81:
599:
585:
109:
100:
672:
1185:
72:
1132:
889:
This equilibrium does not involve a change in hydrogen ion concentration, which would predict that the equilibrium is independent of pH. The red line on the predominance diagram is not quite horizontal due to the simultaneous equilibrium with the chromate ion. The hydrogen chromate ion may be
1274:
Subsequent leaching of this material at higher temperatures dissolves the chromates, leaving a residue of insoluble iron oxide. Normally the chromate solution is further processed to make chromium metal, but a chromate salt may be obtained directly from the liquor.
1522:
Brito, F.; Ascanioa, J.; Mateoa, S.; Hernándeza, C.; Araujoa, L.; Gili, P.; Martín-Zarzab, P.; Domínguez, S.; Mederos, A. (1997). "Equilibria of chromate(VI) species in acid medium and ab initio studies of these species".
1734:
of chromium (VI) compounds. Chromium (VI) compounds cause cancer of the lung. Also positive associations have been observed between exposure to chromium (VI) compounds and cancer of the nose and nasal sinuses. There is
1433:"Effect of solution acidity on the crystallization of polychromates in uranyl-bearing systems: synthesis and crystal structures of Rb2[(UO2)(Cr2O7)(NO3)2] and two new polymorphs of Rb2Cr3O10"
1692:
730:
and the analytical concentration of chromium. The chromate ion is the predominant species in alkaline solutions, but dichromate can become the predominant ion in acidic solutions.
1608:
Anger, Gerd; Halstenberg, Jost; Hochgeschwender, Klaus; Scherhag, Christoph; Korallus, Ulrich; Knopf, Herbert; Schmidt, Peter; Ohlinger, Manfred (2005). "Chromium
Compounds".
1286:, which can occur as spectacular long red crystals, is the most commonly found chromate mineral. Rare potassium chromate minerals and related compounds are found in the
1343:. The use of chromate compounds in manufactured goods is restricted in the EU (and by market commonality the rest of the world) by EU Parliament directive on the
197:
2638:
2546:
1711:
1344:
1789:
1176:, chromates and dichromates convert into trivalent chromium, Cr, salts of which typically have a distinctively different blue-green color.
397:
1688:
1898:
1169:
was used for a very long time before environmental regulations discouraged its use. When used as oxidizing agents or titrants in a
2631:
189:
1705:
1654:
1589:
1494:
1415:
3160:
2624:
1559:
362:
2414:
1782:
498:
1219:
in the presence of air. The chromium is oxidized to the hexavalent form, while the iron forms iron(III) oxide, Fe
1625:
1431:
Nazarchuk, Evgeny V.; Siidra, Oleg I.; Charkin, Dmitry O.; Kalmykov, Stepan N.; Kotova, Elena L. (2021-02-01).
1850:
127:
598:
292:
17:
3124:
2465:
1775:
1356:
632:
322:
1739:
in experimental animals for the carcinogenicity of chromium (VI) compounds. Chromium (VI) compounds are
903:
584:
3155:
2879:
2560:
2522:
2303:
1512:. A comprehensive database of published data on equilibrium constants of metal complexes and ligands.
1432:
31:
2868:
1165:
are only very slightly soluble in water and are thus used as pigments. The lead-containing pigment
348:
340:
1303:
2269:
2227:
2203:
1211:, found as brittle metallic black crystals or granules. Chromite ore is heated with a mixture of
1153:
to protect metals from corrosion and to improve paint adhesion. Chromate and dichromate salts of
3114:
2911:
2432:
2293:
2283:
1642:
1554:, translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter,
2580:
2373:
2363:
2348:
2317:
2251:
2237:
2145:
2041:
2031:
2026:
1996:
1812:
1579:
1486:
1407:
1062:
shows that chromates are weaker oxidizing agent in alkaline solution than in acid solution.
1013:
2988:
2383:
2327:
2140:
2051:
2016:
2011:
1978:
1963:
1949:
1939:
1832:
1162:
733:
Further condensation reactions can occur in strongly acidic solution with the formation of
723:
683:
675:
604:
570:
310:
302:
164:
156:
1757:
8:
3024:
2771:
2447:
2393:
2213:
2061:
1882:
1376:
1308:
1149:, mainly sodium dichromate, were produced in 1985. Chromates and dichromates are used in
1146:
145:
38:
226:
218:
3150:
2753:
2733:
2713:
2703:
2675:
2353:
2006:
1798:
1460:
1379:
of chromium. Thus, when pCr = 2, the chromium concentration is 10 mol/L.
631:, replaces one or more oxygen atoms. In acid solution the unstable blue peroxo complex
590:
1536:
3046:
3006:
2970:
2933:
2839:
2815:
2455:
1701:
1650:
1621:
1585:
1555:
1490:
1464:
1452:
1411:
1212:
1173:
648:
612:
2805:
2723:
2689:
2532:
2508:
2189:
2184:
2115:
1613:
1581:
Toxic
Substances Controls Guide: Federal Regulation of Chemicals in the Environment
1532:
1478:
1444:
1399:
1216:
425:
1607:
281:
273:
2781:
2743:
2661:
1731:
1059:
1009:
563:
559:
1509:
2903:
2597:
1287:
1150:
492:
478:
30:
This article is about the salts of the chromium(VI) anion. For other uses, see
1762:
3144:
2825:
2795:
1617:
1456:
1340:
1328:
1166:
1139:
997:
value for this reaction shows that it can be ignored at pH > 4.
2616:
939:
is not well characterized. Reported values vary between about −0.8 and 1.6.
3085:
2479:
1448:
1320:
1316:
1154:
1016:
it to oxidation state +3. In acid solution the aquated Cr ion is produced.
891:
483:
80:
54:
671:
1521:
1324:
1184:
71:
42:
1291:
247:
2082:
1158:
1135:
468:
50:
108:
1767:
1193:
1131:
799:
784:
46:
1758:
National
Pollutant Inventory - Chromium(VI) and compounds fact sheet
1550:
Holleman, Arnold
Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.),
1327:. Also positive associations have been observed between exposure to
942:
The dichromate ion is a somewhat weaker base than the chromate ion:
491:
Except where otherwise noted, data are given for materials in their
99:
1279:
1200:
1189:
652:
644:
616:
555:
551:
237:
196:
188:
1345:
Restriction of
Hazardous Substances (RoHS) Directive (2002/95/EC)
1323:). Inhaling particles of hexavalent chromium compounds can cause
567:
259:
1647:
Industrial
Minerals & Rocks: Commodities, Markets, and Uses
1332:
682:
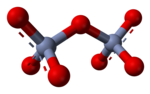
In aqueous solution, chromate and dichromate anions exist in a
1430:
1058:
In alkaline solution chromium(III) hydroxide is produced. The
1375:
pCr is equal to the negative of the decimal logarithm of the
1312:
1170:
208:
177:
327:
2075:
1336:
726:
shows that the position of the equilibrium depends on both
1891:
1826:
787:
of chromium(VI) have structures made up of tetrahedral CrO
1012:. Commonly three electrons are added to a chromium atom,
655:
results in the formation of the more stable complex CrO(O
1670:
1437:
573:, chromate and dichromate ions can be interconvertible.
727:
1700:. Lyon: International Agency for Research on Cancer.
1577:
1179:
1008:The chromate and dichromate ions are fairly strong
844:It is also in equilibrium with the dichromate ion:
1640:
1199:The primary chromium ore is the mixed metal oxide
1477:
1398:
1003:
3142:
1763:Demonstration of chromate-dichromate equilibrium
280:
272:
1694:Volume 100C: Arsenic, Metals, Fibres, and Dusts
1145:Approximately 136,000 tonnes (150,000 tons) of
163:
155:
34:. See also for disambiguation of derived terms.
1610:Ullmann's Encyclopedia of Industrial Chemistry
1549:
2646:
2632:
1783:
1040:+ 14 H + 6 e → 2 Cr + 7 H
473:115.994 g mol and 215.988 g mol
381:dichromate: InChI=1S/2Cr.7O/q;;;;;;;2*-1
1578:Worobec, Mary Devine; Hogue, Cheryl (1992).
890:protonated, with the formation of molecular
2639:
2625:
1790:
1776:
1188:Crocoite specimen from the Red Lead Mine,
1603:
1601:
1543:
309:
301:
1681:
1573:
1571:
1183:
1130:
670:
1278:Chromate containing minerals are rare.
666:
347:
339:
246:
14:
3143:
1797:
1641:Papp, John F.; Lipin Bruce R. (2006).
1634:
1598:
647:molecule, which may be extracted into
576:
371:chromate: InChI=1S/Cr.4O/q;;;2*-1
2620:
1771:
1568:
1294:– the only known dichromate mineral.
384:Key: SOCTUWSJJQCPFX-UHFFFAOYSA-N
374:Key: ZCDOYSPFYFSLEW-UHFFFAOYSA-N
225:
217:
1687:
529:salts contain the dichromate anion,
1304:Hexavalent chromium § Toxicity
262:
24:
513:salts contain the chromate anion,
410:dichromate: O=(=O)()O(=O)(=O)
107:
98:
79:
70:
25:
3172:
1751:
1180:Natural occurrence and production
1741:carcinogenic to humans (Group 1)
643:, is formed; it is an uncharged
597:
583:
1663:
1126:
794:The hydrogen chromate ion, HCrO
495:(at 25 °C , 100 kPa).
1515:
1503:
1471:
1424:
1392:
1369:
1114:
1046:
1004:Oxidation–reduction properties
977:
831:
13:
1:
1537:10.1016/S0277-5387(97)00128-9
1386:
2076:Organochromium(II) compounds
7:
1892:Organochromium(I) compounds
1827:Organochromium(0) compounds
1357:Chromate conversion coating
1350:
1297:
633:Chromium(VI) oxide peroxide
615:, giving products in which
94:
66:
10:
3177:
3161:Transition metal oxyanions
1671:"Mines, Minerals and More"
1301:
562:and are moderately strong
36:
29:
3076:
2963:
2902:
2861:
2654:
2648:Chromates and dichromates
2596:
2446:
2407:
2341:
2133:
2108:
1932:
1875:
1805:
1584:. BNA Books. p. 13.
1483:Chemistry of the Elements
1481:; Earnshaw, Alan (1997).
1404:Chemistry of the Elements
1402:; Earnshaw, Alan (1997).
1122: = −0.13 V
489:
418:
393:
358:
138:
126:
121:
93:
65:
1618:10.1002/14356007.a07_067
1362:
1054: = 1.33 V
61:Chromate and dichromate
37:Not to be confused with
2606:Chromate and dichromate
1612:. Weinheim: Wiley-VCH.
791:units sharing corners.
405:chromate: (=O)(=O)
132:Chromate and dichromate
1449:10.1515/zkri-2020-0078
1196:
1142:
679:
112:
103:
84:
75:
1649:(7th ed.). SME.
1487:Butterworth-Heinemann
1479:Greenwood, Norman N.
1408:Butterworth-Heinemann
1400:Greenwood, Norman N.
1302:Further information:
1187:
1163:alkaline earth metals
1134:
912:for the equilibrium
674:
611:Chromates react with
128:Systematic IUPAC name
111:
102:
83:
74:
724:predominance diagram
684:chemical equilibrium
676:Predominance diagram
667:Acid–base properties
605:potassium dichromate
1737:sufficient evidence
1728:sufficient evidence
1552:Inorganic Chemistry
1377:molar concentration
1309:Hexavalent chromium
1147:hexavalent chromium
577:Chemical properties
62:
39:chromite (compound)
27:Chromium(VI) anions
1799:Chromium compounds
1730:in humans for the
1197:
1143:
680:
591:potassium chromate
499:Infobox references
113:
104:
85:
76:
60:
3138:
3137:
2959:
2958:
2614:
2613:
2109:Chromium(II, III)
2104:
2103:
1928:
1927:
1871:
1870:
1707:978-92-832-0135-9
1656:978-0-87335-233-8
1591:978-0-87179-752-0
1531:(21): 3835–3846.
1510:IUPAC SC-Database
1496:978-0-08-037941-8
1417:978-0-08-037941-8
1311:compounds can be
1213:calcium carbonate
1174:chemical reaction
986: = 1.18
613:hydrogen peroxide
507:Chemical compound
505:
504:
346:dichromate:
323:CompTox Dashboard
308:dichromate:
279:dichromate:
245:dichromate:
224:dichromate:
198:Interactive image
195:dichromate:
190:Interactive image
162:dichromate:
117:
116:
89:
88:
16:(Redirected from
3168:
3156:Oxidizing agents
3130:
3120:
3110:
3095:
3068:
3042:
3020:
3002:
2984:
2951:
2929:
2894:
2875:
2859:
2858:
2853:
2835:
2821:
2811:
2801:
2791:
2777:
2767:
2749:
2739:
2729:
2719:
2709:
2699:
2685:
2671:
2641:
2634:
2627:
2618:
2617:
2586:
2575:
2556:
2542:
2528:
2518:
2504:
2489:
2475:
2461:
2438:
2428:
2399:
2389:
2379:
2369:
2359:
2333:
2323:
2313:
2299:
2289:
2279:
2265:
2247:
2233:
2223:
2209:
2199:
2180:
2125:
2096:
2073:
2072:
2067:
2057:
2047:
2037:
2022:
2002:
1992:
1974:
1959:
1945:
1920:
1889:
1888:
1864:
1846:
1824:
1823:
1818:
1792:
1785:
1778:
1769:
1768:
1746:
1745:
1723:
1722:
1716:
1710:. Archived from
1699:
1685:
1679:
1678:
1667:
1661:
1660:
1638:
1632:
1631:
1605:
1596:
1595:
1575:
1566:
1564:
1547:
1541:
1540:
1519:
1513:
1507:
1501:
1500:
1489:. p. 1009.
1485:(2nd ed.).
1475:
1469:
1468:
1428:
1422:
1421:
1406:(2nd ed.).
1396:
1380:
1373:
1290:. Among them is
1217:sodium carbonate
1115:
1112:
1111:
1110:
1101:
1100:
1099:
1089:
1087:
1086:
1076:
1075:
1074:
1047:
1039:
1038:
1037:
1029:
1028:
1010:oxidizing agents
978:
974:
972:
971:
968:
959:
958:
955:
935:
933:
932:
929:
880:
879:
878:
870:
869:
859:
858:
857:
840: ≈ 5.9
832:
828:
827:
826:
816:
815:
814:
782:
781:
780:
772:
771:
757:
756:
755:
747:
746:
718:
712:
711:
708:
699:
698:
695:
630:
629:
628:
601:
587:
564:oxidizing agents
549:
548:
547:
539:
538:
524:
523:
522:
462:
461:
460:
452:
451:
441:
440:
439:
426:Chemical formula
351:
343:
331:
329:
313:
305:
284:
276:
264:
250:
229:
221:
200:
192:
167:
159:
95:
67:
63:
59:
21:
3176:
3175:
3171:
3170:
3169:
3167:
3166:
3165:
3141:
3140:
3139:
3134:
3129:
3125:
3119:
3115:
3109:
3105:
3101:
3097:
3094:
3090:
3086:
3072:
3067:
3063:
3059:
3055:
3051:
3047:
3041:
3037:
3033:
3029:
3025:
3019:
3015:
3011:
3007:
3001:
2997:
2993:
2989:
2983:
2979:
2975:
2971:
2955:
2950:
2946:
2942:
2938:
2934:
2928:
2924:
2920:
2916:
2912:
2904:Chromate esters
2898:
2892:
2888:
2884:
2880:
2873:
2869:
2862:Chlorochromates
2857:
2852:
2848:
2844:
2840:
2834:
2830:
2826:
2820:
2816:
2810:
2806:
2800:
2796:
2790:
2786:
2782:
2776:
2772:
2766:
2762:
2758:
2754:
2748:
2744:
2738:
2734:
2728:
2724:
2718:
2714:
2708:
2704:
2698:
2694:
2690:
2684:
2680:
2676:
2670:
2666:
2662:
2650:
2645:
2615:
2610:
2592:
2585:
2581:
2573:
2569:
2565:
2561:
2555:
2551:
2547:
2541:
2537:
2533:
2527:
2523:
2517:
2513:
2509:
2503:
2499:
2495:
2491:
2488:
2484:
2480:
2474:
2470:
2466:
2460:
2456:
2442:
2437:
2433:
2427:
2423:
2419:
2415:
2403:
2398:
2394:
2388:
2384:
2378:
2374:
2368:
2364:
2358:
2354:
2337:
2332:
2328:
2322:
2318:
2312:
2308:
2304:
2298:
2294:
2288:
2284:
2278:
2274:
2270:
2264:
2260:
2256:
2252:
2246:
2242:
2238:
2232:
2228:
2222:
2218:
2214:
2208:
2204:
2198:
2194:
2190:
2178:
2174:
2170:
2166:
2162:
2158:
2154:
2150:
2146:
2129:
2124:
2120:
2116:
2100:
2095:
2091:
2087:
2083:
2071:
2066:
2062:
2056:
2052:
2046:
2042:
2036:
2032:
2021:
2017:
2001:
1997:
1991:
1987:
1983:
1979:
1972:
1968:
1964:
1958:
1954:
1950:
1944:
1940:
1924:
1919:
1915:
1911:
1907:
1903:
1899:
1887:
1867:
1863:
1859:
1855:
1851:
1845:
1841:
1837:
1833:
1822:
1817:
1813:
1801:
1796:
1754:
1749:
1732:carcinogenicity
1720:
1718:
1714:
1708:
1697:
1686:
1682:
1669:
1668:
1664:
1657:
1639:
1635:
1628:
1606:
1599:
1592:
1576:
1569:
1562:
1548:
1544:
1520:
1516:
1508:
1504:
1497:
1476:
1472:
1429:
1425:
1418:
1410:. p. 637.
1397:
1393:
1389:
1384:
1383:
1374:
1370:
1365:
1353:
1306:
1300:
1285:
1270:
1266:
1262:
1258:
1254:
1250:
1246:
1242:
1238:
1234:
1226:
1222:
1210:
1206:
1182:
1129:
1121:
1113:
1109:
1107:
1106:
1105:
1103:
1098:
1095:
1094:
1093:
1091:
1085:
1082:
1081:
1080:
1078:
1073:
1070:
1069:
1068:
1066:
1060:redox potential
1053:
1045:
1043:
1036:
1033:
1032:
1031:
1027:
1024:
1023:
1022:
1020:
1006:
1000:
996:
985:
976:
969:
966:
965:
963:
956:
953:
952:
950:
946:
930:
927:
926:
924:
920:
916:
910:
901:
897:
884:
877:
874:
873:
872:
868:
865:
864:
863:
861:
856:
853:
852:
851:
849:
839:
830:
825:
822:
821:
820:
818:
813:
810:
809:
808:
806:
797:
790:
779:
776:
775:
774:
770:
767:
766:
765:
763:
754:
751:
750:
749:
745:
742:
741:
740:
738:
716:
709:
706:
705:
703:
696:
693:
692:
690:
669:
662:
658:
642:
638:
627:
624:
623:
622:
620:
607:
602:
593:
588:
579:
560:oxidation state
546:
543:
542:
541:
537:
534:
533:
532:
530:
521:
518:
517:
516:
514:
508:
501:
496:
459:
456:
455:
454:
450:
447:
446:
445:
443:
438:
435:
434:
433:
431:
428:
414:
411:
406:
401:
400:
389:
386:
385:
382:
376:
375:
372:
366:
365:
354:
338:chromate:
332:
325:
316:
300:chromate:
287:
271:chromate:
265:
253:
232:
216:chromate:
203:
187:chromate:
181:
170:
154:chromate:
148:
134:
133:
58:
35:
28:
23:
22:
15:
12:
11:
5:
3174:
3164:
3163:
3158:
3153:
3136:
3135:
3133:
3132:
3127:
3122:
3117:
3112:
3107:
3103:
3099:
3092:
3088:
3082:
3080:
3074:
3073:
3071:
3070:
3065:
3061:
3057:
3053:
3049:
3044:
3039:
3035:
3031:
3027:
3022:
3017:
3013:
3009:
3004:
2999:
2995:
2991:
2986:
2981:
2977:
2973:
2967:
2965:
2961:
2960:
2957:
2956:
2954:
2953:
2948:
2944:
2940:
2936:
2931:
2926:
2922:
2918:
2914:
2908:
2906:
2900:
2899:
2897:
2896:
2890:
2886:
2882:
2877:
2871:
2865:
2863:
2856:
2855:
2850:
2846:
2842:
2837:
2832:
2828:
2823:
2818:
2813:
2808:
2803:
2798:
2793:
2788:
2784:
2779:
2774:
2769:
2764:
2760:
2756:
2751:
2746:
2741:
2736:
2731:
2726:
2721:
2716:
2711:
2706:
2701:
2696:
2692:
2687:
2682:
2678:
2673:
2668:
2664:
2658:
2656:
2652:
2651:
2644:
2643:
2636:
2629:
2621:
2612:
2611:
2609:
2608:
2602:
2600:
2598:Polyatomic ion
2594:
2593:
2591:
2590:
2588:(hypothetical)
2583:
2577:
2571:
2567:
2563:
2558:
2553:
2549:
2544:
2539:
2535:
2530:
2525:
2520:
2515:
2511:
2506:
2501:
2497:
2493:
2486:
2482:
2477:
2472:
2468:
2463:
2458:
2452:
2450:
2444:
2443:
2441:
2440:
2435:
2430:
2425:
2421:
2417:
2411:
2409:
2405:
2404:
2402:
2401:
2396:
2391:
2386:
2381:
2376:
2371:
2366:
2361:
2356:
2351:
2345:
2343:
2339:
2338:
2336:
2335:
2330:
2325:
2320:
2315:
2310:
2306:
2301:
2296:
2291:
2286:
2281:
2276:
2272:
2267:
2262:
2258:
2254:
2249:
2244:
2240:
2235:
2230:
2225:
2220:
2216:
2211:
2206:
2201:
2196:
2192:
2187:
2182:
2176:
2172:
2168:
2164:
2160:
2156:
2152:
2148:
2143:
2137:
2135:
2131:
2130:
2128:
2127:
2122:
2118:
2112:
2110:
2106:
2105:
2102:
2101:
2099:
2098:
2093:
2089:
2085:
2079:
2077:
2070:
2069:
2064:
2059:
2054:
2049:
2044:
2039:
2034:
2029:
2024:
2019:
2014:
2009:
2004:
1999:
1994:
1989:
1985:
1981:
1976:
1970:
1966:
1961:
1956:
1952:
1947:
1942:
1936:
1934:
1930:
1929:
1926:
1925:
1923:
1922:
1917:
1913:
1909:
1905:
1901:
1895:
1893:
1886:
1885:
1879:
1877:
1873:
1872:
1869:
1868:
1866:
1861:
1857:
1853:
1848:
1843:
1839:
1835:
1830:
1828:
1821:
1820:
1815:
1809:
1807:
1803:
1802:
1795:
1794:
1787:
1780:
1772:
1766:
1765:
1760:
1753:
1752:External links
1750:
1748:
1747:
1706:
1680:
1675:www.mindat.org
1662:
1655:
1633:
1626:
1597:
1590:
1567:
1560:
1542:
1514:
1502:
1495:
1470:
1443:(1–2): 11–21.
1423:
1416:
1390:
1388:
1385:
1382:
1381:
1367:
1366:
1364:
1361:
1360:
1359:
1352:
1349:
1331:compounds and
1299:
1296:
1288:Atacama desert
1283:
1272:
1271:
1268:
1264:
1260:
1256:
1252:
1248:
1244:
1240:
1236:
1232:
1224:
1220:
1208:
1204:
1181:
1178:
1151:chrome plating
1128:
1125:
1124:
1123:
1119:
1108:
1096:
1083:
1071:
1056:
1055:
1051:
1041:
1034:
1025:
1005:
1002:
994:
988:
987:
983:
961:
948:
937:
936:
922:
918:
908:
899:
895:
887:
886:
882:
875:
866:
854:
842:
841:
837:
823:
811:
795:
788:
777:
768:
760:tetrachromates
752:
743:
720:
719:
714:
701:
668:
665:
660:
656:
651:. Addition of
640:
636:
625:
609:
608:
603:
596:
594:
589:
582:
578:
575:
544:
535:
519:
506:
503:
502:
497:
493:standard state
490:
487:
486:
481:
479:Conjugate acid
475:
474:
471:
465:
464:
457:
448:
436:
429:
424:
421:
420:
416:
415:
413:
412:
409:
407:
404:
396:
395:
394:
391:
390:
388:
387:
383:
380:
379:
377:
373:
370:
369:
361:
360:
359:
356:
355:
353:
352:
344:
335:
333:
321:
318:
317:
315:
314:
306:
297:
295:
289:
288:
286:
285:
277:
268:
266:
258:
255:
254:
252:
251:
242:
240:
234:
233:
231:
230:
222:
213:
211:
205:
204:
202:
201:
193:
184:
182:
175:
172:
171:
169:
168:
160:
151:
149:
144:
141:
140:
136:
135:
131:
130:
124:
123:
119:
118:
115:
114:
105:
91:
90:
87:
86:
77:
26:
9:
6:
4:
3:
2:
3173:
3162:
3159:
3157:
3154:
3152:
3149:
3148:
3146:
3131:
3123:
3121:
3113:
3111:
3084:
3083:
3081:
3079:
3075:
3069:
3045:
3043:
3023:
3021:
3005:
3003:
2987:
2985:
2969:
2968:
2966:
2962:
2952:
2932:
2930:
2910:
2909:
2907:
2905:
2901:
2895:
2878:
2876:
2867:
2866:
2864:
2860:
2854:
2838:
2836:
2824:
2822:
2814:
2812:
2804:
2802:
2794:
2792:
2780:
2778:
2770:
2768:
2752:
2750:
2742:
2740:
2732:
2730:
2722:
2720:
2712:
2710:
2702:
2700:
2688:
2686:
2674:
2672:
2660:
2659:
2657:
2653:
2649:
2642:
2637:
2635:
2630:
2628:
2623:
2622:
2619:
2607:
2604:
2603:
2601:
2599:
2595:
2589:
2587:
2578:
2576:
2559:
2557:
2545:
2543:
2531:
2529:
2521:
2519:
2507:
2505:
2478:
2476:
2464:
2462:
2454:
2453:
2451:
2449:
2445:
2439:
2431:
2429:
2413:
2412:
2410:
2406:
2400:
2392:
2390:
2382:
2380:
2372:
2370:
2362:
2360:
2352:
2350:
2347:
2346:
2344:
2340:
2334:
2326:
2324:
2316:
2314:
2302:
2300:
2292:
2290:
2282:
2280:
2268:
2266:
2250:
2248:
2236:
2234:
2226:
2224:
2212:
2210:
2202:
2200:
2188:
2186:
2183:
2181:
2144:
2142:
2139:
2138:
2136:
2134:Chromium(III)
2132:
2126:
2114:
2113:
2111:
2107:
2097:
2081:
2080:
2078:
2074:
2068:
2060:
2058:
2050:
2048:
2040:
2038:
2030:
2028:
2025:
2023:
2015:
2013:
2010:
2008:
2005:
2003:
1995:
1993:
1977:
1975:
1962:
1960:
1948:
1946:
1938:
1937:
1935:
1931:
1921:
1897:
1896:
1894:
1890:
1884:
1881:
1880:
1878:
1874:
1865:
1849:
1847:
1831:
1829:
1825:
1819:
1811:
1810:
1808:
1804:
1800:
1793:
1788:
1786:
1781:
1779:
1774:
1773:
1770:
1764:
1761:
1759:
1756:
1755:
1744:
1742:
1738:
1733:
1729:
1717:on 2020-03-17
1713:
1709:
1703:
1696:
1695:
1690:
1684:
1676:
1672:
1666:
1658:
1652:
1648:
1644:
1637:
1629:
1623:
1619:
1615:
1611:
1604:
1602:
1593:
1587:
1583:
1582:
1574:
1572:
1563:
1561:0-12-352651-5
1557:
1553:
1546:
1538:
1534:
1530:
1526:
1518:
1511:
1506:
1498:
1492:
1488:
1484:
1480:
1474:
1466:
1462:
1458:
1454:
1450:
1446:
1442:
1438:
1434:
1427:
1419:
1413:
1409:
1405:
1401:
1395:
1391:
1378:
1372:
1368:
1358:
1355:
1354:
1348:
1346:
1342:
1341:nasal sinuses
1338:
1334:
1330:
1329:chromium (VI)
1326:
1322:
1318:
1314:
1310:
1305:
1295:
1293:
1289:
1281:
1276:
1230:
1229:
1228:
1218:
1214:
1202:
1195:
1191:
1186:
1177:
1175:
1172:
1168:
1167:chrome yellow
1164:
1160:
1156:
1152:
1148:
1141:
1140:Chrome yellow
1137:
1133:
1118:
1065:
1064:
1063:
1061:
1050:
1019:
1018:
1017:
1015:
1011:
1001:
998:
993:
982:
945:
944:
943:
940:
915:
914:
913:
911:
907:
893:
847:
846:
845:
836:
805:
804:
803:
801:
792:
786:
761:
736:
731:
729:
725:
689:
688:
687:
685:
677:
673:
664:
654:
650:
646:
634:
618:
614:
606:
600:
595:
592:
586:
581:
580:
574:
572:
569:
565:
561:
557:
553:
528:
512:
500:
494:
488:
485:
482:
480:
477:
476:
472:
470:
467:
466:
430:
427:
423:
422:
417:
408:
403:
402:
399:
392:
378:
368:
367:
364:
357:
350:
349:DTXSID5074004
345:
342:
341:DTXSID7065675
337:
336:
334:
324:
320:
319:
312:
307:
304:
299:
298:
296:
294:
291:
290:
283:
278:
275:
270:
269:
267:
261:
257:
256:
249:
244:
243:
241:
239:
236:
235:
228:
223:
220:
215:
214:
212:
210:
207:
206:
199:
194:
191:
186:
185:
183:
179:
174:
173:
166:
161:
158:
153:
152:
150:
147:
143:
142:
137:
129:
125:
120:
110:
106:
101:
97:
96:
92:
82:
78:
73:
69:
68:
64:
56:
52:
48:
44:
40:
33:
19:
3077:
2647:
2605:
2579:
2448:Chromium(VI)
2342:Chromium(IV)
1933:Chromium(II)
1740:
1736:
1727:
1725:
1719:. Retrieved
1712:the original
1693:
1683:
1674:
1665:
1646:
1636:
1609:
1580:
1551:
1545:
1528:
1524:
1517:
1505:
1482:
1473:
1440:
1436:
1426:
1403:
1394:
1371:
1321:IARC Group 1
1317:carcinogenic
1307:
1277:
1273:
1198:
1155:heavy metals
1144:
1127:Applications
1116:
1057:
1048:
1007:
999:
991:
989:
980:
941:
938:
905:
892:chromic acid
888:
843:
834:
793:
759:
735:trichromates
734:
732:
721:
700:+ 2 H ⇌ Cr
681:
678:for chromate
610:
526:
510:
509:
484:Chromic acid
139:Identifiers
55:tetrachromat
18:Chromate ion
2964:Dichromates
2408:Chromium(V)
1876:Chromium(I)
1806:Chromium(0)
1325:lung cancer
1267:+ 8 CO
1259:+ 2 Fe
1251:→ 8 Na
1239:+ 8 Na
1231:4 FeCr
1159:lanthanides
1138:painted in
550:. They are
419:Properties
227:CHEBI:33141
219:CHEBI:35404
43:monochromat
3145:Categories
1721:2020-01-05
1643:"Chromite"
1627:3527306730
1525:Polyhedron
1387:References
1247:+ 7 O
1136:School bus
902:, but the
783:. All poly
558:in the +6
527:Dichromate
469:Molar mass
311:9LKY4BFN2V
303:9S2Y101D6M
176:3D model (
165:13907-47-6
157:13907-45-4
146:CAS Number
51:trichromat
3151:Chromates
2655:Chromates
1726:There is
1691:(2012) .
1465:231808339
1457:2196-7105
1194:Australia
800:weak acid
785:oxyanions
552:oxyanions
47:dichromat
2939:(OSi(OCH
2171:][CH
1351:See also
1298:Toxicity
1292:lópezite
1280:Crocoite
1201:chromite
1190:Tasmania
1090:+ 3 e →
1014:reducing
653:pyridine
645:covalent
617:peroxide
571:solution
566:. In an
556:chromium
511:Chromate
238:DrugBank
32:Chromate
3078:Related
3026:[NH
2841:[NH
2147:[Cr
1335:of the
1282:, PbCrO
925:⇌ HCrO
848:2
798:, is a
635:, CrO(O
568:aqueous
463:
260:PubChem
248:DB14182
3048:[C
2917:(OC(CH
2889:NH]CrO
2881:[C
2570:NH]CrO
2562:[C
2305:Cr(ClO
2229:Cr(OH)
1814:Cr(CO)
1704:
1653:
1624:
1588:
1558:
1493:
1463:
1455:
1414:
1333:cancer
1203:, FeCr
1092:Cr(OH)
758:, and
398:SMILES
122:Names
3116:EuCrO
2817:PbCrO
2807:CdCrO
2797:ZnCrO
2773:NiCrO
2745:BaCrO
2735:SrCrO
2725:CaCrO
2715:MgCrO
2705:BeCrO
2467:CrO(O
2191:Cr(NO
1980:Cr(CH
1715:(PDF)
1698:(PDF)
1461:S2CID
1363:Notes
1313:toxic
1171:redox
990:The p
960:⇌ Cr
691:2 CrO
649:ether
363:InChI
282:24503
274:24461
209:ChEBI
178:JSmol
53:, or
2870:KCrO
2759:(CrO
2524:CrOF
2420:Cr(O
2385:CrBr
2375:CrCl
2349:CrSi
2319:CrBr
2295:CrCl
2205:CrPO
2151:O(CH
2084:Cr(C
2053:CrBr
2043:CrCl
2027:CrSe
2018:CrSO
1998:CrSi
1965:Cr(C
1916:(CO)
1860:(CO)
1834:Cr(C
1702:ISBN
1689:IARC
1651:ISBN
1622:ISBN
1586:ISBN
1556:ISBN
1491:ISBN
1453:ISSN
1412:ISBN
1339:and
1337:nose
1315:and
1215:and
1161:and
1102:+ 5
1077:+ 4
850:HCrO
829:+ H;
807:HCrO
722:The
663:py.
442:and
293:UNII
3126:CrO
3091:CrO
3056:NH]
2935:CrO
2913:CrO
2849:CrO
2831:CrO
2787:CrO
2695:CrO
2681:CrO
2667:CrO
2582:CrF
2548:CrO
2534:CrO
2510:CrO
2485:CrO
2457:CrO
2434:CrF
2395:CrI
2365:CrF
2355:CrO
2329:CrI
2285:CrF
2257:(SO
2185:CrN
2141:CrB
2063:CrI
2033:CrF
2012:CrS
2007:CrO
1941:CrH
1883:CrH
1852:CrC
1614:doi
1533:doi
1445:doi
1441:236
1255:CrO
1067:CrO
973:+ H
947:HCr
934:+ H
921:CrO
898:CrO
894:, H
881:+ H
819:CrO
713:+ H
554:of
515:CrO
432:CrO
328:EPA
263:CID
3147::
3102:Cr
3060:Cr
3034:Cr
3012:Cr
3008:Ag
2994:Cr
2976:Cr
2972:Na
2893:Cl
2874:Cl
2827:Pb
2783:Ag
2755:Fe
2691:Cs
2663:Na
2574:Cl
2552:Br
2538:Cl
2496:Cr
2275:Te
2271:Cr
2253:Cr
2239:Cr
2215:Cr
2175:CO
2167:O)
2163:(H
2155:CO
2117:Cr
1984:CO
1951:Cr
1904:(C
1900:Cr
1724:.
1673:.
1645:.
1620:.
1600:^
1570:^
1529:16
1527:.
1459:.
1451:.
1439:.
1435:.
1347:.
1243:CO
1227::
1192:,
1157:,
1104:OH
1021:Cr
967:2−
862:Cr
860:⇌
817:⇌
802::
778:13
764:Cr
762:,
753:10
739:Cr
737:,
728:pH
707:2−
694:2−
686:.
619:,
531:Cr
525:.
444:Cr
49:,
45:,
41:,
3128:5
3118:4
3108:7
3106:O
3104:2
3100:2
3098:H
3096:/
3093:4
3089:2
3087:H
3066:7
3064:O
3062:2
3058:2
3054:5
3052:H
3050:5
3040:7
3038:O
3036:2
3032:2
3030:]
3028:4
3018:7
3016:O
3014:2
3010:2
3000:7
2998:O
2996:2
2992:2
2990:K
2982:7
2980:O
2978:2
2974:2
2949:2
2947:)
2945:3
2943:)
2941:3
2937:2
2927:2
2925:)
2923:3
2921:)
2919:3
2915:2
2891:3
2887:5
2885:H
2883:5
2872:3
2851:4
2847:2
2845:]
2843:4
2833:5
2829:2
2819:4
2809:4
2799:4
2789:4
2785:2
2775:4
2765:3
2763:)
2761:4
2757:2
2747:4
2737:4
2727:4
2717:4
2707:4
2697:4
2693:2
2683:4
2679:2
2677:K
2669:4
2665:2
2640:e
2633:t
2626:v
2584:6
2572:3
2568:5
2566:H
2564:5
2554:2
2550:2
2540:2
2536:2
2526:4
2516:2
2514:F
2512:2
2502:7
2500:O
2498:2
2494:2
2492:H
2490:/
2487:4
2483:2
2481:H
2473:2
2471:)
2469:2
2459:3
2436:5
2426:4
2424:)
2422:2
2418:3
2416:K
2397:4
2387:4
2377:4
2367:4
2357:2
2331:3
2321:3
2311:3
2309:)
2307:4
2297:3
2287:3
2277:3
2273:2
2263:3
2261:)
2259:4
2255:2
2245:3
2243:S
2241:2
2231:3
2221:3
2219:O
2217:2
2207:4
2197:3
2195:)
2193:3
2179:]
2177:2
2173:3
2169:3
2165:2
2161:6
2159:)
2157:2
2153:3
2149:3
2123:2
2121:C
2119:3
2094:2
2092:)
2090:5
2088:H
2086:5
2065:2
2055:2
2045:2
2035:2
2020:4
2000:2
1990:2
1988:)
1986:2
1982:3
1973:)
1971:4
1969:O
1967:2
1957:2
1955:C
1953:3
1943:2
1918:6
1914:2
1912:)
1910:5
1908:H
1906:5
1902:2
1862:3
1858:6
1856:H
1854:6
1844:2
1842:)
1840:6
1838:H
1836:6
1816:6
1791:e
1784:t
1777:v
1743:.
1677:.
1659:.
1630:.
1616::
1594:.
1565:.
1539:.
1535::
1499:.
1467:.
1447::
1420:.
1319:(
1284:4
1269:2
1265:3
1263:O
1261:2
1257:4
1253:2
1249:2
1245:3
1241:2
1237:4
1235:O
1233:2
1225:3
1223:O
1221:2
1209:4
1207:O
1205:2
1120:0
1117:ε
1097:3
1088:O
1084:2
1079:H
1072:4
1052:0
1049:ε
1044:O
1042:2
1035:7
1030:O
1026:2
995:a
992:K
984:a
981:K
979:p
975:,
970:7
964:O
962:2
957:7
954:−
951:O
949:2
931:4
928:−
923:4
919:2
917:H
909:a
906:K
904:p
900:4
896:2
885:O
883:2
876:7
871:O
867:2
855:4
838:a
835:K
833:p
824:4
812:4
796:4
789:4
773:O
769:4
748:O
744:3
717:O
715:2
710:7
704:O
702:2
697:4
661:2
659:)
657:2
641:2
639:)
637:2
626:2
621:O
545:7
540:O
536:2
520:4
458:7
453:O
449:2
437:4
330:)
326:(
180:)
57:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.