111:
57:
260:
Magnus, Philip; Turnbull, Rachel (2006). "Thermal and Acid-Catalyzed
Hofmann–Martius Rearrangement of 3-N-Aryl-2-oxindoles into 3-(Arylamino)-2-oxindoles".
110:
86:
centers around dissociation of the reactant with the positively charged organic residue R attacking the aniline ring in a
56:
318:
145:
79:
134:
87:
122:
118:
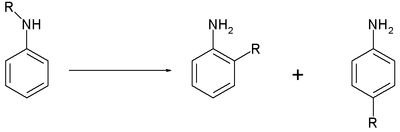
33:
37:
74:
The reaction is also known to work for aryl ethers and two conceptually related reactions are the
25:
231:"XV.—Intramolecular rearrangement of the alkylarylamines: Formation of 4-amino-n-butylbenzene"
68:
8:
140:
75:
83:
277:
48:
21:
269:
242:
211:
180:
40:
312:
215:
184:
281:
246:
273:
230:
199:
168:
63:
When the catalyst is a metal halide the reaction is also called the
44:
299:
29:
228:
166:
43:-alkylated aniline. The reaction requires heat, and the
93:
In one study this rearrangement was applied to a 3-N(CH
302:at 80 °C gives 30% 2-o (ortho) and 37% 2-p (para)
310:
259:
204:Berichte der Deutschen Chemischen Gesellschaft
197:
173:Berichte der Deutschen Chemischen Gesellschaft
117:The reaction is named after German chemists
229:Reilly, J.; Hickinbottom, W. J. (1920).
169:"Methylirung der Phenylgruppe im Anilin"
167:Hofmann, A. W.; Martius, C. A. (1871).
311:
200:"Umwandlung des Anilins in Toluidin"
13:
14:
330:
65:Reilly–Hickinbottom rearrangement
109:
55:
288:
253:
222:
191:
160:
1:
153:
18:Hofmann–Martius rearrangement
7:
128:
10:
335:
146:Fischer–Hepp rearrangement
123:Carl Alexander von Martius
119:August Wilhelm von Hofmann
80:Fischer–Hepp rearrangement
28:converting an N-alkylated
135:Friedel–Crafts alkylation
88:Friedel–Crafts alkylation
216:10.1002/cber.18720050241
185:10.1002/cber.18710040271
319:Rearrangement reactions
198:Hofmann, A. W. (1872).
26:rearrangement reaction
32:to the corresponding
247:10.1039/ct9201700103
71:and Joseph Reilly).
69:Wilfred Hickinbottom
141:Fries rearrangement
76:Fries rearrangement
84:reaction mechanism
274:10.1021/ol061191z
137:-like reactions:
49:hydrochloric acid
22:organic chemistry
326:
303:
292:
286:
285:
257:
251:
250:
226:
220:
219:
195:
189:
188:
164:
113:
59:
47:is an acid like
334:
333:
329:
328:
327:
325:
324:
323:
309:
308:
307:
306:
293:
289:
262:Organic Letters
258:
254:
227:
223:
196:
192:
165:
161:
156:
131:
104:
100:
96:
12:
11:
5:
332:
322:
321:
305:
304:
287:
268:(16): 3497–9.
252:
221:
210:(2): 720–722.
190:
158:
157:
155:
152:
151:
150:
149:
148:
143:
130:
127:
115:
114:
105:)-2-oxindole:
102:
98:
94:
61:
60:
9:
6:
4:
3:
2:
331:
320:
317:
316:
314:
301:
297:
291:
283:
279:
275:
271:
267:
263:
256:
248:
244:
240:
236:
232:
225:
217:
213:
209:
205:
201:
194:
186:
182:
178:
174:
170:
163:
159:
147:
144:
142:
139:
138:
136:
133:
132:
126:
124:
120:
112:
108:
107:
106:
91:
89:
85:
81:
77:
72:
70:
67:(named after
66:
58:
54:
53:
52:
50:
46:
42:
39:
35:
31:
27:
23:
19:
295:
290:
265:
261:
255:
238:
235:J. Chem. Soc
234:
224:
207:
203:
193:
176:
172:
162:
116:
92:
73:
64:
62:
17:
15:
241:: 103–137.
179:(2): 742.
154:References
36:and / or
313:Category
294:heating
282:16869644
129:See also
78:and the
45:catalyst
300:toluene
30:aniline
280:
82:. Its
34:ortho
24:is a
278:PMID
121:and
41:aryl
38:para
16:The
298:in
270:doi
243:doi
239:117
212:doi
181:doi
97:)(C
20:in
315::
276:.
264:.
237:.
233:.
206:.
202:.
175:.
171:.
125:.
90:.
51:.
296:1
284:.
272::
266:8
249:.
245::
218:.
214::
208:5
187:.
183::
177:4
103:5
101:H
99:6
95:3
Text is available under the Creative Commons Attribution-ShareAlike License. Additional terms may apply.