65:
112:
104:
135:
These compounds dissolve in polar solvents to give ionic solutions that contain highly solvated anions and cations. Alkali halides dissolve large amounts of the corresponding alkali metal: caesium is completely miscible at all temperatures above the melting point.
139:
The table below provides links to each of the individual articles for these compounds. The numbers beside the compounds show the electronegativity difference between the elements based on the
128:
solids, although as finely ground powders appear white. They melt at high temperature, usually several hundred degrees to colorless liquids. Their high melting point reflects their high
423:
53:. These compounds are the often commercially significant sources of these metals and halides. The best known of these compounds is
375:
358:
89:
353:
Greenwood, N. N.; & Earnshaw, A. (1997). Chemistry of the
Elements (2nd Edn.), Oxford:Butterworth-Heinemann.
405:
107:
Ball-and-stick model of the coordination of Na and Cl in NaCl. Most alkali metal halides adopt this structure.
448:
17:
100:
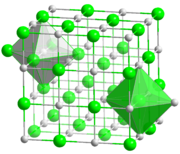
lattice that accommodates coordination number of eight for the larger metal cation (and the anion also).
132:. At still higher temperatures, these liquids evaporate to give gases composed of diatomic molecules.
397:
390:
438:
8:
97:
93:
85:
246:
209:
38:
453:
401:
371:
354:
283:
252:
215:
443:
320:
289:
258:
234:
221:
197:
42:
326:
295:
271:
240:
203:
72:
54:
458:
332:
308:
277:
129:
432:
314:
140:
370:
Wells, A.F. (1984) Structural
Inorganic Chemistry, Oxford: Clarendon Press.
46:
125:
58:
170:
27:
Family of compounds consisting of an alkali metal bonded to a halogen
229:
192:
175:
64:
392:
Inorganic chemistry : principles of structure and reactivity
266:
180:
160:
111:
103:
50:
96:
of six. Caesium chloride, bromide, and iodide crystallize in a
88:
lattices. In this structure both the metals and halides feature
388:
Huheey, James E.; Keiter, Ellen A.; Kieter, Richard L. (1993).
303:
165:
68:
115:
Ball-and-stick model of the coordination of Cs and Cl in CsCl
396:(4. ed.). Cambridge, Massachusetts : Harper. pp.
143:. The higher the number is, the more ionic the solid is.
424:
Tastes of the alkali metal halides (except fluorides)
389:
387:
430:
84:Most alkali metal halides crystallize with the
124:The alkali metal halides exist as colourless
110:
102:
63:
14:
431:
151:
381:
24:
417:
25:
470:
90:octahedral coordination geometry
364:
347:
13:
1:
341:
119:
79:
7:
10:
475:
92:, in which each ion has a
186:
154:
71:is the mineral form of
116:
108:
76:
114:
106:
67:
449:Alkali metal halides
37:, are the family of
31:Alkali metal halides
98:body-centered cubic
94:coordination number
86:face-centered cubic
39:inorganic compounds
117:
109:
77:
45:MX, where M is an
339:
338:
16:(Redirected from
466:
412:
411:
395:
385:
379:
368:
362:
351:
149:
148:
130:lattice energies
43:chemical formula
21:
474:
473:
469:
468:
467:
465:
464:
463:
429:
428:
420:
418:Further reading
415:
408:
386:
382:
369:
365:
352:
348:
344:
189:
146:
122:
82:
73:sodium chloride
55:sodium chloride
28:
23:
22:
15:
12:
11:
5:
472:
462:
461:
456:
451:
446:
441:
427:
426:
419:
416:
414:
413:
406:
380:
363:
345:
343:
340:
337:
336:
330:
324:
318:
312:
306:
300:
299:
293:
287:
281:
275:
269:
263:
262:
256:
250:
244:
238:
232:
226:
225:
219:
213:
207:
201:
195:
190:
187:
184:
183:
178:
173:
168:
163:
157:
156:
155:Alkali metals
153:
121:
118:
81:
78:
35:alkali halides
26:
9:
6:
4:
3:
2:
471:
460:
457:
455:
452:
450:
447:
445:
442:
440:
439:Alkali metals
437:
436:
434:
425:
422:
421:
409:
403:
399:
394:
393:
384:
377:
376:0-19-855370-6
373:
367:
360:
359:0-7506-3365-4
356:
350:
346:
334:
331:
328:
325:
322:
319:
316:
313:
310:
307:
305:
302:
301:
297:
294:
291:
288:
285:
282:
279:
276:
273:
270:
268:
265:
264:
260:
257:
254:
251:
248:
245:
242:
239:
236:
233:
231:
228:
227:
223:
220:
217:
214:
211:
208:
205:
202:
199:
196:
194:
191:
185:
182:
179:
177:
174:
172:
169:
167:
164:
162:
159:
158:
150:
147:
144:
142:
141:Pauling scale
137:
133:
131:
127:
113:
105:
101:
99:
95:
91:
87:
74:
70:
66:
62:
60:
56:
52:
48:
44:
40:
36:
32:
19:
18:Alkali halide
391:
383:
366:
349:
145:
138:
134:
123:
83:
47:alkali metal
34:
30:
29:
126:crystalline
49:and X is a
433:Categories
407:006042995X
342:References
120:Properties
59:table salt
171:Potassium
80:Structure
41:with the
454:Crystals
230:Chlorine
193:Fluorine
188:Halogens
176:Rubidium
444:Halides
267:Bromine
181:Caesium
161:Lithium
152:
51:halogen
404:
374:
357:
335:(1.8)
323:(1.7)
304:Iodine
298:(2.1)
286:(2.0)
261:(2.3)
249:(2.2)
224:(3.3)
212:(3.2)
166:Sodium
69:Halite
459:Salts
329:(1.7)
317:(1.6)
311:(1.5)
292:(2.0)
280:(1.9)
274:(1.8)
255:(2.2)
243:(2.1)
237:(2.0)
218:(3.2)
206:(3.1)
200:(3.0)
33:, or
402:ISBN
372:ISBN
355:ISBN
296:CsBr
290:RbBr
278:NaBr
272:LiBr
259:CsCl
253:RbCl
241:NaCl
235:LiCl
398:377
333:CsI
327:RbI
315:NaI
309:LiI
284:KBr
247:KCl
222:CsF
216:RbF
204:NaF
198:LiF
61:.
435::
400:.
321:KI
210:KF
57:,
410:.
378:.
361:.
75:.
20:)
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.