococcal nuclease
1869:Deoxyribonuclease IV
1864:Deoxyribonuclease II
1797:Exodeoxyribonuclease
1457:Alkaline phosphatase
1282:Acetylcholinesterase
476:erectile dysfunction
313:Smooth muscle tissue
268:smooth muscle tissue
1889:UvrABC endonuclease
1859:Deoxyribonuclease I
1582:Protein phosphatase
1518:Protein phosphatase
1316:Bile salt-dependent
1304:PAF acetylhydrolase
1183:1996Sci...273..245K
558:10.1038/nature02582
549:2004Natur.429..780T
344:crossbridge cycling
326:ions flow into the
2185:Enzyme superfamily
2118:Enzyme promiscuity
2022:Mung bean nuclease
1881:Restriction enzyme
1874:Restriction enzyme
434:
369:
272:muscle contraction
232:myosin phosphatase
2341:
2340:
2050:
2049:
2046:
2045:
2042:
2041:
1831:
1830:
1823:Oligonucleotidase
1768:deoxyribonuclease
1736:Steroid sulfatase
1611:Phosphodiesterase
1340:Hormone-sensitive
1177:(5272): 245–248.
648:978-0-12-632310-8
405:glycogen synthase
225:
224:
221:
220:
124:metabolic pathway
2388:
2358:
2357:
2349:
2221:Hanes–Woolf plot
2164:Enzyme activator
2159:Enzyme inhibitor
2133:Enzyme catalysis
2077:
2070:
2063:
2054:
2053:
1899:Endoribonuclease
1885:
1879:
1847:
1846:
1793:
1792:
1779:
1778:
1479:Acid phosphatase
1360:Monoacylglycerol
1270:ester hydrolases
1239:
1232:
1225:
1216:
1215:
1210:
1165:
1155:
1120:
1119:
1109:
1085:
1079:
1078:
1068:
1036:
1030:
1029:
1019:
1009:
1000:(25): 20975–85.
985:
976:
975:
965:
933:
927:
926:
924:
922:
909:
885:
879:
878:
860:
836:
830:
829:
809:
803:
802:
774:
768:
767:
739:
733:
732:
722:
713:(39): 28874–83.
698:
692:
691:
663:
650:
625:
619:
618:
582:
571:
570:
560:
528:
373:eukaryotic cells
297:arachidonic acid
262:O = + phosphate
248:dephosphorylates
242:(specifically a
178:
177:
28:
16:
15:
2396:
2395:
2391:
2390:
2389:
2387:
2386:
2385:
2366:
2365:
2364:
2352:
2344:
2342:
2337:
2249:Oxidoreductases
2235:
2211:Enzyme kinetics
2199:
2195:List of enzymes
2168:
2137:
2108:Catalytic triad
2086:
2081:
2051:
2038:
2005:
1893:
1883:
1877:
1840:
1827:
1815:Exoribonuclease
1809:
1786:
1770:
1766:
1757:
1731:Arylsulfatase L
1726:Arylsulfatase B
1721:Arylsulfatase A
1696:
1609:
1600:
1439:
1407:
1269:
1257:
1243:
1213:
1146:(11): 7047–54.
1129:
1127:Further reading
1124:
1123:
1086:
1082:
1037:
1033:
986:
979:
934:
930:
920:
918:
900:(1–3): 270–85.
886:
882:
837:
833:
826:
810:
806:
775:
771:
740:
736:
699:
695:
664:
653:
626:
622:
583:
574:
543:(6993): 780–4.
529:
516:
511:
484:
439:
422:phosphorus atom
419:
410:
401:
394:
360:
352:phosphate group
341:
315:
309:
282:, or PP1), the
261:
31:
12:
11:
5:
2394:
2384:
2383:
2378:
2363:
2362:
2339:
2338:
2336:
2335:
2322:
2309:
2296:
2283:
2270:
2257:
2243:
2241:
2237:
2236:
2234:
2233:
2228:
2223:
2218:
2213:
2207:
2205:
2201:
2200:
2198:
2197:
2192:
2187:
2182:
2176:
2174:
2173:Classification
2170:
2169:
2167:
2166:
2161:
2156:
2151:
2145:
2143:
2139:
2138:
2136:
2135:
2130:
2125:
2120:
2115:
2110:
2105:
2100:
2094:
2092:
2088:
2087:
2080:
2079:
2072:
2065:
2057:
2048:
2047:
2044:
2043:
2040:
2039:
2037:
2036:
2031:
2026:
2025:
2024:
2013:
2011:
2007:
2006:
2004:
2003:
1998:
1997:
1996:
1991:
1986:
1981:
1971:
1966:
1961:
1956:
1955:
1954:
1949:
1944:
1939:
1929:
1928:
1927:
1918:
1903:
1901:
1895:
1894:
1892:
1891:
1886:
1871:
1866:
1861:
1855:
1853:
1844:
1833:
1832:
1829:
1828:
1826:
1825:
1819:
1817:
1811:
1810:
1808:
1807:
1801:
1799:
1790:
1776:
1759:
1758:
1756:
1755:
1750:
1745:
1740:
1739:
1738:
1733:
1728:
1723:
1710:
1708:
1698:
1697:
1695:
1694:
1689:
1679:
1674:
1665:
1660:
1655:
1650:
1649:
1648:
1638:
1637:
1636:
1631:
1621:
1615:
1613:
1602:
1601:
1599:
1598:
1593:
1588:
1579:
1578:
1577:
1559:
1558:
1557:
1547:
1542:
1537:
1532:
1527:
1526:
1525:
1515:
1514:
1513:
1503:
1498:
1493:
1476:
1475:
1474:
1469:
1464:
1453:
1451:
1441:
1440:
1438:
1437:
1432:
1427:
1421:
1419:
1409:
1408:
1406:
1405:
1400:
1394:
1393:
1392:
1391:
1386:
1381:
1370:
1369:
1368:
1367:
1365:Diacylglycerol
1362:
1357:
1352:
1347:
1342:
1337:
1332:
1327:
1318:
1307:
1306:
1301:
1296:
1294:Pectinesterase
1291:
1290:
1289:
1284:
1277:Cholinesterase
1273:
1271:
1259:
1258:
1242:
1241:
1234:
1227:
1219:
1212:
1211:
1166:
1130:
1128:
1125:
1122:
1121:
1080:
1031:
977:
928:
880:
831:
825:978-0123694416
824:
804:
769:
734:
693:
651:
620:
572:
513:
512:
510:
507:
506:
505:
500:
495:
490:
483:
480:
446:GTP activates
438:
435:
417:
408:
400:
397:
392:
359:
356:
339:
311:Main article:
308:
305:
264:
263:
259:
230:, also called
223:
222:
219:
218:
213:
207:
206:
201:
195:
194:
189:
183:
182:
174:
173:
164:
158:
157:
146:
139:
138:
133:
127:
126:
121:
115:
114:
109:
103:
102:
97:
91:
90:
85:
79:
78:
73:
67:
66:
62:
61:
56:
50:
49:
44:
38:
37:
33:
32:
29:
21:
20:
9:
6:
4:
3:
2:
2393:
2382:
2379:
2377:
2374:
2373:
2371:
2361:
2356:
2351:
2350:
2347:
2333:
2329:
2328:
2323:
2320:
2316:
2315:
2310:
2307:
2303:
2302:
2297:
2294:
2290:
2289:
2284:
2281:
2277:
2276:
2271:
2268:
2264:
2263:
2258:
2255:
2251:
2250:
2245:
2244:
2242:
2238:
2232:
2229:
2227:
2224:
2222:
2219:
2217:
2214:
2212:
2209:
2208:
2206:
2202:
2196:
2193:
2191:
2190:Enzyme family
2188:
2186:
2183:
2181:
2178:
2177:
2175:
2171:
2165:
2162:
2160:
2157:
2155:
2154:Cooperativity
2152:
2150:
2147:
2146:
2144:
2140:
2134:
2131:
2129:
2126:
2124:
2121:
2119:
2116:
2114:
2113:Oxyanion hole
2111:
2109:
2106:
2104:
2101:
2099:
2096:
2095:
2093:
2089:
2085:
2078:
2073:
2071:
2066:
2064:
2059:
2058:
2055:
2035:
2032:
2030:
2027:
2023:
2020:
2019:
2018:
2015:
2014:
2012:
2008:
2002:
1999:
1995:
1992:
1990:
1987:
1985:
1982:
1980:
1977:
1976:
1975:
1972:
1970:
1967:
1965:
1962:
1960:
1957:
1953:
1950:
1948:
1945:
1943:
1940:
1938:
1935:
1934:
1933:
1930:
1926:
1922:
1919:
1917:
1913:
1910:
1909:
1908:
1905:
1904:
1902:
1900:
1896:
1890:
1887:
1882:
1875:
1872:
1870:
1867:
1865:
1862:
1860:
1857:
1856:
1854:
1852:
1848:
1845:
1843:
1838:
1834:
1824:
1821:
1820:
1818:
1816:
1812:
1806:
1803:
1802:
1800:
1798:
1794:
1791:
1789:
1784:
1780:
1777:
1774:
1769:
1764:
1760:
1754:
1751:
1749:
1746:
1744:
1741:
1737:
1734:
1732:
1729:
1727:
1724:
1722:
1719:
1718:
1717:
1716:
1715:arylsulfatase
1712:
1711:
1709:
1707:
1703:
1699:
1693:
1690:
1687:
1683:
1680:
1678:
1675:
1673:
1669:
1666:
1664:
1661:
1659:
1656:
1654:
1651:
1647:
1644:
1643:
1642:
1639:
1635:
1632:
1630:
1627:
1626:
1625:
1624:Phospholipase
1622:
1620:
1617:
1616:
1614:
1612:
1607:
1603:
1597:
1594:
1592:
1589:
1587:
1583:
1580:
1576:
1572:
1568:
1565:
1564:
1563:
1560:
1556:
1553:
1552:
1551:
1548:
1546:
1543:
1541:
1538:
1536:
1533:
1531:
1528:
1524:
1521:
1520:
1519:
1516:
1512:
1509:
1508:
1507:
1504:
1502:
1499:
1497:
1494:
1492:
1488:
1484:
1480:
1477:
1473:
1470:
1468:
1465:
1463:
1460:
1459:
1458:
1455:
1454:
1452:
1450:
1446:
1442:
1436:
1433:
1431:
1428:
1426:
1423:
1422:
1420:
1418:
1414:
1410:
1404:
1401:
1399:
1396:
1395:
1390:
1387:
1385:
1382:
1380:
1377:
1376:
1375:
1374:Phospholipase
1372:
1371:
1366:
1363:
1361:
1358:
1356:
1353:
1351:
1348:
1346:
1343:
1341:
1338:
1336:
1333:
1331:
1328:
1326:
1322:
1319:
1317:
1314:
1313:
1312:
1309:
1308:
1305:
1302:
1300:
1297:
1295:
1292:
1288:
1285:
1283:
1280:
1279:
1278:
1275:
1274:
1272:
1268:
1264:
1260:
1255:
1251:
1247:
1240:
1235:
1233:
1228:
1226:
1221:
1220:
1217:
1208:
1204:
1200:
1196:
1192:
1188:
1184:
1180:
1176:
1172:
1167:
1163:
1159:
1154:
1149:
1145:
1141:
1140:J. Biol. Chem
1137:
1132:
1131:
1117:
1113:
1108:
1103:
1100:(6): 725–44.
1099:
1095:
1091:
1084:
1076:
1072:
1067:
1062:
1058:
1054:
1051:(2): 177–85.
1050:
1046:
1042:
1035:
1027:
1023:
1018:
1013:
1008:
1003:
999:
995:
991:
984:
982:
973:
969:
964:
959:
955:
951:
948:(4): 531–40.
947:
943:
939:
932:
917:
913:
908:
903:
899:
895:
891:
884:
876:
872:
868:
864:
859:
854:
851:(3): 468–84.
850:
846:
842:
835:
827:
821:
817:
816:
808:
800:
796:
792:
788:
785:(1): 325–41.
784:
780:
773:
765:
761:
757:
753:
749:
745:
738:
730:
726:
721:
716:
712:
708:
707:J. Biol. Chem
704:
697:
689:
685:
681:
677:
673:
669:
662:
660:
658:
656:
649:
645:
641:
640:0-12-632310-0
637:
633:
629:
624:
616:
612:
608:
604:
600:
596:
593:(4): 325–41.
592:
588:
581:
579:
577:
568:
564:
559:
554:
550:
546:
542:
538:
534:
527:
525:
523:
521:
519:
514:
504:
501:
499:
496:
494:
491:
489:
486:
485:
479:
477:
473:
469:
463:
461:
455:
453:
449:
445:
429:
425:
423:
414:
406:
396:
389:
385:
382:
381:cell division
378:
374:
364:
355:
353:
348:
345:
337:
333:
329:
325:
320:
314:
304:
302:
298:
294:
289:
285:
281:
277:
273:
269:
258:phosphate + H
257:
256:
255:
253:
249:
245:
241:
237:
233:
229:
217:
214:
212:
208:
205:
202:
200:
196:
193:
190:
188:
184:
179:
175:
172:
168:
165:
163:
162:Gene Ontology
159:
156:
153:
150:
147:
144:
140:
137:
134:
132:
128:
125:
122:
120:
116:
113:
110:
108:
104:
101:
100:NiceZyme view
98:
96:
92:
89:
86:
84:
80:
77:
74:
72:
68:
63:
60:
57:
55:
51:
48:
45:
43:
39:
34:
27:
22:
17:
2327:Translocases
2324:
2311:
2298:
2285:
2272:
2262:Transferases
2259:
2246:
2103:Binding site
1884:}}
1878:{{
1842:Endonuclease
1773:ribonuclease
1713:
1496:Nucleotidase
1417:Thioesterase
1174:
1170:
1143:
1139:
1097:
1093:
1083:
1048:
1044:
1034:
997:
993:
945:
941:
931:
919:. Retrieved
897:
893:
883:
848:
844:
834:
814:
807:
782:
778:
772:
750:(2): 780–4.
747:
743:
737:
710:
706:
696:
674:(4): 201–6.
671:
667:
631:
623:
590:
586:
540:
536:
478:can result.
472:hypertension
464:
456:
440:
402:
390:
386:
370:
349:
316:
265:
235:
231:
227:
226:
88:BRENDA entry
2098:Active site
2017:Nuclease S1
1788:Exonuclease
1682:Lecithinase
1511:Calcineurin
1449:Phosphatase
1355:Lipoprotein
1345:Endothelial
994:J Biol Chem
907:10.2741/lee
76:IntEnz view
59:86417-96-1
36:Identifiers
2370:Categories
2301:Isomerases
2275:Hydrolases
2142:Regulation
1330:Pancreatic
1267:Carboxylic
744:J Cell Sci
509:References
503:Rho kinase
448:Rho-kinase
367:catalysis.
145:structures
112:KEGG entry
2180:EC number
1907:RNase III
1765:(includes
1706:Sulfatase
1619:Autotaxin
1483:Prostatic
1335:Lysosomal
1250:esterases
1246:Hydrolase
942:Circ. Res
460:inhibitor
399:Mechanism
358:Structure
330:from the
288:manganese
252:myosin II
238:), is an
65:Databases
2376:EC 3.1.3
2204:Kinetics
2128:Cofactor
2091:Activity
2001:RNase T1
1763:Nuclease
1398:Cutinase
1207:37249779
1116:20551073
1075:10639096
1026:22544752
972:19131646
921:March 9,
916:10077543
875:13903804
867:19879837
779:Genomics
764:11839776
729:17636256
688:14627615
628:Page 174
615:27448238
567:15164081
482:See also
377:glycogen
307:Function
216:proteins
204:articles
192:articles
149:RCSB PDB
47:3.1.3.53
2360:Biology
2314:Ligases
2084:Enzymes
1974:RNase E
1969:RNase Z
1964:RNase A
1959:RNase P
1932:RNase H
1550:Phytase
1350:Hepatic
1325:Lingual
1321:Gastric
1199:8662509
1179:Bibcode
1171:Science
1162:6304072
1066:2269761
1017:3375521
963:2649695
799:9570949
607:9635276
545:Bibcode
452:telokin
432:Ser-19.
328:cytosol
324:calcium
246:) that
171:QuickGO
136:profile
119:MetaCyc
54:CAS no.
2346:Portal
2288:Lyases
1912:Drosha
1837:3.1.21
1805:RecBCD
1783:3.1.11
1403:PETase
1311:Lipase
1205:
1197:
1160:
1114:
1073:
1063:
1024:
1014:
970:
960:
914:
873:
865:
822:
797:
762:
727:
686:
646:
638:
613:
605:
565:
537:Nature
488:Myosin
474:, and
468:asthma
413:serine
240:enzyme
199:PubMed
181:Search
167:AmiGO
155:PDBsum
95:ExPASy
83:BRENDA
71:IntEnz
42:EC no.
2240:Types
1921:Dicer
1876:;see
1702:3.1.6
1672:PDE4B
1668:PDE4A
1606:3.1.4
1575:IMPA3
1571:IMPA2
1567:IMPA1
1445:3.1.3
1413:3.1.2
1263:3.1.1
1203:S2CID
871:S2CID
611:S2CID
319:actin
131:PRIAM
2332:list
2325:EC7
2319:list
2312:EC6
2306:list
2299:EC5
2293:list
2286:EC4
2280:list
2273:EC3
2267:list
2260:EC2
2254:list
2247:EC1
1839:-31:
1785:-16:
1771:and
1677:PDE5
1663:PDE3
1658:PDE2
1653:PDE1
1545:PTEN
1530:OCRL
1523:PP2A
1472:ALPP
1467:ALPL
1462:ALPI
1256:3.1)
1195:PMID
1158:PMID
1112:PMID
1071:PMID
1022:PMID
968:PMID
923:2015
912:PMID
863:PMID
845:Cell
820:ISBN
795:PMID
760:PMID
725:PMID
684:PMID
644:ISBN
636:ISBN
630:in:
603:PMID
563:PMID
498:Rhoa
444:RhoA
301:cAMP
299:and
211:NCBI
152:PDBe
107:KEGG
1994:4/5
1187:doi
1175:273
1148:doi
1144:258
1102:doi
1061:PMC
1053:doi
1049:522
1012:PMC
1002:doi
998:287
958:PMC
950:doi
946:104
902:doi
853:doi
849:139
787:doi
752:doi
748:115
715:doi
711:282
676:doi
595:doi
553:doi
541:429
416:MLC
187:PMC
143:PDB
2372::
1952:2C
1947:2B
1942:2A
1923::
1914::
1704::
1584::
1573:,
1569:,
1485:)/
1447::
1415::
1384:A2
1379:A1
1265::
1254:EC
1248::
1201:.
1193:.
1185:.
1173:.
1156:.
1142:.
1138:.
1110:.
1098:16
1096:.
1092:.
1069:.
1059:.
1047:.
1043:.
1020:.
1010:.
996:.
992:.
980:^
966:.
956:.
944:.
940:.
910:.
896:.
892:.
869:.
861:.
847:.
843:.
793:.
783:49
781:.
758:.
746:.
723:.
709:.
705:.
682:.
672:27
670:.
654:^
642:,
609:.
601:.
591:19
589:.
575:^
561:.
551:.
539:.
535:.
517:^
470:,
418:20
409:20
393:20
340:20
254::
169:/
2348::
2334:)
2330:(
2321:)
2317:(
2308:)
2304:(
2295:)
2291:(
2282:)
2278:(
2269:)
2265:(
2256:)
2252:(
2076:e
2069:t
2062:v
1989:3
1984:2
1979:1
1937:1
1775:)
1688:)
1684:(
1670:/
1646:1
1634:D
1629:C
1608::
1489:/
1481:(
1389:B
1323:/
1252:(
1238:e
1231:t
1224:v
1209:.
1189::
1181::
1164:.
1150::
1118:.
1104::
1077:.
1055::
1028:.
1004::
974:.
952::
925:.
904::
898:4
877:.
855::
828:.
801:.
789::
766:.
754::
731:.
717::
690:.
678::
617:.
597::
569:.
555::
547::
260:2
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.